Advanced upper tract urothelial carcinoma (UTUC) remains a challenging disease with limited effective treatment options, particularly in metastatic settings. In this retrospective study, patients with dMMR/MSI-H UTUC—a biologically distinct subgroup enriched for sensitivity to immunotherapy—showed remarkable benefit from single-agent immune checkpoint inhibitors (ICIs), achieving an objective response rate of 83%, including 67% complete responses.
With a median progression-free survival of 65.9 months and overall survival not yet reached after long-term follow-up, the findings suggest that biomarker-selected patients with dMMR/MSI-H UTUC may experience deep and durable remissions with immunotherapy alone, supporting a more personalized treatment approach in this rare but aggressive cancer subtype
Authors: Mohammad Jad Moussa, MD, MS, Alexander Y. Andreev-Drakhlin, MD, Aradhana M. Venkatesan, MD, Surena F. Matin, MD, Lianchun Xiao, MSc, Rebecca S.S. Tidwell, MS, Amishi Y. Shah, MD, Ana C. Adriazola, NP, Leah Shaw, MSN, NP, Jianjun Gao, MD, PhD, John K. Lin, MD, MSHP, Sangeeta Goswami, MD, PhD, Pavlos Msaouel, MD, PhD, Charles C. Guo, MD, Nizar M. Tannir, MD, Arlene O. Siefker-Radtke, MD, Omar Alhalabi, MD, and Matthew T. Campbell, MD, MS
Background
Upper tract urothelial carcinoma (UTUC) represents a relatively rare but clinically aggressive subset of urothelial cancers, often associated with poorer outcomes compared with bladder cancer. A distinct biological feature of UTUC is the higher prevalence of deficient DNA mismatch repair (dMMR) and microsatellite instability-high (MSI-H) status, occurring approximately three times more frequently than in bladder tumors.
These molecular alterations lead to increased tumor mutational burden and enhanced neoantigen formation, rendering tumors more immunogenic and potentially more responsive to immune checkpoint inhibitors (ICIs). While ICIs such as pembrolizumab and nivolumab have transformed the treatment landscape across multiple malignancies, evidence supporting their role specifically in dMMR/MSI-H advanced UTUC remains limited. This study aimed to evaluate the real-world efficacy and durability of single-agent ICI therapy in this biologically enriched patient population.
Methods
This retrospective analysis reviewed institutional data from patients diagnosed with advanced UTUC between 2015 and 2024. Inclusion criteria required confirmed dMMR or MSI-H status, established through immunohistochemistry (IHC), polymerase chain reaction (PCR), or germline testing. Clinical data, molecular characteristics, treatment exposure, and survival outcomes were systematically collected. Time-to-event outcomes, including progression-free survival (PFS) and overall survival (OS), were analyzed using the Kaplan–Meier method. Objective response rates (ORR) were assessed according to RECIST v1.1 criteria.
Study Design
This was a single-center, retrospective cohort study involving 24 patients with dMMR/MSI-H advanced UTUC. Among them, 38% had locally advanced or unresectable disease, while 62% presented with metastatic disease at treatment initiation. The majority of patients demonstrated favorable performance status, with 83% having ECOG 0–1.
Patients received single-agent immune checkpoint inhibitors, predominantly pembrolizumab (71%), followed by nivolumab (17%) and atezolizumab (13%). Approximately 54% of patients had prior exposure to systemic chemotherapy, while nearly half were treated with ICIs in a chemotherapy-naïve or delayed setting. Molecular profiling revealed a strong association with hereditary cancer predisposition, with Lynch syndrome confirmed in 67% of cases, primarily driven by mutations in MSH2 and MSH6 genes.
Results
The study demonstrated remarkably high efficacy of single-agent ICI therapy in this molecular subgroup. The objective response rate reached 83%, including 67% complete responses (CRs) and 17% partial responses (PRs). Importantly, no patients experienced primary progressive disease as their best response.
Survival outcomes were notably favorable. The median progression-free survival (PFS) was 65.9 months, with 12-month and 24-month PFS rates of 95.2% and 78.8%, respectively. At a median follow-up of approximately 56.9 months, the median overall survival (OS) was not reached, indicating sustained long-term benefit. Furthermore, 42% of patients remained progression-free at 4 years, highlighting the durability of response.
The median time to best response was 3.9 months, suggesting relatively rapid tumor regression following ICI initiation. The median duration of response reached 63.1 months, reinforcing the long-lasting nature of treatment benefit.
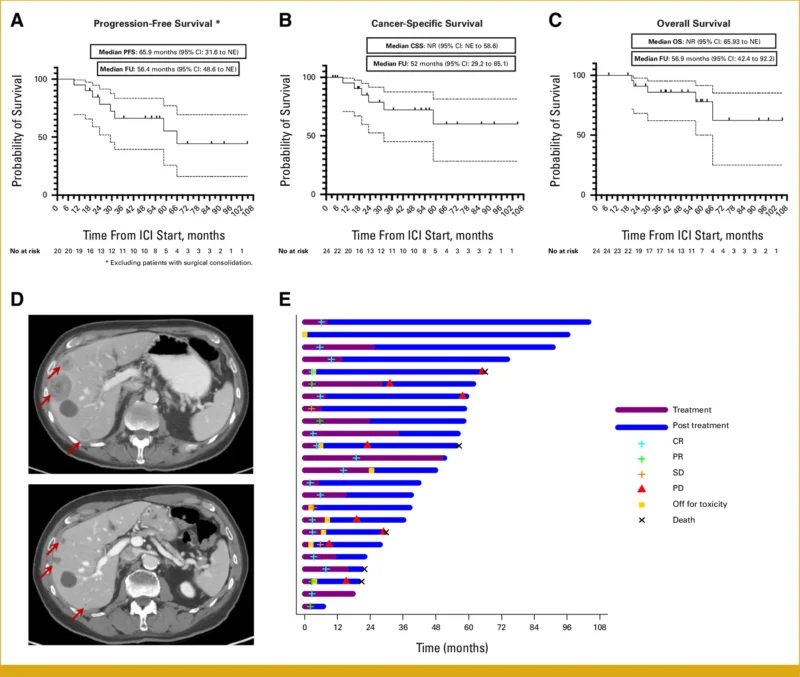
A subset of patients (17%) underwent surgical consolidation following immunotherapy, achieving favorable pathological outcomes, including complete eradication of disease (ypT0N0) in some cases.
In terms of safety, 33% of patients experienced grade ≥3 immune-related adverse events (irAEs). These included bullous pemphigoid, hepatitis, pancytopenia, colitis, and endocrine or rheumatologic toxicities. Despite these events, most toxicities were manageable, and treatment discontinuation occurred in a minority of cases due to adverse effects.
Key Findings
Single-agent immune checkpoint inhibition demonstrated exceptional clinical activity in patients with dMMR/MSI-H advanced UTUC. The observed 83% response rate and 67% complete response rate substantially exceed historical outcomes seen with platinum-based chemotherapy, where response rates are typically below 40%.
The durability of benefit represents a defining feature of this study. With a median PFS exceeding 5 years and OS not reached, outcomes appear significantly improved compared with conventional treatment paradigms, where most patients experience disease progression within 1–2 years.
The high prevalence of Lynch syndrome (67%) underscores the importance of germline and tumor-based molecular testing, not only for treatment selection but also for identifying hereditary cancer syndromes.
Additionally, the study suggests a potential role for ICIs in organ-preserving strategies, as durable responses may allow avoidance or delay of radical surgery in selected patients.
Conclusion
In this retrospective cohort, single-agent immune checkpoint inhibitors demonstrated unprecedented efficacy and durability in patients with dMMR/MSI-H advanced upper tract urothelial carcinoma. The high response rates, prolonged survival outcomes, and potential for treatment-free intervals support the integration of biomarker-driven immunotherapy into standard clinical practice for this subgroup.
However, given the study’s limitations—including small sample size, single-institution design, and lack of a comparator arm—these findings should be interpreted as hypothesis-generating. Prospective, multi-institutional trials are needed to validate these results, optimize treatment duration, and define the role of ICIs in earlier-stage disease.
Ultimately, this study emphasizes a paradigm shift toward precision oncology in UTUC, where molecular profiling directly informs therapeutic decision-making and enables more effective, individualized treatment strategies


