For years, radiotherapy to the chest in stage IV non-small cell lung cancer was largely viewed as a palliative tool, used mainly to relieve symptoms such as cough, pain, hemoptysis, or airway compromise. A new consensus statement from the International Association for the Study of Lung Cancer, published in the Journal of Thoracic Oncology, argues that this framework is no longer broad enough for the modern era. The panel reviewed the available randomized evidence on definitive radiotherapy to the primary lung tumor in metastatic NSCLC and concluded that the strongest support currently exists in EGFR-mutant oligometastatic disease, while the data in patients without actionable genomic alterations remain promising but still insufficient for firm routine recommendations.
Why the Primary Tumor Is Back in Focus
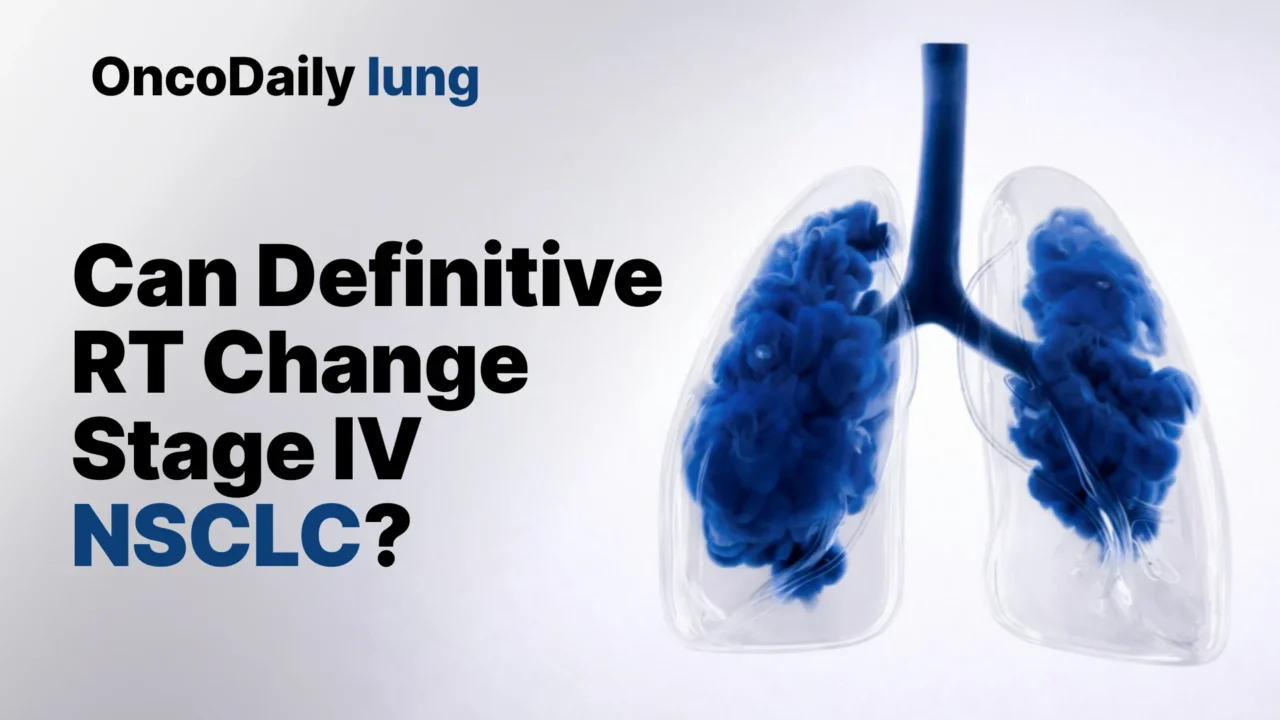
The renewed interest in aggressive local treatment is not only about better radiation technology. It is also about a changing understanding of how metastatic NSCLC behaves. The biological rationale is increasingly supported by evolutionary data suggesting that, in at least some patients, the primary lung tumor continues to act as a source of metastatic spread. In the TRACERx analysis published in Nature, investigators found that in 13 of 19 evaluable cases, dissemination appeared to arise solely from the primary tumor, reinforcing the idea that local control of the thoracic disease may have consequences beyond symptom relief alone.
That concept becomes more relevant as systemic therapy improves. Patients with metastatic NSCLC now live longer with targeted therapies and immunotherapy than they did a decade ago, which means uncontrolled thoracic disease has more time to become clinically significant. The IASLC panel frames definitive radiotherapy to the primary tumor not as a replacement for systemic therapy, but as a possible complement to it in carefully selected patients.
Where the Evidence Is Strongest: EGFR-Mutant Disease
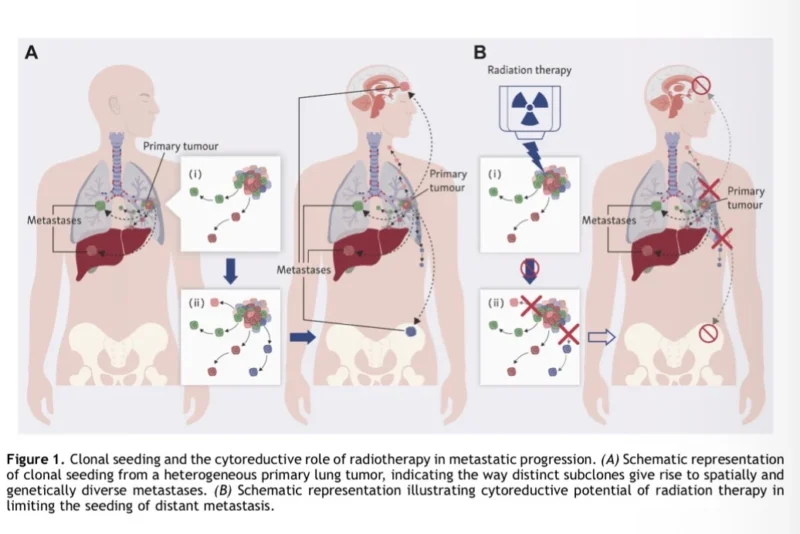
The clearest signal comes from EGFR-mutant oligometastatic NSCLC. In the phase III study by Sun and colleagues, adding thoracic radiotherapy to first-line EGFR TKI therapy improved both progression-free survival and overall survival, with median PFS increasing from 10.6 to 17.1 months and median OS from 26.2 to 34.4 months. The authors also reported improved locoregional control, with toxicity considered acceptable, although treatment-related adverse events were higher in the combined-modality arm.
The SINDAS trial pointed in the same direction. In that randomized study, upfront radiotherapy added to first-generation EGFR TKI therapy improved median PFS from 12.5 to 20.2 months and median OS from 17.6 to 25.5 months in patients with synchronous EGFR-mutated oligometastatic NSCLC. However, SINDAS treated not only the thoracic primary tumor but also other oligometastatic sites, which means it supports the broader role of local consolidative therapy more than it isolates the precise contribution of the primary lesion alone.
This is why the IASLC consensus is careful in its wording. It does not claim that every metastatic EGFR-mutant case should automatically receive definitive thoracic RT. But it does state that early RT, commonly framed as consolidation after initial systemic therapy, has become a credible life-prolonging strategy in this subgroup.
The Picture Is Less Clear in Non-Driver-Positive Disease
For patients without actionable genomic alterations, the panel is much more cautious. The consensus notes that randomized evidence specifically isolating the effect of radiotherapy to the primary lung tumor is still limited in this setting. Preliminary results are encouraging, but they do not yet justify a universal recommendation for definitive thoracic RT across all stage IV NSCLC populations.
That caution is justified. Trials such as CURB support the broader principle that ablative radiotherapy can prolong progression-free survival in oligoprogressive metastatic NSCLC, but they do not directly answer the narrower question of how much benefit comes from treating the thoracic primary itself. In CURB, SBRT added to standard systemic therapy increased PFS substantially in oligoprogressive NSCLC, reinforcing the value of local therapy in selected resistant sites, but not specifically establishing primary-tumor irradiation as the decisive component.
So the consensus lands in a clinically reasonable place: for non-AGA stage IV NSCLC, definitive primary-tumor RT is an important investigational strategy, not yet a settled standard.
Timing Matters, but the Best Sequence Is Still Being Defined
One of the most practical questions in clinic is when to deliver RT. Should it be given upfront, during systemic therapy, or only after initial disease control is documented?
The IASLC panel leans toward early integration, especially in EGFR-mutant disease, often in the form of consolidation after induction systemic therapy. That reflects the idea that waiting until overt oligoprogression may miss an earlier window where local control could be more meaningful. At the same time, the statement acknowledges that the optimal sequence remains uncertain, particularly in patients without actionable drivers.
This nuance matters. In practice, earlier RT may help control the most biologically important lesion before resistant clones dominate, but delayed RT after initial systemic response may allow better patient selection and smaller treatment volumes. The consensus does not pretend this tension has been fully resolved. Instead, it identifies timing as one of the most urgent questions for future prospective trials.
Dose and Target Volumes Are Now Central Questions
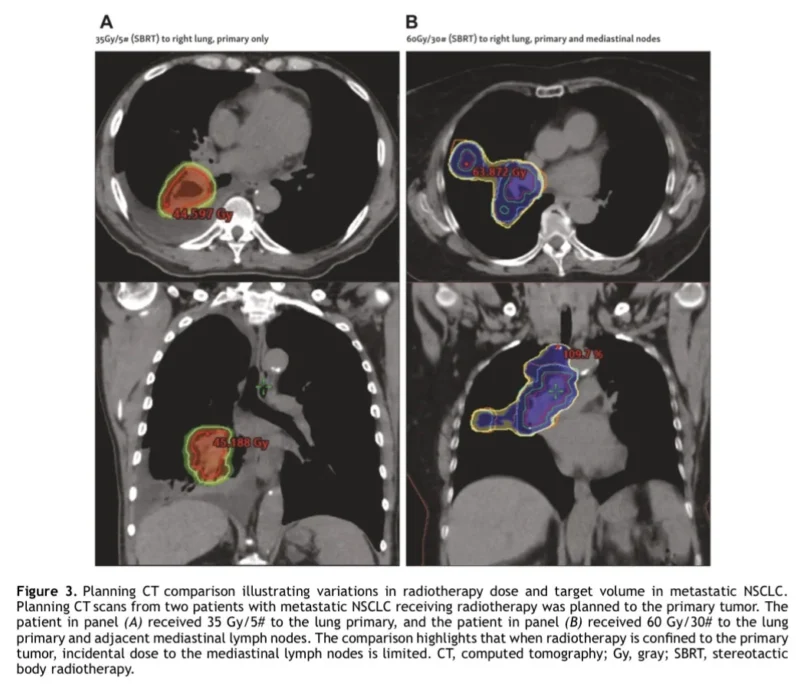
Another important contribution of the paper is that it moves beyond the simple question of whether to irradiate and asks how to do it. The panel suggests that higher-dose thoracic RT may improve outcomes, but also emphasizes that fractionation should be individualized according to tumor location to reduce cardiopulmonary toxicity. A centrally located primary tumor is not the same technical problem as a peripheral lesion, and the consensus makes clear that dose intensity cannot be separated from anatomy.
Target volumes remain another unresolved issue. The statement notes that the optimal thoracic target volume is still uncertain, especially when it comes to whether involved mediastinal lymph nodes should always be included. That question is increasingly relevant in the immunotherapy era, where preserving lymphatic immune architecture may theoretically matter, but prospective validation is still lacking.
In other words, the debate is no longer just “Should we radiate?” It is increasingly “What dose, what field, and at what cost?”
Safety Looks Additive, Not Harmless
One of the more useful clinical messages from the consensus is its pragmatic assessment of safety. The panel states that when RT is combined with systemic therapy, toxicity appears generally additive rather than clearly synergistically amplified, but that this still requires close monitoring. Pneumonitis remains the most obvious shared concern, particularly when thoracic radiation is layered onto drugs that already carry pulmonary risk.
That is an important distinction. Additive toxicity is not trivial toxicity. It means clinicians may be able to combine modalities safely in selected patients, but only with careful sequencing, dose planning, and follow-up. The consensus is especially cautious around combinations involving large thoracic fields and concurrent immunotherapy, where enthusiasm should not outrun safety data.
What This Means for Practice Now
The most useful way to read this IASLC statement is not as a new mandate, but as a marker of where the field is moving. It supports the idea that definitive-dose RT to the primary lung tumor in stage IV NSCLC is no longer a fringe concept. In EGFR-mutant oligometastatic disease, it is already supported by randomized evidence. In other stage IV NSCLC populations, it remains promising, biologically plausible, and increasingly relevant, but not yet mature enough for blanket recommendations.
For multidisciplinary teams, the message is straightforward: the primary tumor deserves more attention than it used to. But patient selection remains everything. Performance status, disease burden, tumor location, systemic therapy plan, and toxicity risk all still matter. The consensus does not simplify metastatic NSCLC. It reflects how much more complex, and potentially more effective, its treatment has become.
You can read full article here