Perioperative immunotherapy has already changed the standard of care in resectable non-small cell lung cancer, but the next phase of progress will likely depend on whether new immune targets can further increase pathologic complete response and long-term survival. At the AACR Annual Meeting 2026, interim phase 2 data from the MATISSE trial introduced one such strategy: adding IPH5201, an anti-CD39 monoclonal antibody, to durvalumab plus platinum-based chemotherapy in patients with resectable stage II to IIIA NSCLC.
The early signal is provocative, particularly in PD-L1-positive disease, where the reported pCR rates were numerically higher than those previously reported with durvalumab plus chemotherapy alone. (Barlesi et al., 2026).
This is an early dataset, and caution is necessary. Still, MATISSE offers a biologically interesting look at how dual inhibition of CD39 and PD-L1 might push perioperative immunotherapy beyond current benchmarks.

Why CD39 07:03:37 Matters in Resectable NSCLC
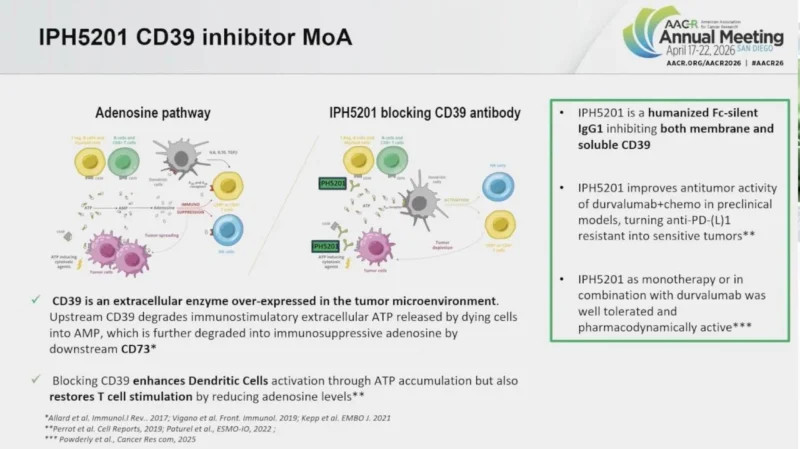
The rationale behind MATISSE is grounded in tumor immunobiology. In the tumor microenvironment, CD39 ectonucleotidase contributes to the generation of immunosuppressive adenosine, which can dampen antitumor immune activity. IPH5201 is designed to block both membrane and soluble CD39, reduce immunosuppressive adenosine signaling, and preserve immunostimulatory ATP, thereby shifting the tumor microenvironment toward a more immune-reactive state. (Barlesi et al., 2026).
That makes CD39 an appealing partner for PD-L1 blockade. In resectable NSCLC, where perioperative checkpoint inhibition already improves pathologic response and event-free survival, the question is whether an additional immune-modulating layer can drive deeper tumor eradication before surgery.
The MATISSE Trial Design
MATISSE (NCT05742607) is an open-label, single-arm phase 2 study evaluating IPH5201 in combination with neoadjuvant platinum-based chemotherapy and perioperative durvalumab in previously untreated patients with resectable stage II to IIIA NSCLC. The primary endpoint is centered on pathologic complete response, with additional biomarker work assessing soluble and membrane CD39 occupancy in blood and tumor CD39 expression (Barlesi et al., 2026).
The analysis presented at AACR 2026 was based on the planned interim look after enrollment of 40 patients. Because this is not a randomized comparison, the results should be interpreted as signal-generating rather than practice-changing. Still, in perioperative lung cancer, pCR remains one of the most watched early endpoints because of its established association with longer-term outcomes.
Safety Appears Manageable
At the interim analysis, 40 patients had been enrolled. The combination showed a favorable safety profile, with no new emerging safety signal compared with durvalumab plus chemotherapy. Treatment-related adverse events occurred in 89.5% of patients, but they were mostly grade 1 or 2. Importantly, 35 of 40 patients, or 87.5%, went on to receive the planned surgery (Barlesi et al., 2026).
That surgical completion rate matters. In perioperative studies, treatment intensification only becomes clinically meaningful if patients can still reach definitive surgery safely and on time. Based on these interim data, adding CD39 blockade does not appear to have disrupted the overall operability pathway.
The pCR Signal Is Strongest in PD-L1-Positive Disease
In the full interim cohort, the pathologic complete response rate was 27.5%, with 11 of 40 patients achieving pCR. That figure alone is respectable, but the more interesting signal emerged in PD-L1-defined subgroups.
Among patients with PD-L1 ≥1%, the pCR rate reached 35.7%. In the subgroup with PD-L1 ≥50%, the pCR rate rose to 50%. These subgroup pCR rates are clinically interesting because the investigators note that they are numerically higher than the 21.2% and 27.5% pCR rates reported with durvalumab plus chemotherapy in AEGEAN for the corresponding PD-L1 subgroups, although this remains an indirect cross-trial comparison. (Barlesi et al., 2026).
This does not prove superiority, because MATISSE is single-arm and cross-trial comparisons must always be interpreted with caution. Still, the signal is strong enough to justify continued enrollment and further evaluation, especially in PD-L1-positive patients.

Biomarker Findings Add Biological Credibility
One of the most important strengths of this interim report is that it does not rely only on clinical pCR data. The biomarker findings support pharmacodynamic activity consistent with the intended mechanism of IPH5201.
The study showed complete and sustained saturation of soluble and membrane CD39 in blood, supporting real pharmacodynamic target engagement. In addition, investigators observed a trend toward higher baseline CD39-positive cell density in tumors, as well as higher CD8-positive cell density, among patients who achieved pCR or major pathologic response (Barlesi et al., 2026).
This is especially relevant because it suggests that tumor CD39-positive cell density may represent an emerging biomarker of pathologic response. If confirmed, CD39-positive immune contexture could help identify the patients most likely to benefit from this treatment intensification strategy.
How MATISSE Fits Into the Current Perioperative Landscape
The current perioperative standard in resectable NSCLC already includes PD-(L)1 blockade plus chemotherapy, so MATISSE is entering a competitive and fast-evolving field. That means a new regimen must do more than show activity. It must demonstrate that it can meaningfully improve depth of response, remain surgically feasible, and ideally identify a subgroup where the added complexity is justified.
This is where MATISSE becomes interesting. Rather than trying to replace chemoimmunotherapy, it is attempting to layer a rational immune target on top of an established perioperative backbone. The strongest signal so far appears to be in PD-L1-positive disease, particularly PD-L1 ≥50%, where the 50% pCR rate is difficult to ignore, even in a small sample.

Important Limitations
There are several reasons to stay cautious. MATISSE is a single-arm phase 2 study with only 40 patients in this interim analysis. The pCR findings are encouraging, but they come from small subgroup numbers. There is no randomized control arm within the study itself, so interpretation depends partly on historical comparison. Long-term endpoints such as event-free survival and overall survival are not yet available, and those will ultimately determine whether the biologic promise of CD39 inhibition translates into clinically meaningful improvement.
That said, early perioperative development often begins exactly this way: with a strong biologic rationale, a manageable safety profile, and a pCR signal that looks sufficiently competitive to justify deeper study.
The Clinical Takeaway
The interim phase 2 MATISSE data suggest that adding IPH5201, a CD39-blocking antibody, to durvalumab plus platinum-based neoadjuvant chemotherapy may deepen pathologic response in resectable NSCLC, particularly in PD-L1-positive tumors. The regimen appeared feasible, showed no new emerging safety signal, and demonstrated complete and sustained saturation of soluble and membrane CD39 in blood. The biomarker findings also point to tumor CD39-positive cell density as a potentially relevant emerging marker of response (Barlesi et al., 2026).
For now, MATISSE should be viewed as an early, hypothesis-generating signal that supports continued evaluation of CD39 blockade in perioperative NSCLC. It does not change practice today. But it does suggest that the next step in perioperative NSCLC may not be a new checkpoint inhibitor alone. It may be a smarter way of reshaping the tumor microenvironment around the checkpoint strategies we already use.
You can read full abstract here


