Generate Biomedicines priced its U.S. initial public offering on February 26, 2026, selling 25 million shares at $16 each for gross proceeds of $400 million, with trading expected on the Nasdaq under the ticker “GENB.” In February 2026, the offering was described as the largest biotechnology IPO to date for the year and part of an unusually active month for biotech listings.
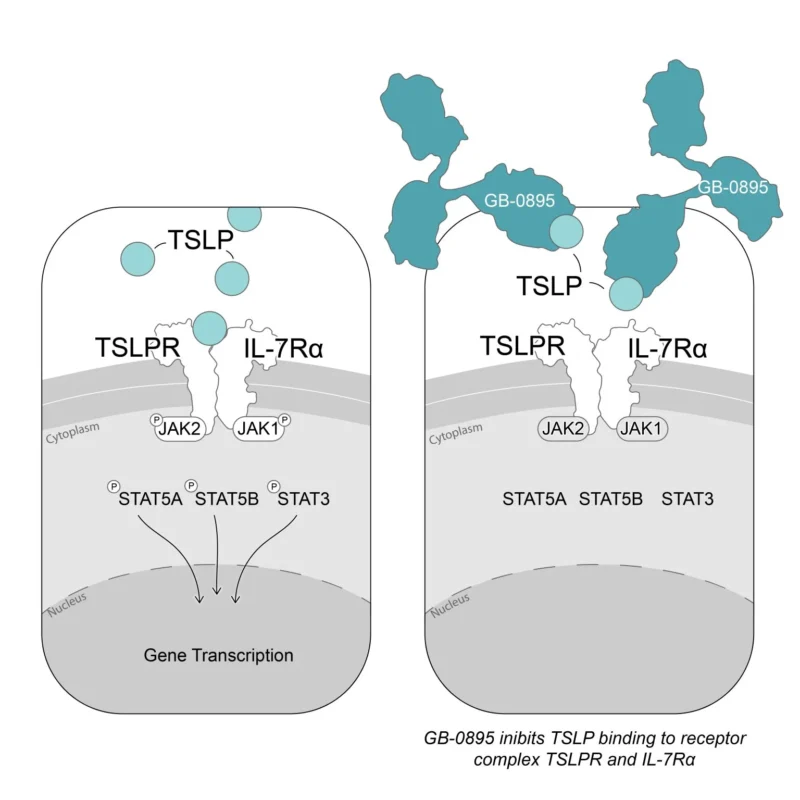
The company positions itself as an AI-driven drug developer focused on designing protein-based therapeutics, with a stated pipeline emphasis spanning immunology and oncology. The lead clinical program, GB-0895, targets thymic stromal lymphopoietin (TSLP) and is in Phase 3 studies for severe asthma, while an additional study evaluates GB-0895 in COPD.
Alexandra Snyder/LinkedIn
Although the near-term clinical value driver is not an oncology asset, the platform’s oncology applicability is central to how the company is being framed in capital markets coverage. On the oncology side, Generate lists preclinical-stage programs including:
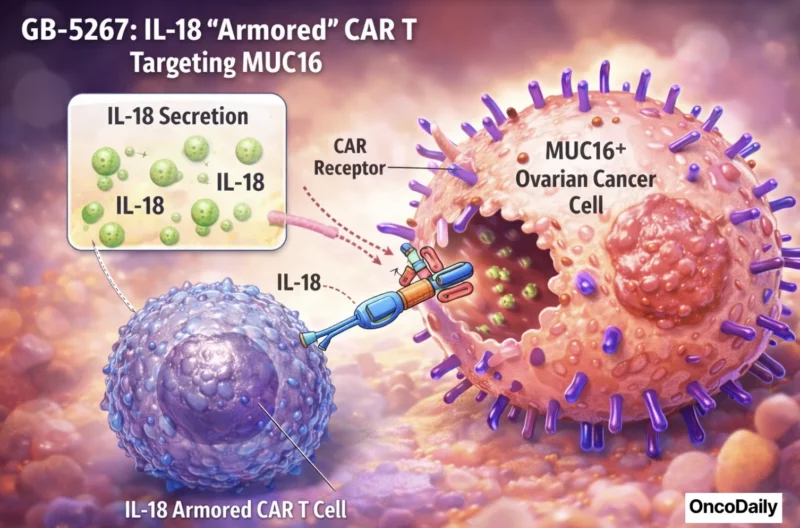
- GB-5267, an IL-18 “armored” CAR T targeting MUC16 for metastatic ovarian cancer (the company notes an IND “study may proceed” status in December 2025).
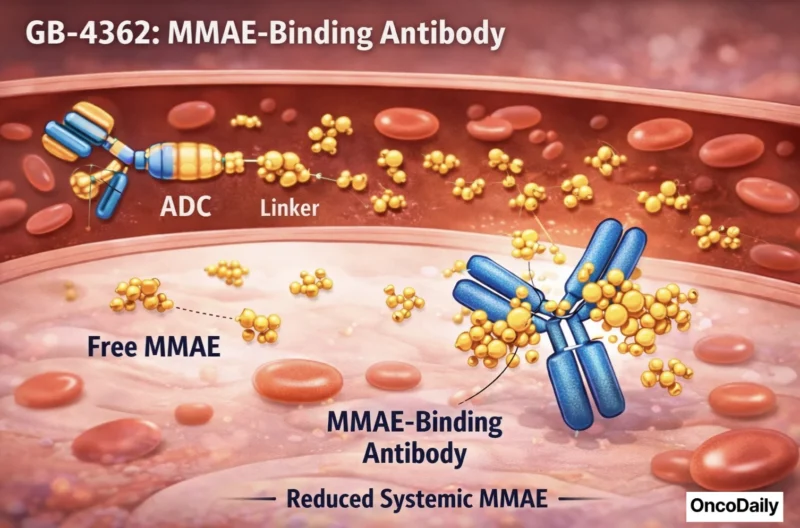
- GB-4362, an antibody designed to bind free MMAE (monomethyl auristatin E) to reduce systemic toxicities associated with MMAE-based antibody-drug conjugates when payload is released.
In addition to IPO proceeds, industry reporting cited more than $800 million in prior venture financing and approximately $110 million in collaboration payments from Amgen and Novartis tied to the company’s platform.
Why this matters in oncology
The IPO is a real-time signal about what public-market investors are currently willing to fund: platform narratives that claim to compress discovery timelines and generate multiple modalities, including assets relevant to cancer. At the same time, IPO coverage has highlighted a key diligence issue for AI-centric biotech valuations: industry observers still point out that a fully AI-discovered drug has not yet achieved U.S. Food and Drug Administration approval, leaving oncology investors and partners to judge platforms by translational performance rather than marketing claims.
Written by: Semiramida Nina Markosyan, Editor, OncoDaily Canada


