Novartis is acquiring Synnovation Therapeutics’ next-generation PI3Kα inhibitor program for $2 billion upfront, a bold move to strengthen its breast cancer pipeline. The lead drug (SNV4818) is a mutant-selective PI3Kα inhibitor designed to target PIK3CA-mutant cancer cells while sparing healthy cells, potentially enabling a safer triple-combination therapy in hormone receptor-positive, HER2-negative (HR+/HER2-) breast cancer. The deal, announced March 20, 2026, underscores Novartis’s strategy to deliver better efficacy with fewer side effects by reviving a triple-therapy approach that earlier drugs couldn’t tolerate.
Deal Overview: $2B Upfront to Bolster Novartis’s Breast Cancer Arsenal
Novartis will pay $2 billion in cash upfront (plus up to $1 billion in future milestones) to acquire Pikavation Therapeutics, a Synnovation subsidiary developing pan–mutant-selective PI3Kα inhibitors. The crown jewel is SNV4818, an oral PI3Kα inhibitor in Phase 1/2 trials. By absorbing this program, Novartis aims to defend its HR+ breast cancer franchise amid intensifying competition.
The Swiss pharma already markets Piqray (alpelisib), the first PI3Kα drug approved in HR+ breast cancer (2019), and Kisqali (ribociclib), a leading CDK4/6 inhibitor. But Piqray’s sales have been constrained by side effects and a new rival from Roche (Itovebi) that moved into earlier-line therapy. By acquiring SNV4818, Novartis positions itself to potentially introduce a next-generation therapy that could be combined with Kisqali and endocrine treatment, further strengthening its leadership in PIK3CA-mutant breast cancer, pending successful clinical outcomes.
Wenqing Yao, Ph.D., CEO of Synnovation Therapeutics, highlighted the potential of the partnership, stating,
We believe Novartis’s global capabilities and commitment to patients with cancer will accelerate the development of SNV4818 for patients with PI3Kα mutation–driven solid tumors beyond what Synnovation could achieve alone.

Wenqing Yao/Philadelphia Business Journal
Under the terms of the agreement, Novartis will assume global responsibility for the development and commercialization of SNV4818, while Synnovation continues advancing its broader oncology pipeline. The transaction is expected to close in the first half of 2026, subject to regulatory approvals.
Mutant-Selective PI3Kα: A Fix for Toxicity, Key to Triple Therapy
Older PI3Kα inhibitors have been life-extending but hard to tolerate. Novartis’s Piqray and Roche’s new Itovebi (inavolisib) both block PI3Kα broadly (mutant and wild-type), which can trigger severe hyperglycemia, rash, and GI side effects by disrupting normal insulin signaling. These toxicities often force dose reductions and have prevented using PI3K inhibitors alongside other therapies. Notably, a Novartis trial of triple therapy, combining Piqray + Kisqali + fulvestrant, was halted when over half of patients required dose cuts due to side effects. This derailed hopes of a powerful three-drug regimen.
SNV4818’s design directly tackles this challenge. It is engineered to hit only the mutant PI3Kα that drives cancer growth, sparing the wild-type enzyme in healthy tissues. By avoiding off-target toxicity, SNV4818 aims to allow full-strength dosing and long-term use, even in combination with hormonal and CDK4/6 therapies. Preclinical tests showed strong activity against common PIK3CA mutations and minimal impact on normal PI3Kα.
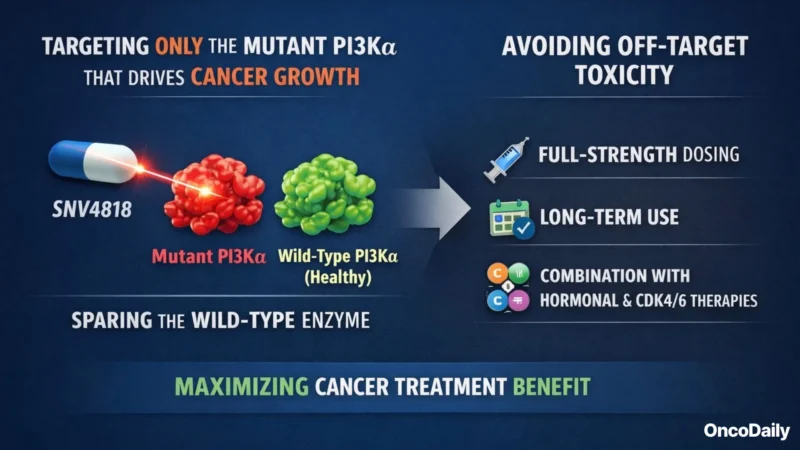
Shreeram Aradhye, M.D., Novartis’s President of Development, explained that:
By focusing on the mutated form in tumors, SNV4818 aims to reduce unwanted side effects, support more consistent dosing, and make it easier to combine with hormonal therapy and other treatments earlier in care.

Shreeram Aradhye/Novartis
In short, mutant-selective targeting could unlock the long-sought triple therapy approach by making it tolerable for patients.
How a Safer PI3Kα Inhibitor Could Benefit Patients:
- Prolonged Disease Control: Triple blockade of ER, CDK4/6, and PI3Kα could significantly delay tumor progression, as hinted by prior trials (adding first-gen Piqray doubled median PFS from ~5.7 to 11 months in resistant cases). With better safety, more patients might stay on treatment long enough to realize this benefit.
- Earlier Use of Triple Therapy: Currently, PI3K inhibitors are mostly used in later lines due to toxicity. A cleaner drug could be moved up to first-line therapy, attacking tumor mutations before resistance emerges. Roche’s inavolisib already showed that adding a PI3Kα inhibitor up front can extend PFS to ~15 months vs 7.3 months on standard therapy. SNV4818 might replicate or exceed that, if it’s well tolerated.
- Fewer Interruptions & Hospital Visits: By reducing severe side effects like high blood sugar, SNV4818 may lessen the need for intervention (e.g., insulin for hyperglycemia) and allow patients to stay at optimal doses. This improves quality of life and outcomes.
Data Snapshot & Development Timeline
Ongoing Phase 1/2 Trial: SNV4818 is being evaluated in an open-label Phase 1/2 study (NCT06736704) for patients with PIK3CA-mutant cancers, including metastatic HR+/HER2- breast cancer. Initiated in early 2025, the trial has multiple arms: SNV4818 monotherapy, and SNV4818 combined with fulvestrant (AstraZeneca’s Faslodex) and palbociclib (Pfizer’s Ibrance). These combinations mirror the standard care for HR+ breast cancer, testing whether adding SNV4818 is safe and determines an optimal dose. Primary endpoints focus on safety/tolerability and identifying a recommended Phase 2 dose, while secondary outcomes will look for signs of anti-tumor activity. The estimated primary completion is April 2027, meaning initial efficacy data could emerge in late 2026 or 2027.
Novartis’s Plans: Once the acquisition closes, Novartis will likely expand SNV4818 trials swiftly if Phase 1 data are promising. Analysts expect a Phase 3 trial in first-line PIK3CA-mutant breast cancer (adding SNV4818 to Kisqali + endocrine therapy) to start by 2027, positioning the drug for possible approval around 2029. Novartis noted that SNV4818 “fits naturally alongside CDK inhibitors as well as endocrine therapies as part of a potential combination regimen”, signaling its intent to test triple therapy head-on. The company also indicated interest in exploring SNV4818 in other solid tumors driven by PIK3CA mutations over time. For now, breast cancer remains the primary target and the area of greatest unmet need for this approach.
Clinical Benchmark, Piqray: The promise of SNV4818 builds on the trail blazed by Piqray. In the pivotal SOLAR-1 trial, Piqray (when added to fulvestrant) improved median progression-free survival to 11.0 months vs 5.7 months with endocrine therapy alone. This proved that inhibiting PI3Kα can thwart cancer growth in PIK3CA-mutant patients. However, real-world use of Piqray has been limited by side effects, in trials, nearly 37% of patients on Piqray suffered high-grade hyperglycemia, and many had dose interruptions. With SNV4818, Novartis aims to keep the efficacy while eliminating the worst toxicity, thereby achieving the full potential of PI3K inhibition.
Regulatory Outlook: Since SNV4818 is a novel compound in early trials, it has no special FDA designations yet (unlike FluoGuide’s FG001 which earned Fast Track). However, if Phase 2 results show clear benefit with improved safety, Novartis could seek Breakthrough Therapy status to expedite Phase 3 and review, especially given the large patient population (~40% of HR+/HER2- metastatic breast cancers carry PIK3CA mutations) and the need for better tolerated options.
Competitive Landscape: Next-Gen PI3K Inhibitors on the Rise
Novartis’s $2B bet comes as rivals race to develop their own PI3Kα-targeted therapies for breast cancer:
| Therapy (Company) | Mechanism | Status / Indication | Key Data / Notes |
| Piqray (Novartis) | Pan-PI3Kα inhibitor (mutant + wild-type) | Approved 2019 (2L HR+/HER2- MBC with PIK3CA mutation) | PFS +5.3 mo vs placebo (11.0 vs 5.7 mo in SOLAR-1). First PI3Kα inhibitor; significant hyperglycemia (~37% Grade 3/4) often limits dosing. |
| Itovebi (Roche) | Pan-PI3Kα inhibitor (non-selective) | Approved 2024 (1L HR+/HER2- MBC, PIK3CA-mutant) | PFS +7.7 mo vs placebo (15.0 vs 7.3 mo) with OS benefit. First PI3Kα in 1L; improved tolerability vs Piqray but still causes metabolic & GI side effects. |
| Tersolisib (Lilly) | Mutant-selective PI3Kα inhibitor | Phase 3 (1L HR+/HER2- MBC, PIK3CA-mutant; combo with CDK4/6 + endocrine) | Acquired 2025 (from Scorpion). Aim: frontline triple therapy; no clinical results released yet. Seen as a key competitor to Novartis’s program. |
| Zovegalisib (Relay) | Allosteric, mutant-selective PI3Kα inhibitor | Phase 3 (2L HR+/HER2- MBC, post-CDK4/6; with fulvestrant) | Received FDA Breakthrough Therapy status (2026). Targets mutant enzyme via unique binding site. Early trials showed promising safety/PK; positioned for second-line (Piqray’s niche). |
| SNV4818 (Novartis/Synnovation) | Pan–mutant-selective PI3Kα inhibitor | Phase 1/2 (PIK3CA-mutant solid tumors; HR+ MBC in combo with fulvestrant + palbociclib) | Designed to spare wild-type PI3Kα → reduced toxicity. Broadly active against PIK3CA mutations; aims to enable triple therapy in 1L HR+ breast cancer. Deal worth $2B upfront (Mar 2026) to acquire program. |
MBC = metastatic breast cancer; HR+ = hormone receptor-positive; HER2- = HER2-negative.
The market opportunity is substantial. With around 40% of advanced HR+ breast cancer patients having PIK3CA mutations, there is a large eligible population for these therapies. Roche’s confidence in Itovebi (projecting $2.3B peak sales) suggests that a best-in-class PI3Kα inhibitor could become a blockbuster. Novartis, by investing early in SNV4818, is positioning to capture that value if the drug lives up to its promise.
Key Takeaways
- $2B Acquisition to Reinforce Pipeline: Novartis is acquiring Synnovation’s PI3Kα inhibitor program (SNV4818) for $2 billion upfront + $1 billion in milestones. The deal secures a next-generation targeted therapy to strengthen Novartis’s breast cancer portfolio as older drugs face competition and patent risks.
- Mutant-Selective PI3Kα Inhibitor: SNV4818 is a mutant-selective PI3Kα inhibitor that attacks cancer-driving PIK3CA mutations but spares wild-type PI3Kα. This precision aims to reduce side effects (like hyperglycemia) that plague first-generation PI3K inhibitors, allowing patients to tolerate treatment better.
- Reviving Triple Therapy in HR+ Breast Cancer: By improving tolerability, SNV4818 could enable a potent triple therapy, combining endocrine therapy, a CDK4/6 inhibitor (e.g. Kisqali), and a PI3Kα inhibitor, in PIK3CA-mutant HR+/HER2- breast cancer. Novartis’s earlier attempt at triple therapy with Piqray failed due to toxicity, but a safer agent may unlock significant progression-free survival benefits seen in concept.
- Clinical Progress & Timeline: SNV4818 is in an ongoing Phase 1/2 trial (monotherapy and with fulvestrant + palbociclib) that began in 2025. Initial safety data will guide dose selection; primary completion is expected in 2027. Novartis plans to accelerate Phase 3 studies (potentially in first-line therapy) if early results are positive, aiming for approval by ~2029.
- Competitive Pressure: Novartis’s move is partly defensive, rivals are advancing their own PI3Kα inhibitors. Roche’s Itovebi (approved 1L in 2024) doubled PFS and shows an OS trend, grabbing the first-mover advantage. Eli Lilly’s tersolisib and Relay’s zovegalisib are in Phase 3 trials. Several biotechs (OnKure, Sensei, etc.) have early programs. Novartis paid a premium to ensure its leadership in the next wave of PI3K-targeted therapy.
- Strategic Importance for Novartis: This acquisition bolsters Novartis’s high-value HR+ breast cancer franchise. Kisqali (ribociclib) already leads in CDK4/6 inhibitors (2025 sales ~$4.8B), and Piqray established the PIK3CA-mutant niche. SNV4818 could protect and expand these revenues by offering a superior option once Piqray (which had ~$505M in 2023 sales) is outpaced. It also showcases Novartis’s commitment to precision oncology and willingness to invest heavily for innovation.
- Better Outcomes for Patients: If successful, SNV4818’s mutant-selective approach will mean more patients can safely receive PI3Kα inhibitors earlier in their treatment journey. This could translate into longer disease control and survival for the ~40% of metastatic HR+ breast cancer patients with PIK3CA mutations. Patients could benefit from the synergy of triple therapy without the burden of frequent high-grade side effects.
- Broader Implications: A tolerable PI3Kα inhibitor like SNV4818 may eventually find use beyond breast cancer as PIK3CA mutations appear in other tumors (e.g. endometrial, head & neck). While Novartis’s immediate focus is breast cancer, success here would validate the mutant-selective strategy and potentially open doors to treating PI3K-driven cancers more broadly with combination regimens. It would exemplify how refining a drug’s selectivity can resurrect a potent therapeutic approach that was previously limited by toxicity.
Read more biotech insights on OncoDaily Biotech.
Written by: Semiramida Nina Markosyan, Editor, OncoDaily Canada