The treatment landscape of HR-positive, HER2-negative metastatic breast cancer continues to evolve, particularly in the post–CDK4/6 inhibitor setting, where therapeutic options remain limited and resistance is common. In this context, atirmociclib, a next-generation CDK4 inhibitor, has demonstrated encouraging efficacy in the randomized Phase 2 FOURLIGHT-1 trial, positioning itself as a potential new strategy for patients progressing after prior CDK4/6 inhibitor therapy (Pfizer, 2026).
What Was The FOURLIGHT-1 Trial Designed to Answer?
The FOURLIGHT-1 study was designed to address a critical clinical question: can continued targeting of the cyclin-dependent kinase pathway provide benefit after progression on prior CDK4/6 inhibitor-based therapy? Unlike earlier trials conducted in treatment-naïve settings, this study specifically focused on patients whose disease had already demonstrated resistance to standard endocrine-based targeted therapy.
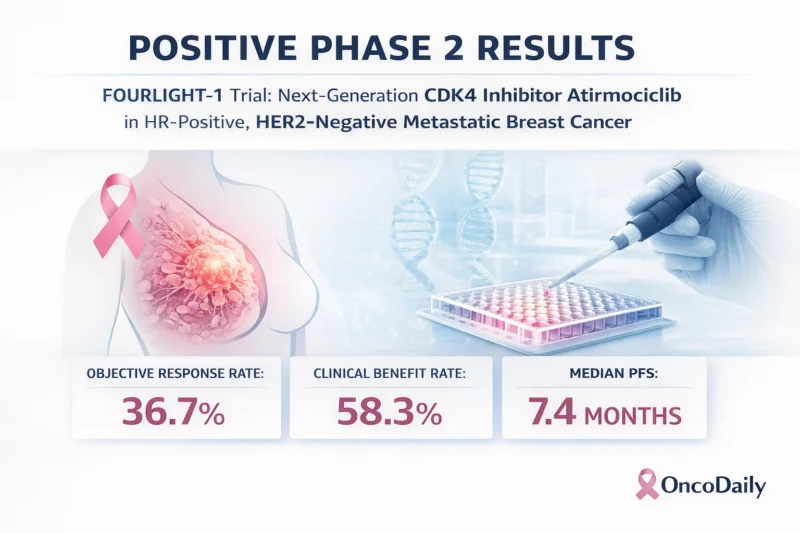
FOURLIGHT-1 was an interventional, randomized, open-label, multicenter Phase 2 trial, enrolling 264 patients across 14 countries with HR-positive, HER2-negative advanced or metastatic breast cancer (Pfizer, 2026). Patients were randomized to receive atirmociclib in combination with fulvestrant or physician-relevant comparator regimens, including fulvestrant alone or everolimus plus exemestane, reflecting real-world second-line treatment strategies.
The primary endpoint of the study was investigator-assessed progression-free survival, while secondary endpoints included overall survival, objective response rate, duration of response, and clinical benefit rate, ensuring a comprehensive evaluation of both disease control and long-term outcomes.
Atirmociclib Demonstrates Clinically Meaningful Benefit
The topline results of FOURLIGHT-1 demonstrated that atirmociclib significantly improved progression-free survival compared with control therapy. Pfizer reported a hazard ratio of 0.60, corresponding to a 40% reduction in the risk of disease progression or death, with a 95% confidence interval of 0.440 to 0.825 and a p-value of 0.0007 (Pfizer, 2026).
For patients who have already received a CDK4/6 inhibitor, these findings are particularly important. This setting is often characterized by diminishing endocrine sensitivity and limited targeted options. The magnitude of benefit observed in FOURLIGHT-1 suggests that atirmociclib may re-establish disease control even after prior CDK4/6 exposure, raising important questions about the biology of resistance and pathway re-targeting.
An additional layer of clinical relevance comes from the patient population itself. According to Pfizer, more than 90% of patients initiated atirmociclib within three months of their last CDK4/6 inhibitor therapy, indicating that the trial reflects an early post-progression setting rather than a heavily pretreated cohort.
Consistency Across Clinically Relevant Subgroups
One of the most notable aspects of the FOURLIGHT-1 results is the consistency of benefit across predefined subgroups. Pfizer reported that the progression-free survival advantage with atirmociclib was observed regardless of performance status, menopausal status, presence of visceral metastases, duration of prior CDK4/6 inhibitor exposure, and the specific CDK4/6 inhibitor previously used (Pfizer, 2026).
This consistency strengthens the overall interpretation of the trial and suggests that the activity of atirmociclib is not confined to a narrow subset of patients. In clinical practice, where heterogeneity is the rule rather than the exception, such broad applicability is particularly valuable.
Safety Profile and Tolerability
Beyond efficacy, tolerability remains a critical determinant of clinical adoption. In FOURLIGHT-1, atirmociclib was reported to have a manageable safety profile, with 6.4% of patients discontinuing treatment due to adverse events. Importantly, no new safety signals were identified, and the overall safety findings were consistent with previous studies of the drug (Pfizer, 2026).
Although detailed safety data are not yet available in the topline release, the relatively low discontinuation rate provides an early signal that atirmociclib may offer a favorable balance between efficacy and tolerability, a key consideration in long-term metastatic treatment strategies.
Positioning Atirmociclib in the Treatment Landscape
Pfizer describes atirmociclib as an investigational oral CDK4-selective inhibitor, developed as a potential next-generation therapy for HR-positive, HER2-negative breast cancer (Pfizer, 2026). Its selective targeting of CDK4, rather than broader CDK4/6 inhibition, may represent a strategic attempt to refine efficacy while potentially improving tolerability, although detailed mechanistic data were not included in the current release.
Importantly, the development program for atirmociclib extends beyond FOURLIGHT-1. Pfizer noted that a Phase 3 registrational trial in the first-line metastatic setting is ongoing, and a Phase 2 neoadjuvant study in early breast cancer is planned for future presentation.
What Comes Next?
While the FOURLIGHT-1 topline results provide a strong signal of activity, several key questions remain. Overall survival data are not yet mature, with approximately 20% of events reported at the time of analysis, limiting conclusions about long-term benefit.
In addition, detailed efficacy endpoints such as objective response rates, duration of response, and resistance patterns will be critical for fully understanding how atirmociclib compares with existing post-CDK4/6 strategies. Full data presentation at a scientific congress or in a peer-reviewed publication will be essential to define its role in clinical practice.
A Step Forward in Post-CDK4/6 Breast Cancer?
Based on the currently available data, atirmociclib demonstrates a clinically meaningful progression-free survival benefit, consistent subgroup activity, and a manageable safety profile in patients with HR-positive, HER2-negative metastatic breast cancer following prior CDK4/6 inhibitor therapy (Pfizer, 2026).
FOURLIGHT-1 does not yet redefine the treatment paradigm, but it clearly introduces a promising new direction—one that challenges the assumption that CDK pathway inhibition loses relevance after initial resistance and instead suggests that refined, next-generation targeting may still provide benefit in later lines of therapy.


