Advanced EGFR-mutated non-small cell lung cancer has entered a more complex first-line treatment era. Osimertinib monotherapy established a durable and tolerable standard for many patients, while newer systemic intensification strategies, including osimertinib plus chemotherapy and amivantamab plus lazertinib, have improved outcomes but added toxicity, complexity, and treatment burden (Ramalingam et al., 2020; Jänne et al., 2026; Yang et al., 2025). Within this evolving landscape, local ablative therapy, or LAT, is emerging as a possible third pathway: a way to intensify disease control while maintaining the backbone of EGFR TKI monotherapy.
A new systematic review and meta-analysis by Brunetti and colleagues, accepted in the Journal of Thoracic Oncology, evaluated whether integrating LAT into first-line EGFR TKI treatment improves outcomes in advanced EGFR-mutated NSCLC. Across a heterogeneous evidence base, LAT added to EGFR TKI therapy was associated with significantly longer progression-free survival and overall survival compared with EGFR TKI alone, with an acceptable safety profile and no unexpected safety signals (Brunetti et al., 2026).
Why LAT Is Being Reconsidered In EGFR-Mutated NSCLC
Local ablative therapy is already familiar in oncogene-driven NSCLC, particularly for oligoprogressive disease, where focal treatment can allow patients to continue an effective TKI. The question addressed by this review is different: whether LAT should be integrated earlier, as part of first-line therapy, either upfront or after initial disease control with an EGFR TKI.
The rationale is clinically and biologically plausible. EGFR-mutated NSCLC often progresses at pre-existing disease sites, and local control of selected lesions may delay the emergence of widespread resistant disease. EGFR-mutant cancer cells may also have increased radiosensitivity, and EGFR signaling is involved in DNA damage repair and cell survival after radiation exposure (Das et al., 2007; Nyati et al., 2006). These concepts support the hypothesis that EGFR inhibition and radiotherapy-based LAT may work together to prolong disease control.
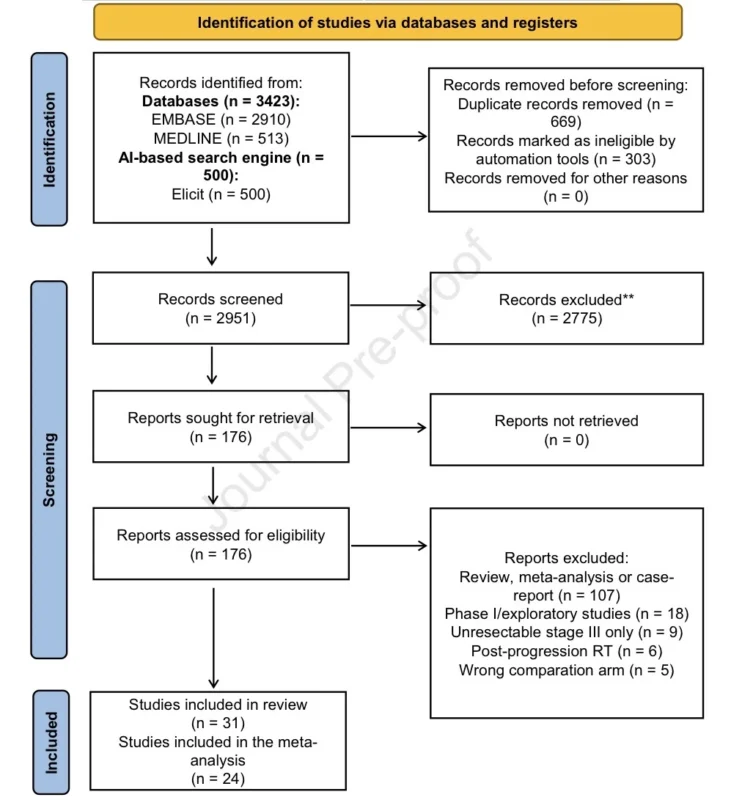
How The Meta-Analysis Was Conducted
The authors searched MEDLINE, Embase, and Elicit through December 2025. The protocol was registered in PROSPERO under CRD420251244650. Eligible studies included adults with advanced or metastatic NSCLC harboring activating EGFR mutations, including common and uncommon variants. EGFR TKIs included first-generation, second-generation, and third-generation agents, such as gefitinib, erlotinib, icotinib, afatinib, dacomitinib, osimertinib, lazertinib, aumolertinib, and furmonertinib (Brunetti et al., 2026).
LAT was primarily defined as ablative-intent radiotherapy, including stereotactic body radiotherapy, stereotactic ablative radiotherapy, stereotactic radiosurgery, and hypofractionated or conventionally fractionated radiotherapy. Surgery and interventional radiology procedures were also eligible when clearly integrated into an EGFR TKI-based strategy. Studies focused only on palliative radiotherapy or LAT for oligoprogression were excluded.
Overall, 31 studies involving 3,355 patients met inclusion criteria. Of these, 24 comparative studies involving 3,012 patients were included in quantitative meta-analysis. This included 6 randomized studies, 2 prospective non-randomized studies, and 16 retrospective observational cohorts. The remaining 7 studies were summarized descriptively (Brunetti et al., 2026).
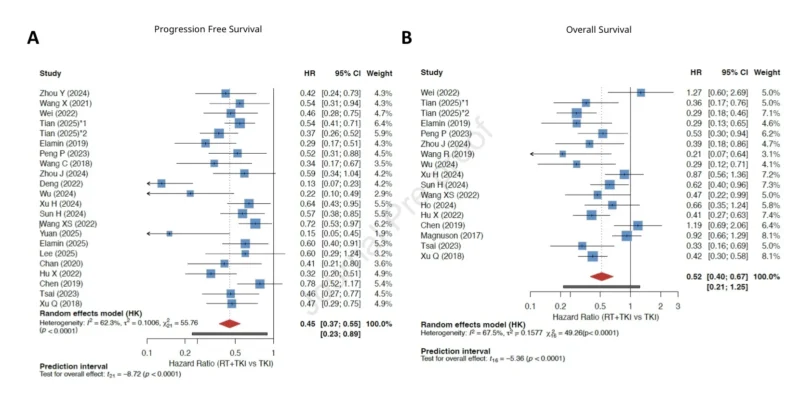
LAT Was Associated With Longer PFS And OS
The primary efficacy signal was consistent. In the pooled random-effects analysis, adding LAT to EGFR TKI monotherapy significantly reduced the risk of disease progression, with a progression-free survival hazard ratio of 0.45 and a 95% confidence interval of 0.37–0.55. Overall survival also favored LAT integration, with a hazard ratio of 0.52 and a 95% confidence interval of 0.40–0.67 (Brunetti et al., 2026).
These results suggest that LAT integration was associated with both disease-control and survival advantages. However, the authors appropriately emphasized the heterogeneity of the evidence base. Disease burden, timing of LAT, treatment site, radiation technique, TKI generation, and study design varied across studies. Statistical heterogeneity was moderate for both PFS and OS, with I² values of 62.3% and 67.5%, respectively.
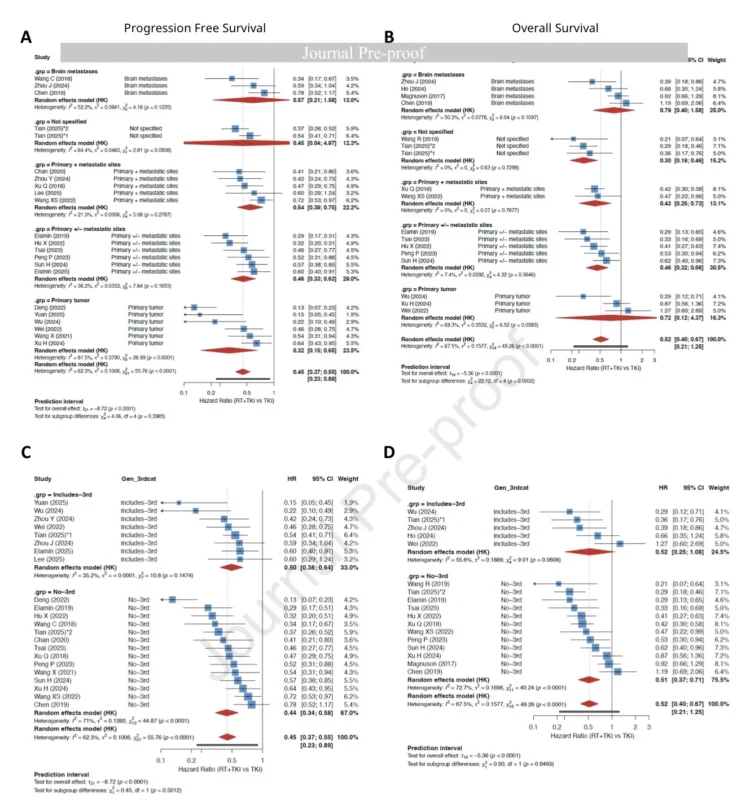
Benefit Was Seen Across Multiple Clinical Subgroups
The review found that the PFS benefit of LAT was not limited to one narrow clinical group. When stratified by baseline disease burden, LAT improved PFS in oligometastatic disease, non-oligometastatic disease, and mixed or unselected populations. The pooled PFS hazard ratio was 0.44 in oligometastatic cohorts, 0.60 in non-oligometastatic cohorts, and 0.47 in mixed or unselected cohorts (Brunetti et al., 2026).
For overall survival, the benefit appeared strongest in oligometastatic and unselected populations. OS hazard ratios were 0.42 in oligometastatic cohorts and 0.50 in unselected populations. The authors did not find a significant OS benefit in the non-oligometastatic subgroup, and they interpreted this cautiously because of limited sample size, retrospective selection, and heterogeneity.
Timing also mattered, but both tested approaches favored LAT. Upfront LAT was associated with improved PFS, with a hazard ratio of 0.47, while consolidative LAT after initial EGFR TKI response or stabilization showed a hazard ratio of 0.44. OS also favored both strategies, with hazard ratios of 0.53 for upfront LAT and 0.49 for consolidative LAT (Brunetti et al., 2026).
Third-Generation EGFR TKIs Remain An Important Evidence Gap
A key clinical question is whether LAT adds value in the modern osimertinib era. The meta-analysis found PFS benefit both in studies using third-generation EGFR TKIs and in those using first- or second-generation or mixed TKI cohorts. For PFS, the hazard ratio was 0.50 in third-generation TKI cohorts and 0.44 in non-third-generation or mixed cohorts (Brunetti et al., 2026).
For OS, the pooled hazard ratio was 0.52 in third-generation TKI cohorts, but the confidence interval crossed 1.00, while non-third-generation or mixed cohorts showed a hazard ratio of 0.51. This does not mean LAT lacks value with third-generation TKIs. Rather, it reflects that prospective randomized evidence with contemporary third-generation EGFR TKIs remains limited.
This is clinically important because current first-line EGFR-mutated NSCLC treatment is increasingly shaped by osimertinib-based or lazertinib-based strategies, including systemic combination regimens. The review supports LAT as a promising strategy, but not yet as a replacement for phase III evidence in the modern systemic treatment era.
Safety Was Manageable, But Pneumonitis Needs Attention
The addition of LAT increased treatment-related adverse events, mainly due to radiotherapy-related toxicity. Across studies, any-grade adverse events in combination arms ranged from approximately 75% to 97%, compared with 50% to 97% in EGFR TKI-only arms. Most events were grade 1–2 (Brunetti et al., 2026).
Grade 3 or higher toxicities were uncommon, ranging from 2% to 18% in LAT arms and 0% to 9% in control arms. Pulmonary toxicity was the most consistent LAT-associated safety signal. Radiation pneumonitis or pneumonia was reported more frequently in LAT arms, with any-grade incidence ranging from 6% to 90%, compared with 0% to 24% with EGFR TKI alone. Severe pneumonitis was uncommon, but the wide variability in reporting limits precise risk estimation.
The review found no unexpected safety signals overall. Still, the safety profile reinforces the need for careful patient selection, high-quality radiotherapy planning, and multidisciplinary discussion, especially when combining thoracic radiotherapy with EGFR TKIs.
What This Means For Clinical Practice
This meta-analysis does not establish LAT plus EGFR TKI monotherapy as a universal first-line standard. The authors are careful to position the findings as hypothesis-strengthening rather than practice-defining. However, the consistency of the PFS and OS signals across multiple subgroups makes LAT a serious candidate for further prospective evaluation.
The clinical appeal is clear. Systemic intensification can improve outcomes but may increase toxicity and treatment complexity. LAT offers a different form of intensification, focusing on durable control of selected disease sites while preserving the tolerability of EGFR TKI monotherapy. This may be particularly relevant for patients with oligometastatic or oligoresidual disease, patients with dominant thoracic disease, or patients who are not ideal candidates for systemic combination strategies.
At the same time, the strategy is highly context-dependent. LAT feasibility depends on disease distribution, lesion location, radiation expertise, prior treatments, organ-at-risk constraints, and patient preference. The benefit is unlikely to be uniform across all advanced EGFR-mutated NSCLC patients.
Limitations That Should Shape Interpretation
The authors identified several limitations. Definitions of oligometastatic and oligoresidual disease varied substantially across studies, and many were applied retrospectively. Several randomized studies were open-label, with investigator-assessed PFS and no blinded central radiology review. Most non-randomized studies were at serious risk of bias because of confounding, patient selection, and lack of random allocation (Brunetti et al., 2026).
Retrospective studies showed larger PFS effect sizes than prospective studies, which may reflect selection bias or immortal time bias. The evidence base was also geographically concentrated, with many studies conducted in China, which may limit generalizability to other health systems and populations.
Another important limitation is the limited representation of third-generation EGFR TKIs. Although third-generation TKI cohorts were included, only about one-third of patients in the pooled analysis received a third-generation agent. This limits confidence in extrapolating the magnitude of benefit to contemporary first-line regimens.
The economic impact of integrating LAT into first-line management was also not assessed. This matters because ablative radiotherapy requires technical resources, multidisciplinary coordination, and center-level expertise.
A Strategy Worth Testing In The Modern EGFR Era
The findings from Brunetti and colleagues support a renewed interest in first-line LAT integration for advanced EGFR-mutated NSCLC. The pooled data suggest that adding LAT to EGFR TKI monotherapy is associated with improved PFS and OS, with toxicity that appears manageable in experienced settings (Brunetti et al., 2026).
The next step is not simply to ask whether LAT works, but to define where it works best. Future trials need to clarify which patients benefit most, whether LAT should be delivered upfront or after initial TKI response, which lesions should be treated, how to integrate LAT with third-generation EGFR TKIs, and whether biomarkers such as circulating tumor DNA can guide timing and selection.
For now, LAT plus EGFR TKI monotherapy should be viewed as a biologically grounded, clinically promising, and multidisciplinary strategy. It may become especially relevant as clinicians seek to balance longer disease control with tolerability, quality of life, and sustainable treatment intensity in advanced EGFR-mutated NSCLC.
Read full article here