Chemotherapy remains one of the most powerful tools in oncology. It has cured early-stage cancers, prolonged survival in advanced disease, and continues to serve as the backbone of countless combination regimens.
Yet one of the most difficult realities in cancer care is this: treatment works, and then it stops working. A tumor shrinks. Symptoms improve. Imaging confirms response. Months later, progression appears.
Understanding why chemotherapy stops working is not about drug failure. It is about cancer biology. In 2026, we recognize that chemotherapy resistance is rarely random. It is driven by evolutionary selection, molecular adaptation, microenvironmental protection, and cellular survival strategies.
Cancer does not simply endure therapy.
It adapts to it.
Cancer Is an Evolving Ecosystem
Tumors are not uniform masses of identical cells. They are genetically diverse populations composed of multiple subclones. This phenomenon, known as intratumoral heterogeneity, has been well described in genomic analyses (Greaves & Maley, 2012, Nature).
Before chemotherapy even begins, resistant cell populations may already exist at low frequency. When treatment is applied, sensitive cells die. Resistant cells survive. This process mirrors Darwinian evolution under selective pressure.
Single-cell sequencing studies demonstrate that resistant clones can pre-exist before therapy exposure (McGranahan & Swanton, 2017, Cell). Chemotherapy often reveals resistance rather than creating it.
This evolutionary selection is one of the central explanations for why chemotherapy stops working in advanced cancer.
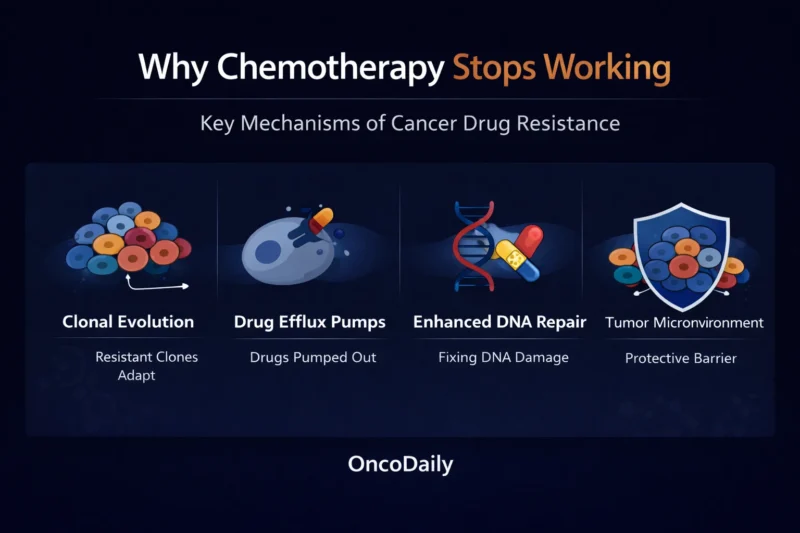
Drug Efflux: The Cellular Defense Mechanism
Some cancer cells acquire the ability to actively remove chemotherapy agents from their interior. This is mediated by ATP-binding cassette (ABC) transporters such as P-glycoprotein.
Overexpression of these transporters reduces intracellular drug concentrations and diminishes cytotoxic effect (Gottesman et al., 2002, Nature Reviews Cancer).
This mechanism has been observed across solid tumors and hematologic malignancies. Although pharmacologic inhibition of efflux pumps has been challenging clinically, their role in chemotherapy resistance remains well established.
When cancer cells pump drugs out faster than they accumulate, treatment effectiveness declines.
Enhanced DNA Repair: Fixing What Chemotherapy Breaks
Many cytotoxic agents function by damaging DNA. Platinum compounds induce crosslinks. Alkylating agents cause base modifications. Anthracyclines generate oxidative stress and DNA strand breaks.
If tumor cells enhance their DNA repair capacity, they can survive chemotherapy-induced damage.
Increased nucleotide excision repair and homologous recombination repair have been linked to platinum resistance (Lord & Ashworth, 2012, Science). Secondary BRCA mutations restoring DNA repair function have been associated with treatment resistance in BRCA-mutated cancers (Norquist et al., 2011, Journal of Clinical Oncology).
As repair pathways strengthen, chemotherapy loses lethality. This adaptive repair is a major contributor to why chemotherapy stops working over time.
The Tumor Microenvironment: Protection Beyond the Cell
Cancer exists within a complex microenvironment composed of stromal cells, fibroblasts, immune cells, extracellular matrix, and abnormal vasculature.
This environment can shield tumor cells from chemotherapy (Meads et al., 2009, Nature Reviews Cancer).
Hypoxia reduces sensitivity to cell cycle-dependent drugs. Dense stromal tissue, particularly in pancreatic cancer, impairs drug penetration. Cancer-associated fibroblasts secrete survival-promoting cytokines.
Even when chemotherapy is biologically active, inadequate drug delivery or protective signaling can blunt its effect. Sometimes chemotherapy stops working because the tumor’s surroundings prevent it from reaching effective concentrations.
Cancer Stem Cells: The Resistant Reservoir
The cancer stem cell model proposes that tumors contain a small population of stem-like cells capable of self-renewal and tumor regeneration (Batlle & Clevers, 2017, Nature Medicine).
These cells often divide slowly and exhibit high levels of drug efflux proteins and anti-apoptotic signaling. Chemotherapy primarily targets rapidly dividing cells. Stem-like cells may survive initial treatment and later repopulate the tumor.
This persistent reservoir provides another explanation for why chemotherapy stops working after initial response.
Epigenetic Plasticity and Drug-Tolerant States
Resistance does not always require permanent genetic mutations.
Epigenetic changes, including chromatin remodeling and histone modification, allow cancer cells to alter gene expression dynamically (Sharma et al., 2010, Cell).
Under chemotherapy stress, a subset of cells may enter a reversible drug-tolerant “persister” state. These cells survive treatment temporarily and resume proliferation once drug pressure decreases.
This cellular plasticity highlights that resistance can be transient, adaptive, and reversible in some contexts.

Activation of Alternative Survival Pathways
Cancer cells frequently activate compensatory signaling pathways in response to cytotoxic stress.
Upregulation of the PI3K/AKT/mTOR pathway, MAPK pathway activation, and increased anti-apoptotic protein expression such as BCL-2 have been implicated in resistance mechanisms (Fruman et al., 2017, Nature Reviews Drug Discovery).
When primary pathways are inhibited, alternative survival networks maintain viability. Modern treatment strategies increasingly combine chemotherapy with targeted inhibitors to prevent such escape.
Pharmacokinetic and Anatomical Barriers
Chemotherapy effectiveness depends on adequate delivery.
Solid tumors often exhibit abnormal vasculature and elevated interstitial pressure, impairing uniform drug distribution (Jain, 2014, Science). The blood–brain barrier restricts penetration into central nervous system metastases.
In these situations, chemotherapy stops working not because of molecular resistance but because therapeutic concentrations cannot be achieved consistently.
Immune Interactions and Resistance
Chemotherapy can induce immunogenic cell death, promoting antigen presentation and immune activation (Galluzzi et al., 2017, Nature Reviews Immunology).
However, tumors may counteract this effect through immune suppression, increased PD-L1 expression, or recruitment of regulatory immune cells.
This immune escape contributes to resistance and explains why chemo-immunotherapy combinations have become standard in several malignancies.
Tumor Burden and Probability of Resistance
The likelihood that resistant clones exist correlates with tumor size and cell number. In early-stage disease, chemotherapy can eradicate microscopic residual disease before resistant clones expand.
In metastatic disease, billions of tumor cells increase the statistical probability of resistance. This probabilistic model helps explain why chemotherapy stops working more frequently in advanced cancer compared with early curative settings.

Monitoring Resistance in 2026
Circulating tumor DNA (ctDNA) analysis now enables dynamic monitoring of tumor evolution (Siravegna et al., 2017, Nature Reviews Clinical Oncology).
Emerging resistance mutations can be detected prior to radiographic progression, allowing earlier therapeutic adaptation. The field is shifting from reactive to anticipatory oncology.
Conclusion: Outthinking Evolution
Why chemotherapy stops working is not due to a single mechanism.
It reflects evolutionary selection, enhanced DNA repair, drug efflux, microenvironmental shielding, stem-like cell survival, epigenetic plasticity, alternative signaling activation, and delivery limitations.
Cancer adapts. Treatment applies pressure. Evolution follows.
In 2026, oncology no longer views chemotherapy resistance as an unexpected event. It is an anticipated biological process, one that can be monitored, delayed, and strategically targeted.
The future of cancer care lies not only in stronger drugs, but in smarter sequencing, rational combinations, and precision-guided adaptation.
Chemotherapy remains powerful.
But staying ahead of resistance is now the true frontier.
You Can Watch More on OncoDaily Youtube TV
Written by Armen Gevorgyan, MD


