This week in oncology biotech, the conversation turned toward transformation — not just of science, but of the industry itself. As the field marks the 50th anniversary of the first biotech company, the deals and debates of the past seven days captured exactly what that half-century has built: an ecosystem bold enough to attempt reprogramming the immune system from inside the body.
Below are 10 featured posts from scientists, executives, and investors shaping the conversation in oncology and life sciences this week.
John F. Crowley – President & CEO, Biotechnology Innovation Organization
THE FUTURE OF BIOTECH…in studio early this morning with CNBC to discuss the 50th anniversary this month of the founding of the first biotech company. We discussed a range of topics and most importantly the real promise that biotech can deliver today and, in the years ahead so that increasingly a doctor when delivering a tough diagnosis does not have to say, “I am sorry, there is nothing we can do.”
With the explosion of biotechnologies that answer is simply no longer acceptable. We have real hope in so many areas- cancer, rare disease, autoimmune disease, brain diseases, and more. We just need to let biotech entrepreneurs flourish and the ecosystem do what it does best. And to make sure that there are no barriers to people accessing these medicines once approved. No one should ever go a day without the medicines they need….no one.
Spencer Knight – Business Development Director, Biotechnology at Hartmann Young
BREAKING NEWS:
Eli Lilly to Acquire In Vivo CAR-T Pioneer for $3.25 BillionEli Lilly and Company is acquiring Kelonia Therapeutics to advance next-generation in vivo CAR-T cancer treatments.
Key Highlights:
Deal structure: $3.25B upfront, total value up to $7B
KLN-1010: First-in-class in vivo CAR-T therapy in Phase 1 for multiple myeloma
Approach: Uses lentiviral gene delivery to reprogram T-cells directly inside the body, eliminating ex vivo manufacturing
Platform: iGPS® technology aims to enable scalable, off-the-shelf cell therapies across multiple diseases
Patient impact: Designed to reduce cost, time, and logistical barriers of traditional CAR-T
This acquisition reflects a shift from personalized, lab-based therapies to scalable in-body treatments that could redefine CAR-T delivery.

Spencer Knight/LinkedIn
Sascha-Oliver Bucher – Biotech Investor, Forty51 Ventures
Forty51 Ventures is proud to announce portfolio company #8: FERROSA Therapeutics.
FERROSA is building a first-in-class bispecific antibody program targeting Anemia of Inflammation — a major unmet need across chronic kidney disease, autoimmune disorders, and oncology.
We are especially excited to have Martin Stern onboard as co-founding CEO, alongside an outstanding group of advisors including Beatriz Goyenechea, Stefano Rivella, and Beatrice Goilav plus everyone behind the scenes who turns ambitious science into successful biotech companies.
At Forty51, we build biotechs around proven biology, clear unmet medical need, and the slightly dangerous idea that maybe the old way of doing things is not good enough.
FERROSA checked all three boxes.
Dessi McEntee – Owner and Head of Toxicology, Toxistrategy LLC
Your nonclinical package for an oncology program looks nothing like a package for a CNS drug.
Same U.S. Food and Drug Administration. Same IND process. Completely different expectations.
Most early-stage biotech teams miss this. They build their nonclinical program to a generic standard—28-day studies, 10x safety margin, full genotox battery—without asking whether the indication actually changes what the FDA expects.
It does. Significantly.
The graphic breaks down the full comparison across five criteria. Save it.
Building a nonclinical program and not sure which standard applies to your indication? Let’s talk.
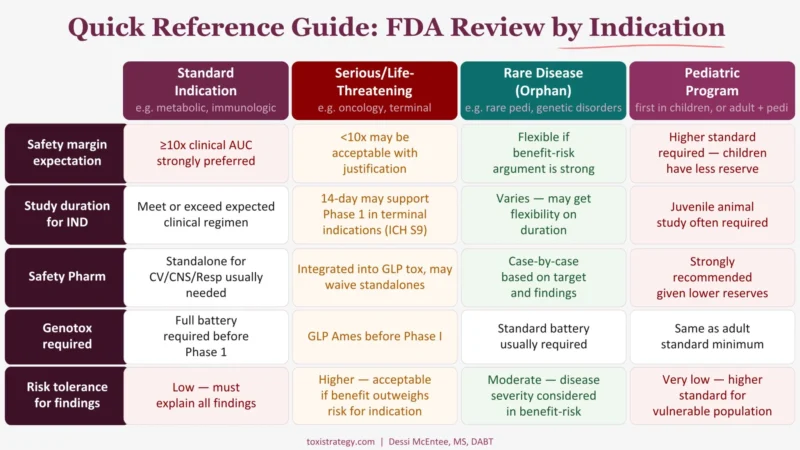
Dessi McEntee/LinkedIn
Deborah Glasser – Head of Specialty Care North America, U.S. Country Lead
If you have ever needed medicine, biotechnology is what makes it possible.
More than 100 million Americans live with a condition that can be treated, however millions more are still waiting for the next breakthrough. This progress depends not only on great science, but on smart policies that help ensure innovation can reach patients.
Biotechnology Innovation Organization (BIO) recently launched the Fight of Our Lives campaign, a national effort to highlight the people behind biotechnology, from patients and families still waiting for answers to the scientists and teams working to deliver them.
In both my work at Sanofi and on the Executive Committee of the Board of BIO, I’m reminded every day that innovation doesn’t happen in a vacuum. Efforts like #FightofOurLives help to elevate biotech’s impact on matters that affect us all; public health, the economy and global scientific progress.
Maria Fardis – Biotech CEO
It has been a great pleasure to have been chair of the Board at Obsidian and as the Company is considering a new path toward becoming a public company. Congratulations to the Company for the outstanding work bringing life-saving therapies to patients. We are grateful to the incredible investors supporting the drug development through the years and in the future.
Jack Shuang Hou – Scientific Director, Jtests
Labcorp Activates Nationwide CDx for KEYTRUDA — Turning Biomarker Access into Immediate Oncology Revenue Flow
A classic Dx–Rx flywheel: Labcorp operationalizes companion diagnostics at scale right after therapy approval.
- CDx as the gatekeeper of immunotherapy access
- Nationwide rollout of PD-L1 IHC 22C3 pharmDx
- Only FDA-approved CDx to identify eligibility for KEYTRUDA in platinum-resistant ovarian cancer
- Targets patients with PD-L1 CPS ≥1
Translation:
No test = no drug access → CDx is revenue infrastructure
- Speed-to-market = real advantage
- Labcorp participated in early validation with Agilent Technologies
- Immediate nationwide deployment post-FDA approval
Strategic insight:
In precision oncology, the winner is not just the best assay — it is the one that is clinically ready on Day 1 of drug approval
- Unlocking a high-value segment
- ~80% ovarian cancer recurrence rate
- Platinum resistance = limited options and poor prognosis
- KEYTRUDA regimen shows PFS and OS benefit (KEYNOTE-B96)
Result:
CDx becomes the front door to an expanded immunotherapy marketTakeaway
Diagnostics drive therapy adoption:
- Pharma creates demand
- Diagnostics identify patients
- Lab networks scale access
Labcorp is controlling the entry point to therapy utilization
Bottom line
Labcorp is reinforcing its role as a national precision oncology access layer, where companion diagnostics directly translate into therapy uptake, clinical decisions, and revenue flow for partners like Merck & Co..

Jack Shuang Hou/LinkedIn
Lindsey Summers – Associate Director, Talent at Day One Biopharmaceuticals
I attended a small, in-person session at Innosphere’s Innovation After Hours featuring Array BioPharma, focused on how one company’s success helped shape Boulder’s life sciences ecosystem. Founders and early leaders shared firsthand insights.
One theme dominated: elite human capital.
- Deep specialists who knew exactly where they added value
- Leaders who stayed close to the science, in the data and at the bench
- Radical transparency enabling faster decisions
- A scrappy, focused culture grounded in scientific credibility
Array operated as a small molecule oncology discovery engine that consistently moved programs into the clinic.
Outcomes:
- 20+ INDs
- Multiple approved drugs impacting patients
Core insight:
The right market + the right people = momentum
In biotech, you need to stay long enough for that momentum to materialize.There’s a tendency to default to major hubs like Boston or the Bay Area. That bias is lazy. This was proof that Boulder has a concentrated pocket of world-class talent and company building.
I’ve worked with leaders at Day One Biopharmaceuticals who came out of the Array ecosystem. The talent standard is obvious and persistent.
Key point:
Access to elite human capital is not location-dependent. It’s network-dependent. Proximity to the right builders and problems matters more than geography.Great companies don’t just build drugs.
They build concentrated talent networks that compound over time.That effect is visible locally in companies like Edgewise Therapeutics, OnKure Therapeutics, Ambrosia Biosciences, and BoCo Bio.
Fahaam Shaikh – Product Development & Global Marketing, Market Access & Strategy Lewens Labs Pvt Ltd
Big Pharma just made a $7 Billion bet on the future of cancer treatment.
Eli Lilly and Company has agreed to acquire Kelonia Therapeutics a biotech pioneering in vivo CAR-T cell therapy in a deal worth up to $7 billion.
What makes this remarkable?
Kelonia’s technology reprograms T-cells inside the body :no cell extraction, no lab modification, no reinfusion.
Just one IV. One shot at fighting cancer. From within.This isn’t just an acquisition it’s a signal.
The next era of oncology isn’t about treating cancer. It’s about teaching the body to fight it.The pharma world is evolving fast. Are you watching?

Fahaam Shaikh/LinkedIn
Artyom Smirnov – Co-Founder, Symptóme
Servier has officially acquired Day One Biopharmaceuticals, completing its tender offer at $21.50 per share
This move is more than just another deal, it’s a strategic push deeper into rare cancers and paediatric oncology.
What this means:
Strengthens Servier’s leadership in low-grade glioma
Expands its oncology footprint in the U.S. market 🇺🇸
Adds both a commercial asset + pipeline upsideKey assets joining Servier:
OJEMDA™ (tovorafenib) – FDA-approved for paediatric low-grade glioma
Emi-Le (emiltatug ledadotin) – next-gen ADC
DAY301 – targeted therapy for rare cancersPaediatric brain tumours remain an area of high unmet need, and this acquisition signals continued investment in niche, high-impact indications.
From a talent and strategy perspective, expect:
Increased demand for oncology clinical development & medical affairs talent
Continued focus on targeted therapies & ADC platforms
Integration challenges (and opportunities) across US–EU operations

Artyom Smirnov/LinkedIn
From the founding of the first biotech company fifty years ago to a single IV infusion designed to reprogram T-cells inside the body, this week traced the full arc of what the industry has dared to imagine, and is now beginning to deliver.
Read more biotech insights on OncoDaily Biotech.
Written by: Semiramida Nina Markosyan, OncoDaily Canada


