This Phase 2 trial explored a critical question in prostate cancer immunotherapy: can active immune priming with DNA vaccines make Pembrolizumab effective in a disease that is typically resistant to checkpoint blockade?
pRather than relying on PD-1 inhibition alone, the study combined pembrolizumab with tumor-specific vaccines targeting prostate antigens, aiming to generate and amplify a T-cell–mediated anti-tumor response.
The results provide a clear signal: this strategy works biologically—but optimization is key.
Study Design
This randomized Phase 2 trial was designed to evaluate whether broadening tumor-specific T-cell activation could enhance the efficacy of Pembrolizumab in metastatic castration-resistant prostate cancer (mCRPC).
Patients were assigned to one of two strategies:
- Single-antigen approach: pTVG-HP vaccine (targeting prostatic acid phosphatase, PAP) + pembrolizumab
- Dual-antigen approach: pTVG-HP + pTVG-AR (targeting androgen receptor) + pembrolizumab
The primary endpoint was 6-month progression-free survival (PFS), with secondary endpoints including overall survival (OS), PSA response, objective response rate (ORR), safety, and immunologic correlates.
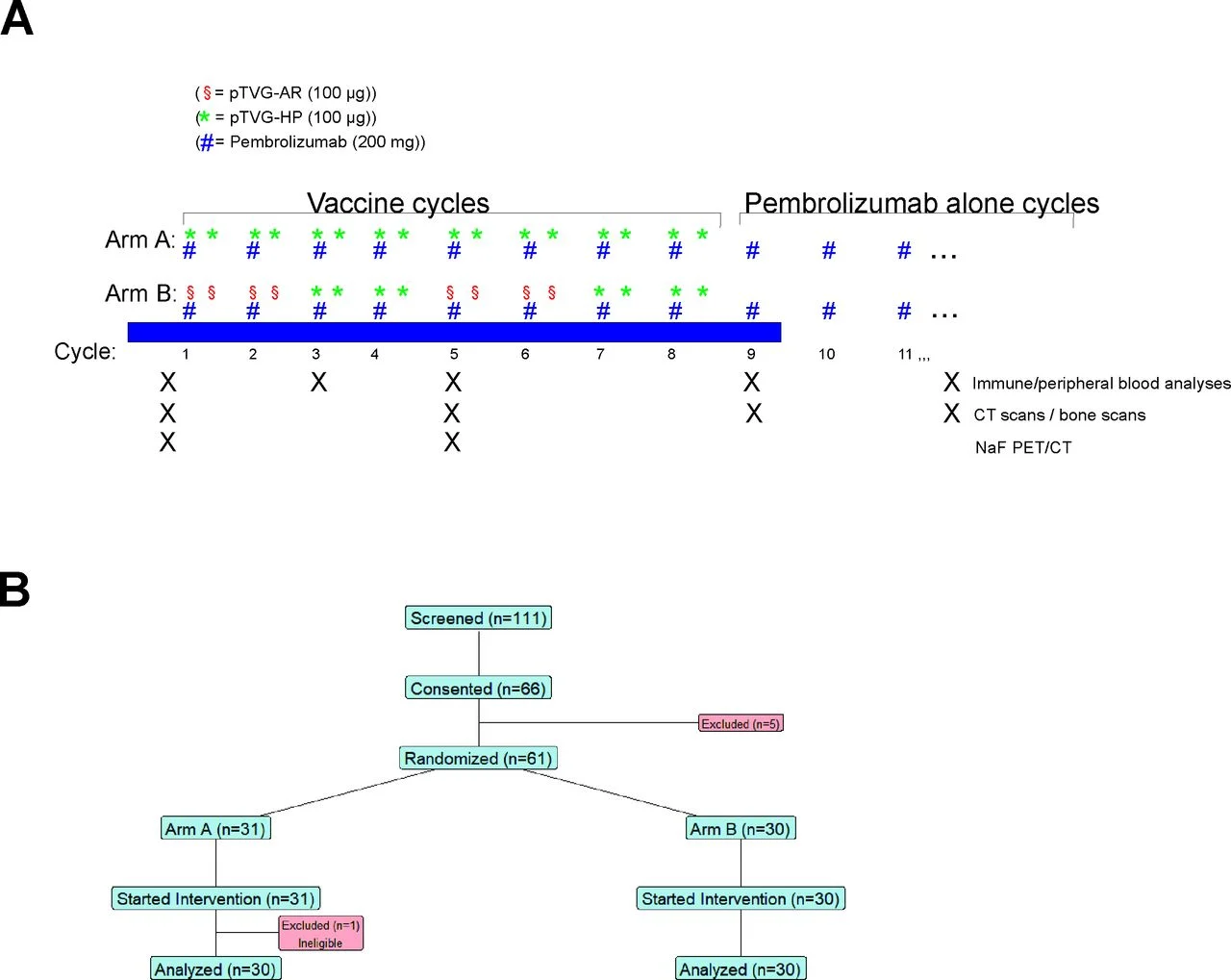
Methods
A total of 60 patients with mCRPC were enrolled and treated in 3-week cycles:
- DNA vaccines were administered on days 1 and 8
- Pembrolizumab was given on day 1 of each cycle
- After 6 months, patients continued pembrolizumab maintenance
- Selected patients received booster vaccinations at PSA progression
Clinical response was assessed using:
- PSA kinetics (every 3–6 weeks)
- Radiographic imaging (RECIST criteria)
- Kaplan–Meier estimates for PFS and OS
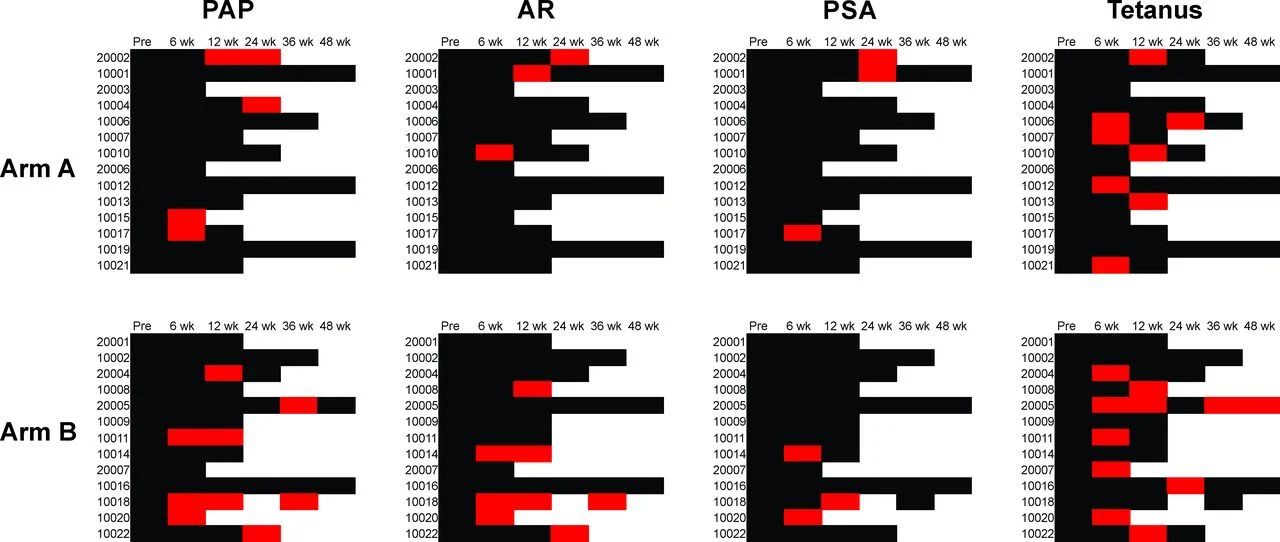
Immune responses were evaluated through:
- Antigen-specific T-cell assays
- Cytokine and chemokine profiling
Results
The study demonstrated consistent and clinically meaningful activity, but importantly, this benefit appeared to be driven by the overall strategy of immune priming rather than by increasing antigen complexity. Across both treatment arms, outcomes were broadly similar, suggesting that adding a second vaccine target did not translate into improved efficacy.
From a disease control perspective, both groups showed comparable results. The median time to progression was approximately 24 weeks in each arm, and the 6-month progression-free survival rates were closely aligned.
6-month PFS:
- 51% (single vaccine)
- 45% (dual vaccine)
These findings indicate that the addition of the second antigen did not enhance the ability to delay disease progression.
A numerical difference in overall survival was observed, with longer survival in the dual-vaccine arm. However, this did not reach statistical significance and should be interpreted with caution.
Median overall survival (OS):
- ~29.9 months (single vaccine)
- ~44.7 months (dual vaccine)
Given the limited sample size and potential baseline differences between groups, this signal remains hypothesis-generating rather than definitive.
Biochemical and radiographic responses further support the presence of real antitumor activity. PSA reductions were seen in a substantial proportion of patients, and objective tumor shrinkage was observed in those with measurable disease.
PSA responses:
- Any decline: ~32%
- ≥50% decline: ~20%
Objective response rate (ORR):
- ~30% in patients with measurable disease
These response rates are notable in mCRPC, particularly in a population not selected for classical immunotherapy-responsive biomarkers.
Importantly, a subset of patients achieved durable disease control, remaining progression-free beyond 6 months. In some cases, patients who received additional booster vaccinations at PSA progression experienced renewed PSA declines, suggesting that the immune response could be reactivated with continued antigen exposure.
Durable benefit:
- ~28% progression-free beyond 6 months
- Evidence of response reactivation with booster vaccination
From a safety standpoint, the combination was generally well tolerated, with most adverse events being low grade. However, the dual-vaccine approach was associated with a modest increase in immune-related toxicity, including signals in tissues that physiologically express androgen receptor.
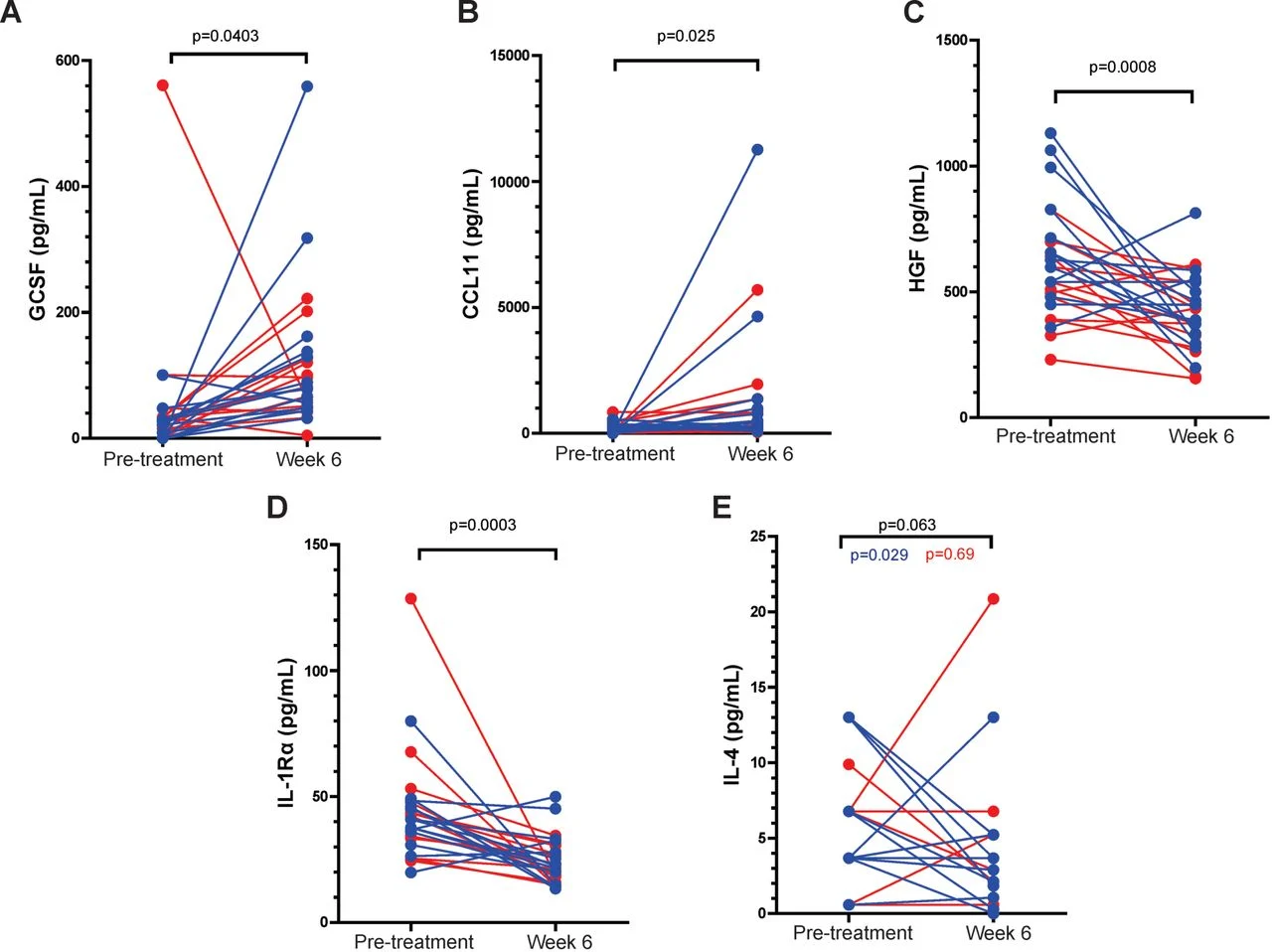
Finally, immunologic analyses provided an unexpected insight. Despite the clinical activity observed, there was limited detectable peripheral T-cell activation, and classic cytokine signatures were not consistently elevated. This suggests that the therapeutic effect may depend more on local tumor immune dynamics or on factors such as timing and scheduling of vaccination, rather than simply the magnitude of systemic immune activation.
When placed in context, these results appear clinically meaningful. Historically, median overall survival in mCRPC trials has been in the range of ~20–30 months, and PD-1 monotherapy has demonstrated minimal activity outside biomarker-selected populations. In contrast, this study reports median OS of ~33.6 months, along with PSA responses (~32%) and an objective response rate of ~30%, suggesting that this is not trivial activity and may reflect a true biologic effect of immune priming combined with checkpoint inhibition.
Limitations
While the study provides important insights, several key limitations should be considered:
- Small sample size (n=60): limits statistical power and may affect reliability of survival comparisons
- No true control arm: absence of pembrolizumab-only or standard-of-care comparator makes it difficult to isolate the vaccine effect
- Not powered for OS: overall survival differences are exploratory and not definitive
- Patient heterogeneity: prior treatments (ARPI, chemotherapy) may influence outcomes
- Limited immune analysis: peripheral blood data may not reflect tumor microenvironment activity
- Suboptimal vaccine schedule: timing may have affected the strength and durability of immune activation
Key Takeaway Message
- Immunotherapy in prostate cancer is limited not by Pembrolizumab itself, but by insufficient immune activation
- DNA vaccine + PD-1 blockade can generate meaningful clinical activity in mCRPC
- Increasing antigen targets (dual vaccines) does not improve efficacy and may increase toxicity
- PSA responses and ORR signals suggest this is not trivial activity
- Success in “cold” tumors depends on effective immune priming, not just checkpoint inhibition
Read Full Article Here


