In non-small cell lung cancer, lymph nodes are usually discussed in one familiar context: staging. They determine prognosis, influence treatment intensity, and often define whether a patient is managed with surgery, systemic therapy, radiation, or a multimodality approach. But a new Frontiers in Immunology mini-review argues that this framing is too narrow. The real story is more biologically important. In NSCLC, tumor-draining lymph nodes (TDLNs) are not passive anatomic checkpoints. They are dynamic immune organs that help determine whether anti-tumor immunity develops, collapses into tolerance, or evolves into a microenvironment permissive for metastasis.
That shift in perspective matters. If the TDLN is actively shaping tumor biology, then it is no longer just a site to confirm nodal involvement. It becomes a clinically relevant space for biomarker discovery, treatment response prediction, and eventually even therapeutic intervention. That is the central message of this review, and it comes at a time when immunotherapy, neoadjuvant treatment, and precision oncology are all forcing the field to think beyond the primary tumor alone (Bain, A. M., 2026).
Why Tumor-Draining Lymph Nodes Deserve More Attention
The review makes a straightforward but important point: although nodal status remains one of the most important determinants of prognosis in NSCLC, the lymph node microenvironment has been far less studied than the primary tumor. The authors argue that this is a missed opportunity because the TDLN is a place where tumor antigens are first processed and presented to the immune system, and where the balance between immune activation and immune tolerance may be decided early.
Under ideal conditions, the TDLN supports anti-tumor immunity. But in NSCLC, that environment becomes progressively remodeled toward immune suppression across cytokine, cellular, genomic, transcriptomic, and metabolic domains. Those changes are not biologic background noise. According to the review, they may influence tumor growth, early metastasis, and responsiveness to treatment.
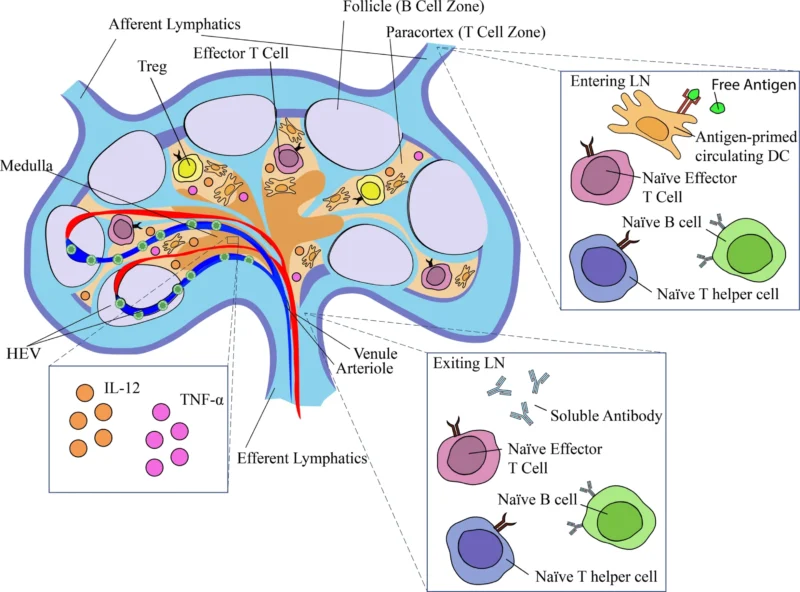
The Lymph Node Is a Functional Immune Organ, Not Just a Metastatic Waystation
The review begins by revisiting lymph node structure, because anatomy is part of the biology. The cortex contains B-cell follicles and germinal centers, while the paracortex is the main site of antigen presentation and T-cell priming. High endothelial venules selectively recruit lymphocytes into the node, and tertiary lymphoid structures in tumors and invasive margins also interact with this broader immune ecosystem. Importantly, tumor-invaded TDLNs are associated with reduced germinal center formation, decreased B-cell infiltration, and diminished memory B-cell formation in the primary tumor context, suggesting that nodal dysfunction may directly weaken anti-tumor immunity beyond the node itself.
This is one of the most useful conceptual points in the paper. The TDLN is not simply downstream from the tumor. It is part of the machinery that helps determine whether effective tumor immunity is built in the first place.
© 2026 Bain, DeMaio, Velez, Tsay and Sterman
NSCLC Progression Involves Active Remodeling of the TDLN
The review describes tumor-induced TDLN remodeling as a kind of pre-metastatic niche. Tumors shape the nodal environment through lymphatic remodeling, growth factor secretion, immunosuppressive cytokines, exosomes, changes in immune cell populations, and defects in antigen presentation. These changes can emerge before overt nodal metastasis develops, meaning the TDLN may already be biologically compromised even when malignant cells have not yet clearly replaced nodal tissue.
That has obvious implications for staging and recurrence risk. But it also has implications for treatment timing. If the TDLN is being conditioned early, then it may influence how well a patient responds to neoadjuvant immunotherapy, radiation, or surgery in ways that are not captured by conventional tumor-only biomarkers.
Early Metastasis to the TDLN Is More Than a Staging Event
The review also emphasizes that metastatic spread to the TDLN is a dynamic process. Tumor cells arriving in the node may be destroyed, pass through, or establish metastases. Once they begin colonizing the node, they usually enter through the subcapsular sinus and then progressively invade deeper structures until nodal parenchyma is replaced. The review highlights CD169-positive macrophages in the subcapsular sinuses as among the first immune cells to encounter afferent lymph, making them biologically important in host defense.
The paper also notes that nodal micrometastases in NSCLC have been associated in several studies with worse disease-free and/or overall survival, while even isolated tumor cells may hold prognostic value, although that remains more controversial. In other words, the TDLN matters long before bulky nodal replacement becomes radiographically obvious (Bain, A. M., 2026).
The Cytokine Milieu in TDLNs Is Distinctly Immunosuppressive
One of the clearer biologic themes in the review is the cytokine profile of tumor-draining lymph nodes. Studies cited by the authors found higher levels of TGF-β in TDLNs compared with non-draining nodes, alongside increases in IL-10 and decreases in inflammatory cytokines such as IL-12 and TNF-α. In one cited EBUS-TBNA study, IL-10 expression in the TDLN correlated with tumor stage, while IL-12 expression correlated inversely with survival.
This is not a subtle finding. It suggests that the nodal immune environment becomes biased toward suppression in proportion to disease burden. Other studies cited in the review also found increases in metastatic nodes of factors such as VEGF-A, angiopoietin-2, uPA, and sHER-2/neu, further reinforcing the idea that the TDLN is both immunologically and biologically active (Bain, A. M., 2026).
Cellular Composition in the TDLN Reflects Suppression, Not Activation
The review’s cellular section is particularly relevant for clinicians thinking about immunotherapy biomarkers. Compared with benign lymph nodes, TDLNs in NSCLC show decreased overall immune-cell percentages, reduced relative lymphocyte abundance, and increased monocytes. Myeloid antigen-presenting cells may be increased, but with lower CD80/CD86 expression, suggesting reduced costimulatory capacity.
Several cited studies found increased regulatory T cells, depletion of CD4-positive effector T cells, and enrichment of PD-1-positive effector and regulatory T cells within tumor-draining nodes. In one study, TDLNs from patients who progressed on immune checkpoint inhibition had higher Treg frequencies than those from responders, raising the possibility that nodal immune composition could help predict benefit from checkpoint blockade.
The review also notes possible differences by histology. In some cohorts, adenocarcinoma-associated TDLNs showed more pronounced FOXP3-positive Treg accumulation than squamous cell carcinoma, with associated reductions in natural killer cells. That is an early but intriguing suggestion that tumor subtype may shape nodal immune suppression differently.
Genomic and Transcriptomic Heterogeneity Extends Into the Node
The review makes another important argument: tumor-draining lymph nodes may help explain some of the heterogeneity that is missed when only the primary tumor is profiled. Numerous studies cited in the paper found discordance between primary tumors and nodal metastases for driver mutations and PD-L1 expression, which could directly affect treatment selection and interpretation of biomarker testing.
Transcriptomic work further deepens this. Bulk RNA sequencing and single-cell RNA sequencing studies cited in the review suggest that metastatic lymph nodes show marked immune remodeling, enrichment of myeloid populations, expansion of suppressive cell states, and altered signaling pathways that may influence both tumor behavior and immune response. More recent single-cell work in lymph node metastases from lung adenocarcinoma identified distinct malignant epithelial phenotypic clusters and developed a prognostic risk score linked to overall survival (Bain, A. M., 2026).
For thoracic oncology, this is highly relevant. If nodal tissue captures clinically meaningful biologic divergence from the primary tumor, then TDLN sampling may eventually have value beyond confirming metastatic involvement.
Metabolism in the TDLN May Become the Next Biomarker Layer
The review also highlights the metabolic environment of the TDLN as an emerging area. It discusses pathways involving tryptophan catabolism, IDO1, kynurenine, glutamine metabolism, asparagine synthetase, and arginase. These pathways are tied to Treg activation, T-cell suppression, tumor immunogenicity, and checkpoint responsiveness.
One cited human study found that while no single metabolomic feature could cleanly distinguish malignant from benign lymph nodes, metabolites such as kynurenine and oxidized glutathione differed in aspirates from patients with NSCLC. Another cited study linked high asparagine synthetase expression in lymph node metastases to improved efficacy of neoadjuvant immunotherapy. These are still early signals, but they suggest that the TDLN may eventually be profiled metabolically as part of a broader biomarker strategy.
Why the TDLN Is So Attractive Clinically: We Can Actually Access It
One of the strongest practical points in the review is that TDLNs in lung cancer are routinely accessible. Modern endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) has made minimally invasive mediastinal sampling standard in thoracic oncology. That gives lung cancer an advantage over some other solid tumors: the biologically relevant lymph node compartment is often already reachable in routine care.
The authors argue that this accessibility makes the TDLN an unusually practical site for biomarker development. Instead of relying only on the primary tumor or peripheral blood, clinicians may eventually be able to use TDLN samples to characterize cytokine patterns, immune-cell composition, gene expression, or metabolomic features that help guide therapy (Bain, A. M., 2026).
TDLNs May Also Be Direct Therapeutic Targets
The review goes a step further by arguing that tumor-draining lymph nodes are not only biomarker sources but also therapeutic targets. Prior research has shown that intact TDLNs are important for response to PD-1/PD-L1 blockade, and neoadjuvant immune checkpoint inhibition appears to induce immune remodeling within the nodal compartment. Conversely, tumor-invaded TDLNs have been associated with poorer pathologic responses and more rapid relapse after neoadjuvant immunotherapy.
The review also discusses radiotherapy. Preclinical work suggests that direct irradiation of the TDLN can attenuate anti-tumor immune activation, whereas the timing of delayed adjuvant nodal irradiation may preserve or improve combined radio-immunotherapy efficacy. That distinction is clinically important because it suggests the node is not simply collateral tissue in a radiation field; it may be part of the therapeutic immune circuit.
The authors also highlight more experimental strategies, including bronchoscopic cryoimmunotherapy, intranodal delivery of lipid nanoparticle mRNA constructs, tumor antigen-pulsed dendritic cells, and adoptive immunotherapy using lymph-node-derived cells. Several ongoing trials are already exploring TDLN-accessible therapeutic platforms, including viral immunotherapy, oncolytic viruses, and hafnium radiosensitizers.
The Bigger Message for Thoracic Oncology
What this review ultimately does is broaden the map of clinically meaningful biology in NSCLC. The field has spent years focusing on the primary tumor, circulating biomarkers, and distant metastases. The TDLN has remained somewhat underrecognized despite sitting at the intersection of staging, immune priming, early dissemination, biomarker sampling, and treatment response. The authors make a convincing case that this should change.
For OncoDaily readers, the most important idea is not that the TDLN is newly important. It has always been important. The difference is that we are now beginning to understand why, and we finally have the tools to study it properly.
The Clinical Takeaway
This 2026 mini-review argues that in NSCLC, tumor-draining lymph nodes are underused but highly relevant immune organs, progressively remodeled toward suppression across cytokine, cellular, genomic, transcriptomic, and metabolic levels. These changes may shape metastasis, influence response to treatment, and offer a realistic opportunity for biomarker development and local therapeutic innovation. Because TDLNs are routinely accessible through techniques such as EBUS-TBNA, they may become one of the most practical next frontiers in thoracic precision oncology.
Read full article here


