This week in oncology biotech, dealmaking dominated the headlines and the numbers were hard to ignore. Merck, Eli Lilly, and Biogen collectively committed over $12 billion across three acquisitions, while AI-biology partnerships reached new scale with Insilico Medicine’s $2.75B Lilly deal and Anthropic’s $400M acquisition of Coefficient Bio. Alongside the M&A surge, Kite Pharma secured full FDA approval for its CAR-T therapy in mantle cell lymphoma, Revolution Medicines opened enrollment in a Phase 3 pancreatic cancer trial, and Blackstone closed the largest private life sciences fund ever raised at $6.3 billion. Below are 10 featured posts from a week that signaled where capital and conviction are converging in the sector.
Wyatt McDonnell – Co-Founder and CEO, Infinimmune
In reposting Infinimmune original announcement, Wyatt McDonnell wrote:
Infinimmune is just 44 months old. Today we announced an $838MM discovery partnership with Merck, one of the world’s best drug developers. This deal is the product of human biology and hard work, and proof of what a small and relentless team can accomplish. We started Infinimmune on the premise that the human immune system is the best source of therapeutic antibodies. Thank you to our partners at Merck, our team, our investors, and the human donors whose immune systems make all of this possible—and whose biology has so much more to teach us.
In the original company post, Infinimmune wrote:
We’re excited to announce a collaboration with Merck (MSD outside the U.S. and Canada) to apply Infinimmune’s human-first antibody discovery platform to identify and develop antibody candidates.
Under the agreement, Infinimmune will deploy its proprietary Anthrobody® platform and GLIMPSE™ antibody language model to discover and optimize therapeutic antibodies directly from human immune repertoires.
The platform enables large-scale screening of millions of single memory B cells across hundreds of targets, identifying naturally paired antibodies and rapidly advancing candidates with strong affinity, high specificity, and favorable drug-like properties.
Merck will lead development and commercialization, with the deal including upfront and milestone payments totaling up to ~$838M.
A clear signal toward the future of antibody discovery: human-first, data-driven, and AI-enabled.
Read our release.
Jianye Zhang – Business Development Center Representative, Jiangxi Zvast Biotechology Co., Ltd
Blackstone just closed its record-breaking $6.3 billion BXLS VI fund, the largest private life sciences fund ever raised—an almost 40% expansion from its predecessor!
This oversubscribed fund reaffirms Blackstone’s standing as a trusted capital provider in the life sciences space, building on the BXLS platform launched in 2018 that now manages $15 billion in assets (as of Q4 2025). What’s more impressive? BXLS boasts an 86% Phase 3 clinical franchise success rate, far outpacing the industry average, and has partnered to deliver 34 regulatory approvals for innovative drugs and devices—including blockbuster therapies like LEQVIO®, AMVUTTRA® and IMBRUVICA®, plus game-changing medical devices such as MiniMed Flex™.
Over the past 12 months alone, BXLS has committed nearly $2 billion in new investments, striking impactful deals with industry leaders: from a R&D funding agreement with Merck on Sacituzumab Tirumotecan, to a $2 billion strategic financing partnership with Alnylam accelerating RNAi therapies, and a $400 million growth capital deal with Teva for TL1A antibody duvakitug, to name a few. It also facilitated Novartis’ potential $3.1 billion acquisition of Anthos Therapeutics, showcasing its ability to source, finance and actively manage large-scale product investments.
Nicholas Galakatos, Global Head of Blackstone Life Sciences, noted that this milestone is a testament to investor confidence in BXLS’ strategy and the platform’s enduring partnership with global pioneers—all to bring life-changing medicines and technologies to patients worldwide.
As the life sciences industry continues to push the boundaries of innovation, Blackstone’s massive new fund is set to fuel the next wave of breakthroughs in drug development and medical technology. Excited to see what transformative advancements this capital will unlock!

Jianye Zhang/LinkedIn
Kite Pharma
The U.S. FDA has granted traditional, full approval to our CAR T-cell therapy for adults with relapsed or refractory mantle cell lymphoma. We’re committed to advancing innovative treatments for patients with a variety of blood cancers. Read more.
Revolution Medicines
We’re pleased to share that we have begun treating patients in RASolute 303, our global Phase 3 trial evaluating our investigational therapy as first line treatment for patients with metastatic pancreatic cancer.
RASolute 303 is testing two approaches: monotherapy and combination therapy with standard of care chemotherapy.
For patients with metastatic PDAC, a disease where approximately 90% of cases are RAS-driven and nearly 80% are diagnosed at advanced stages, new treatment options are urgently needed.

Revolution Medicines/LinkedIn
Thomas Triomphe – Executive Vice President Vaccines, Sanofi
We recently launched a documentary series, called “Making it Real,” which takes an in-depth look at manufacturing sites around Sanofi.
Episode 1 takes you inside Modulus, our cutting-edge facility in Neuville, France. This site is the future of vaccine manufacturing. If a new health crisis emerges, or if people need a different vaccine urgently, we can pivot production platforms in days, not months. This agility can be a lifeline for people counting on our products.
By transforming how we produce, we’re building something that can work for everyone: patients get faster access to the vaccines and medicines they need, healthcare systems gain flexibility and resilience, and the entire pharmaceutical industry benefits from a new, more flexible manufacturing model.
Learn more about Modulus, directly from our team on site.
Jonah Probell – Founder of Lexi Ventures
Wow. Anthropic just paid $400 million for Coefficient Bio, a biotech AI startup with fewer than 10 employees, operating in stealth for only 8 months.
The company, co-founded by Samuel Stanton and Nathan Frey and based in New York City, uses AI to draft drug R&D plans, manage clinical regulatory strategy, identify new drug candidates, and model biomolecules.
The deal was all-stock. Dimension, the VC firm that backed them, reportedly generated a 38,513% paper IRR.
Eric Kauderer-Abrams, Anthropic’s head of Healthcare Life Sciences, threw down the gauntlet. “We want a meaningful percentage of all life science work in the world to run on Claude, in the same way that happens today with coding.”
For seed and pre-seed investors this validates AI-native computational biology as a category. The acquisition price for a sub-10-person, pre-revenue, 8-month-old company reflects how scarce this talent is and how strategically valuable the intersection of foundation models and biology has become.

Jonah Probell/LinkedIn
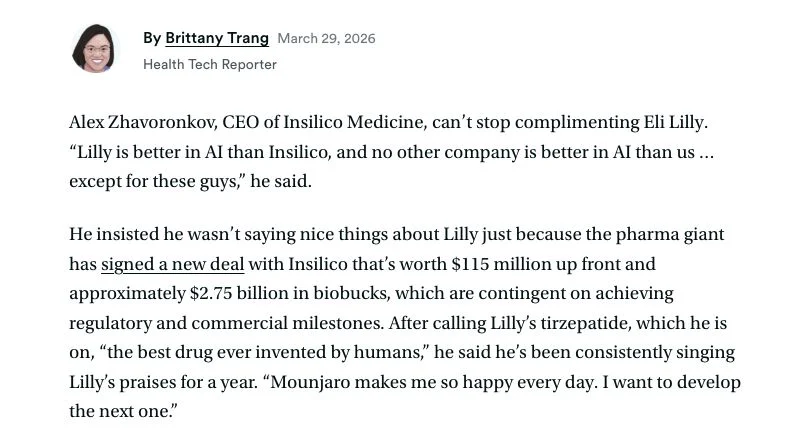
Alex Zhavoronkov – Founder, CEO and CBO at Insilico Medicine
I am pleased to announce a significant deal of up to $2.75B with $115M upfront with Eli Lilly, the maker of several drugs that I rely on as a patient. This deal is crucial for the company as it showcases our ability to design novel molecules with unique properties in a highly competitive market. Since I assumed the role of CBO, we have dramatically increased our deal flow, and this year we anticipate more than doubling our revenue while significantly increasing the number of PCCs delivered and deals made.
Looking ahead, business development and advancements in engineering foundation models for science will remain my top priorities for 2026. I am also excited about the progress of our MMAI Gym for science. Stay tuned for more updates.
I want to extend my gratitude to our team and the team at Lilly for their efforts in making this happen. We look forward to collaborating in the one team mode through the Lilly Gateway Labs.
Alex Zhavoronkov/LinkedIn
Alex Zhavoronkov/LinkedIn
Andaleeb Afaz – Head of Regulatory Affairs, Renata PLC
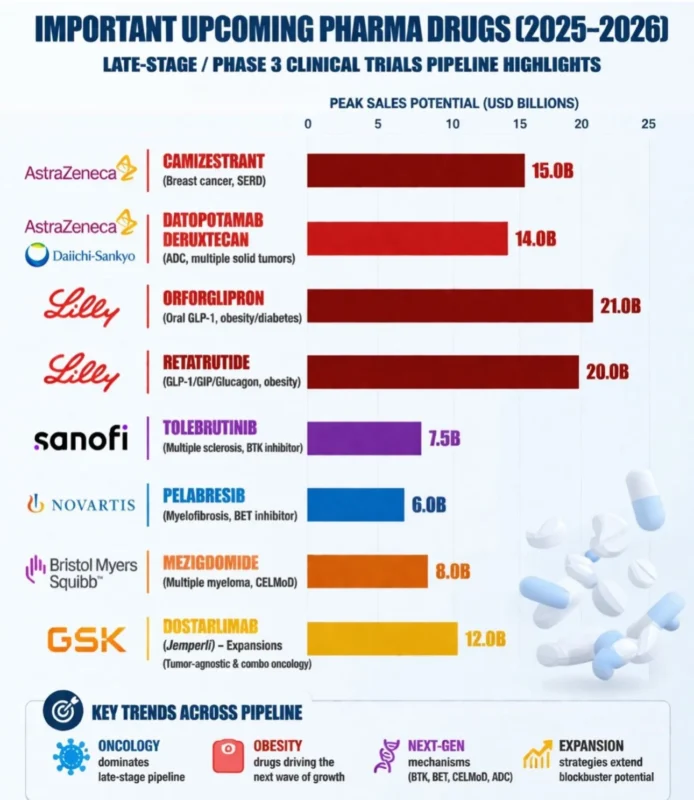
The Next Wave of Pharma Innovation is Coming (2026-27)
Here’s a snapshot of the most exciting late-stage / Phase 3 assets shaping the future of healthcare
Blockbuster pipeline highlights:
– Camizestrant (AstraZeneca) – next-gen SERD in breast cancer
– Datopotamab deruxtecan (AZ / Daiichi Sankyo) – ADC momentum continues
– Orforglipron (Lilly) – oral GLP-1 breakthrough
– Retatrutide (Lilly) – triple agonist redefining obesity
– Tolebrutinib (Sanofi) – high-stakes MS innovation
– Pelabresib (Novartis) – epigenetic targeting
– Mezigdomide (BMS) – next-gen CELMoD strategy
– Dostarlimab expansions (GSK) – immunotherapy growthWhat stands out:
– Oncology still dominates late-stage pipelines
– Obesity & metabolic diseases are becoming massive opportunities
– Novel mechanisms (BTK, BET, ADCs, CELMoDs) are transforming care
– Lifecycle expansion is just as powerful as new launchesThe next 2–3 years could define the next generation of blockbuster drugs—and reshape competitive dynamics across big pharma.
Which of these assets do you think will have the biggest impact?
Andaleeb Afaz/LinkedIn
Isabella Lockhart – Business Consultant – Biotechnology, Hartmann Young
Oncology Update!
Merck has agreed to acquire Terns Pharmaceuticals in a $5.7bn enterprise value deal, adding an early‑stage chronic myeloid leukaemia (CML) asset to its oncology pipeline as it prepares for Keytruda’s patent expiry from 2028.
Why this matters:
– Pipeline resilience: With ~$30bn of annual Keytruda revenue at risk, Merck is moving early to reshape its growth engine.
– Targeted innovation: Terns’ lead asset, TERN‑701, has shown promising clinical data in a rare blood and bone cancer and could challenge Novartis’ Scemblix.
– M&A momentum: This follows Merck’s $10bn Verona Pharma and $9.2bn Cidara deals, reinforcing its position as the most aggressive acquirer in biotech.
– Investor confidence: Merck’s shares are up ~38% since last summer, amid an industry facing an estimated $320bn patent cliff by 2030.Late‑stage trials for TERN‑701 are expected to begin later this year or early 2027, with analysts flagging potential multibillion‑dollar peak sales – neatly aligning with Keytruda’s loss of exclusivity.
A clear signal that scale + science + timing are now inseparable in oncology strategy.
Does Merck’s dealmaking signal a new valuation floor for high‑quality oncology biotech?

Isabella Lockhart/LinkedIn
Spencer Knight – Business Development Director – Biotechnology, Hartmann Young
BREAKING NEWS:
Big Pharma Drops $12B+ on Two BiotechsTwo major deals shook the biotech world today as Eli Lilly and Biogen announced multibillion-dollar acquisitions to expand into neuroscience, sleep medicine, immunology, and rare diseases.
Key Highlights:
– Eli Lilly acquires Centessa Pharmaceuticals for $6.3B upfront– Lead program: cleminorexton, an orexin receptor agonist targeting narcolepsy & idiopathic hypersomnia
– Expands Lilly’s neuroscience and sleep medicine portfolio
– Strategic move into disorders with high unmet need for wakefulness– Biogen acquires Apellis Pharmaceuticals for $5.6B upfront
– Gains two approved drugs: Empaveli® (rare kidney & blood diseases) and Syfovre® (advanced macular degeneration)
– Immediate revenue-generating assets: ~$689M combined 2025 net sales
– Diversifies Biogen beyond neurology into immunology and rare disease
– Both deals reflect a trend of premium M&A in biotech, with companies paying significant upfront cash and milestone-based contingent payments.Congrats to the teams at Eli Lilly, Centessa Pharmaceuticals, Biogen, and Apellis Pharmaceuticals for advancing patient care and driving innovation.

Spencer Knight/LinkedIn
This week made one thing clear: the pace of capital deployment in oncology and life sciences is accelerating, not plateauing. M&A is being used as a precision instrument, whether to shore up pipelines ahead of patent cliffs, acquire AI-native biology talent, or bring approved revenue-generating assets under larger roofs. At the same time, regulatory wins and Phase 3 initiations remind us that behind every deal, clinical execution remains the ultimate measure. From a $838M antibody discovery platform to a record-breaking life sciences fund, this week reflected a sector that is both well-capitalized and increasingly strategic about where it deploys that capital.
Read more biotech insights on OncoDaily Biotech.


