Pancreatic Cancer remains one of the most difficult malignancies to treat, especially in the metastatic setting where therapeutic options after progression on standard chemotherapy are limited and outcomes remain poor. Because more than 90% of PDAC tumors harbor oncogenic KRAS alterations, targeting downstream KRAS signaling has long been an appealing strategy.
Yet single-pathway inhibition has not delivered meaningful clinical benefit, in part because tumor cells can activate adaptive resistance mechanisms such as autophagy. Against this backdrop, investigators explored whether combining MEK inhibition with autophagy blockade could improve outcomes in previously treated KRAS-mutant metastatic PDAC.
This study was published in The Oncologist (Volume 31, Issue 4, April 2026) on February 27, 2026.
Title: Phase I Trial of Binimetinib Plus Hydroxychloroquine in Patients with Previously Treated Metastatic Pancreatic Cancer
Authors: S. Daniel Haldar, Fen Saj, So Jung Hong, Rishi Surana, Lianchun Xiao, J. Jack Lee, Brandon Smaglo, Dan Zhao, Huili Zhu, Ryan Huey, Jason Willis, M. Pia Morelli, Michael Overman, Florencia McAllister, Robert Wolff, Anirban Maitra, David Fogelman, Channing Der, and Shubham Pant.
Methods
This was an investigator-led, single-arm, open-label phase I dose escalation and expansion trial in patients with previously treated metastatic PDAC with a documented KRAS mutation. Key eligibility criteria included ECOG performance status 0–1, adequate organ and marrow function, and at least one prior line of systemic therapy for metastatic disease. Patients with prior MEK inhibitor or autophagy inhibitor exposure were excluded.

The study used a Bayesian optimal interval design to guide dose escalation, with a target dose-limiting toxicity rate of 30%. Binimetinib and hydroxychloroquine were administered orally in continuous 14-day cycles. The tested dose levels were:
- DL1: binimetinib 45 mg + hydroxychloroquine 600 mg twice daily
- DL-1: binimetinib 45 mg + hydroxychloroquine 400 mg twice daily
- DL-2: binimetinib 30 mg + hydroxychloroquine 400 mg twice daily
- DL-1.5: binimetinib 30 mg + hydroxychloroquine 600 mg twice daily
The primary endpoint was the maximum tolerated dose. Secondary endpoints included safety, objective response rate, disease control rate, progression-free survival, and overall survival. Tumor response was evaluated by RECIST v1.1, and survival was estimated by the Kaplan–Meier method.
What is Binimetinib and How Does it work?
Binimetinib is an oral targeted anticancer agent that belongs to the class of MEK inhibitors. It specifically targets MEK1 and MEK2, key components of the mitogen-activated protein kinase (MAPK/ERK) signaling pathway, which regulates essential cellular processes such as proliferation, differentiation, and survival. This pathway is frequently dysregulated in cancer, particularly in tumors harboring KRAS or BRAF mutations, resulting in persistent activation of growth signals and uncontrolled cell division.
Mechanistically, binimetinib inhibits the kinase activity of MEK1/2, thereby preventing the phosphorylation and activation of ERK, the downstream effector responsible for transmitting proliferative signals to the nucleus. By interrupting this signaling cascade, the drug disrupts a critical pathway that drives tumor growth.
Key aspects of its mechanism of action include:
- Inhibition of MEK1 and MEK2, central kinases in the MAPK pathway
- Blockade of ERK activation, preventing downstream signal transduction
- Suppression of signaling involved in cell proliferation and survival
- Activity in tumors with MAPK pathway overactivation, including KRAS- and BRAF-driven cancers
Through these mechanisms, binimetinib reduces tumor cell proliferation and may induce apoptosis, ultimately contributing to the control of tumor progression.
Read more about Binimetinib (Mektovi) on OncoDaily.
Results
Between December 2019 and August 2024, 34 patients were enrolled: 17 in dose escalation and 17 in dose expansion. The median age was 65 years (range, 45–79). Most patients had received multiple prior therapies: 27% had 1 prior line, 38% had 2, 32% had 3, and 3% had 4 or more prior lines. Liver metastases were present in 74% of patients, while peritoneal and distant lymph node metastases were each reported in 29%, and lung metastases in 24%.
Tolerability was a major issue early in development. Among the first three patients treated at the starting dose level, two dose-limiting toxicities occurred: grade 3 creatine phosphokinase elevation with renal impairment attributed to binimetinib, and grade 3 QT prolongation attributed to hydroxychloroquine. After dose de-escalation, the maximum tolerated dose was established as binimetinib 30 mg plus hydroxychloroquine 600 mg twice daily, and this regimen was used for the expansion cohort.
Among 31 patients evaluable for efficacy, 2 patients achieved a partial response and 9 had stable disease, while 20 patients (64.5%) had progressive disease as best response. There were no complete responses. This translated into:
- ORR: 6.5% (95% CI, 0.8%–21.4%)
- DCR: 35.5% (95% CI, 19.2%–54.6%)
- Median PFS: 1.9 months (95% CI, 1.8–2.8)
- Median OS: 5.3 months (95% CI, 3.1–9.0)
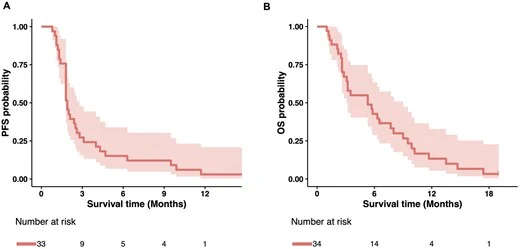
The authors also noted that the two patients with RECIST-defined partial responses had progression-free survival durations of 6.9 months and 4.7 months, respectively. No clear association between KRAS mutation subtype and response was identified in this small cohort.
Adverse Events
The combination of binimetinib and hydroxychloroquine was associated with a challenging safety profile, with treatment-related adverse events observed in a substantial proportion of patients. Toxicities were consistent with known effects of MEK inhibition, but the combination contributed to an increased toxicity burden, leading to dose modifications and early dose de-escalation.
The most common treatment-related adverse events (all grades) included:
- Rash (76%)
- Diarrhea (47%)
- Aspartate aminotransferase (AST) elevation (38%)
- Creatine phosphokinase (CPK) elevation (35%)
- Fatigue (35%)
- Anorexia (32%)
- Nausea (32%)
Other notable toxicities included cardiac-related events, such as cardiac troponin T increase (26%), QT interval prolongation (21%), sinus bradycardia (12%), and decreased ejection fraction (9%), highlighting the need for careful cardiac monitoring during treatment.
Dose-limiting toxicities were observed at the initial dose level and included grade 3 QT prolongation attributed to hydroxychloroquine and grade 3 CPK elevation with renal impairment attributed to binimetinib, which necessitated dose de-escalation and ultimately led to the identification of the maximum tolerated dose.
Serious adverse events were reported during the study, although most were considered unrelated to treatment. One grade 3 pulmonary embolism was classified as possibly related to therapy.
Overall, the safety profile of this combination was characterized by limited tolerability, which significantly impacted dose optimization and clinical feasibility in this heavily pretreated population.
Conclusion
This phase I study showed that binimetinib plus hydroxychloroquine had a challenging toxicity profile and limited clinical activity in patients with previously treated, KRAS-mutant metastatic PDAC. Although the trial successfully identified an MTD, the regimen produced a low response rate, modest disease control, short median progression-free survival, and limited overall survival in a heavily pretreated population. The findings do not support continued development of this combination in its current form for chemorefractory metastatic PDAC.
The full article is available in The Oncologist.
Read about Top 10 Pancreatic Cancer Updates – January 2026 on OncoDaily.