Immune checkpoint inhibitors (ICIs) have transformed the treatment landscape of advanced hepatocellular carcinoma (HCC), yet substantial variability in therapeutic response remains. While ICIs possess long half-lives measured in weeks, emerging evidence across multiple malignancies has suggested that the timing of immunotherapy administration may unexpectedly influence clinical efficacy.
The biological rationale behind this concept is rooted in circadian immunology. T-cell activation, proliferation, migration, cytokine secretion, and immune responsiveness fluctuate throughout the day under regulation of intrinsic clock genes and systemic circadian signals. Experimental models have demonstrated that antitumor immune responses may be stronger when immune activation occurs during specific circadian windows. However, the mechanistic basis and clinical significance of infusion timing in HCC had remained unclear.
This study investigated whether the timing of the first ICI infusion could influence survival outcomes and immune responses in patients with advanced HCC.

You Can Also Read About Chronotherapy and Nivolumab in Metastatic Gastric Cancer: Does Infusion Timing Matter?
Study Design and Methods
Investigators analyzed patients with advanced or unresectable HCC treated with standard-of-care ICI-based therapies at Johns Hopkins between 2021 and 2025. Patients were stratified according to the timing of their first immunotherapy infusion:
- Morning group: first infusion before 12:00 PM
- Afternoon group: first infusion at or after 12:00 PM
The study evaluated:
- Progression-free survival (PFS)
- Overall survival (OS)
- Objective treatment response
- Immune-related adverse events (irAEs)
To explore biologic mechanisms, investigators also performed extensive immune profiling using:
- Cytometry by time-of-flight (CyTOF)
- Serial cytokine analyses
- Peripheral blood mononuclear cell profiling
- Multiplex plasma immune assays
Importantly, the study focused specifically on the timing of the first infusion (C1D1) rather than cumulative infusion timing, addressing a major unanswered question in circadian immunotherapy research.
Clinical Outcomes
The study included 84 patients, with 39 patients receiving their first infusion in the morning and 45 in the afternoon. Baseline clinical characteristics were balanced between groups, minimizing the likelihood that differences were driven by demographic or disease-related confounding factors.
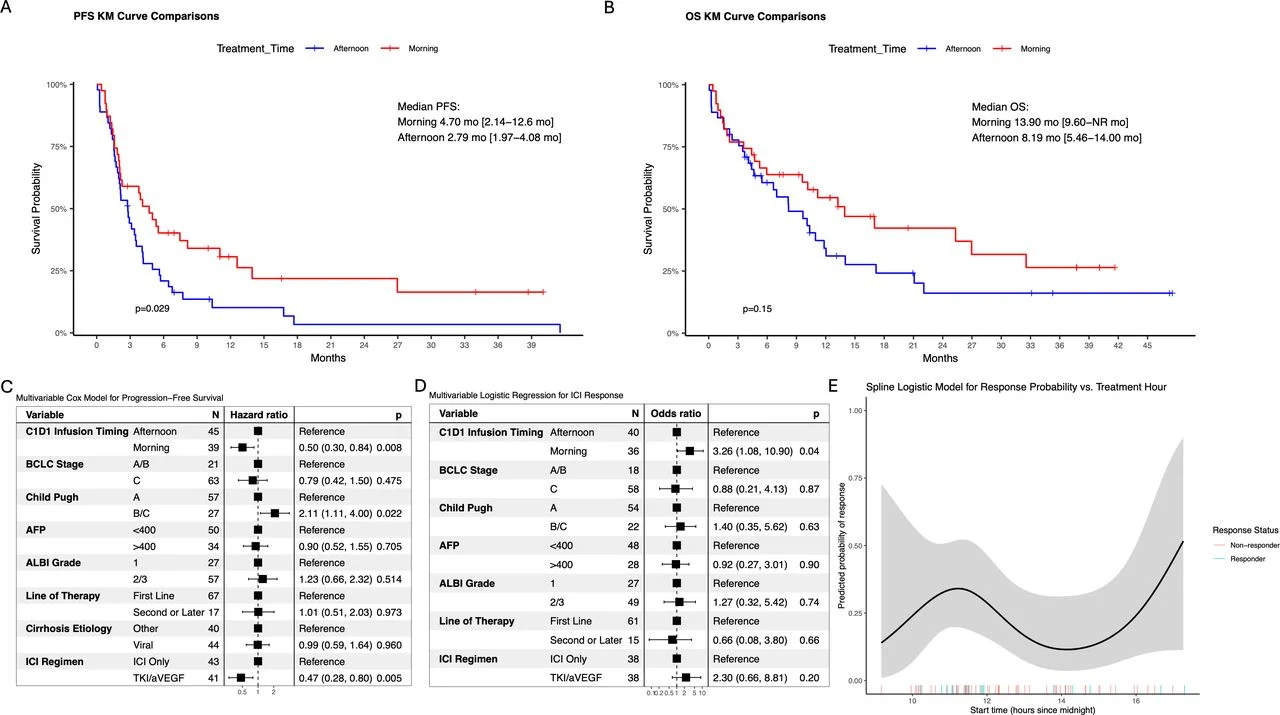
Patients treated in the morning demonstrated significantly superior clinical outcomes compared with those treated later in the day. Morning infusion was associated with improved progression-free survival, higher response rates, and no meaningful increase in clinically significant immune-related toxicity.
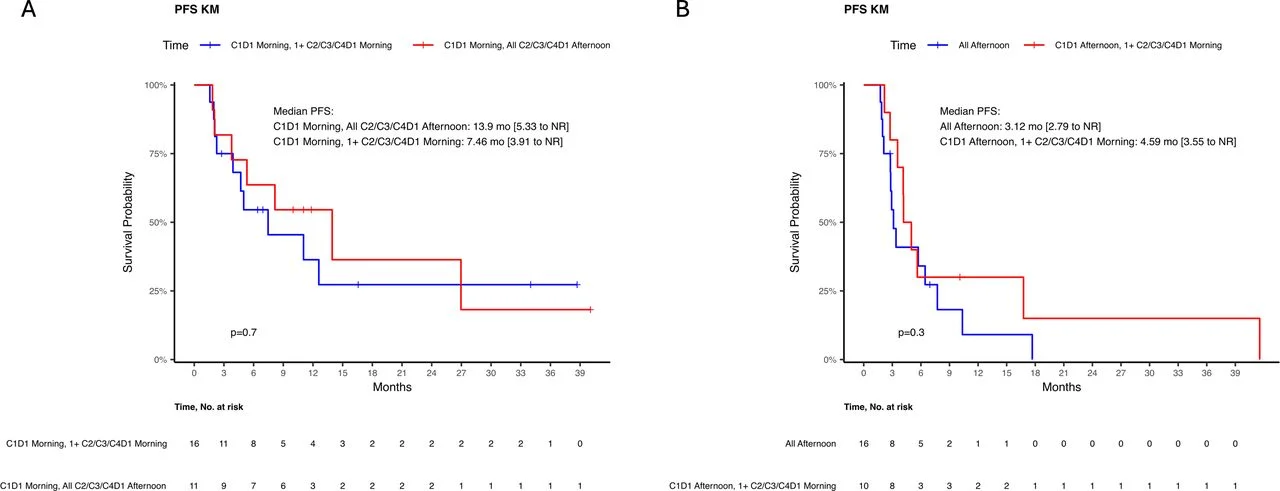
Interestingly, the timing of subsequent infusions after the first dose had no measurable effect on outcomes, suggesting that the initial exposure to checkpoint inhibition may establish an early immune “programming” effect that shapes downstream antitumor immunity.
Key Clinical Results
- Morning first-dose infusion was associated with significantly improved progression-free survival, with a multivariable hazard ratio of 0.50 (95% CI 0.30–0.84; p<0.01). Median PFS was 4.7 months in the morning group versus 2.8 months in the afternoon group.
- Patients treated in the morning demonstrated substantially higher odds of objective response, with a multivariable odds ratio of 3.26 (95% CI 1.08–10.90; p<0.05).
- Late morning administration appeared particularly favorable. Spline modeling suggested that infusions between approximately 10:30 AM and noon were associated with the highest predicted probability of response.
- The timing of later infusions beyond cycle 1 day 1 did not significantly influence outcomes, reinforcing the hypothesis that the first infusion may be biologically decisive.
Immune and Cytokine Findings
One of the most important aspects of this study was the integration of detailed immune profiling to identify biologic correlates underlying the clinical observations.
Investigators demonstrated that morning-treated patients developed a distinct early immune signature characterized by:
- Reduced IL-6 signaling
- Expansion of cytotoxic CD8+ T-cell populations
- Enhanced central memory T-cell responses
IL-6 emerged as a particularly important finding. Patients treated in the afternoon experienced marked IL-6 expansion early during therapy, whereas IL-6 levels decreased in the morning-treated cohort. This observation is clinically relevant because elevated IL-6 has repeatedly been associated with immune suppression, tumor progression, and resistance to checkpoint inhibition across multiple cancers, including HCC.
CyTOF analyses further revealed significant expansion of cytotoxic central memory CD8+ T cells in the morning cohort. Additional trends toward increased cytotoxic effector and effector-memory CD8+ populations were also observed, supporting the concept that circadian timing may influence the quality and durability of T-cell priming after checkpoint blockade.
Safety Profile
Despite superior efficacy outcomes, morning administration was not associated with increased clinically meaningful immune toxicity. The incidence of grade ≥2 immune-related adverse events was similar between groups, and no statistically significant increase in serious toxicity was observed with earlier infusion timing.
This finding is clinically important because it suggests that optimization of infusion timing could potentially improve efficacy without compromising safety.
Biologic and Translational Significance
This study provides some of the strongest mechanistic evidence to date supporting the role of circadian biology in cancer immunotherapy.
The findings suggest that the initial checkpoint inhibitor exposure may occur during a biologically favorable “immune priming window,” when T cells are more capable of entering proliferative, cytotoxic, and durable memory programs. Conversely, later-day administration may coincide with less favorable immune conditions characterized by elevated inflammatory cytokines such as IL-6.
The observation that only the first infusion timing influenced outcomes is particularly intriguing. It suggests that early immune programming rather than cumulative exposure may determine long-term antitumor immunity. This concept aligns with preclinical studies demonstrating that effective checkpoint blockade depends heavily on optimal CD8+ T-cell priming during the earliest phases of immune activation.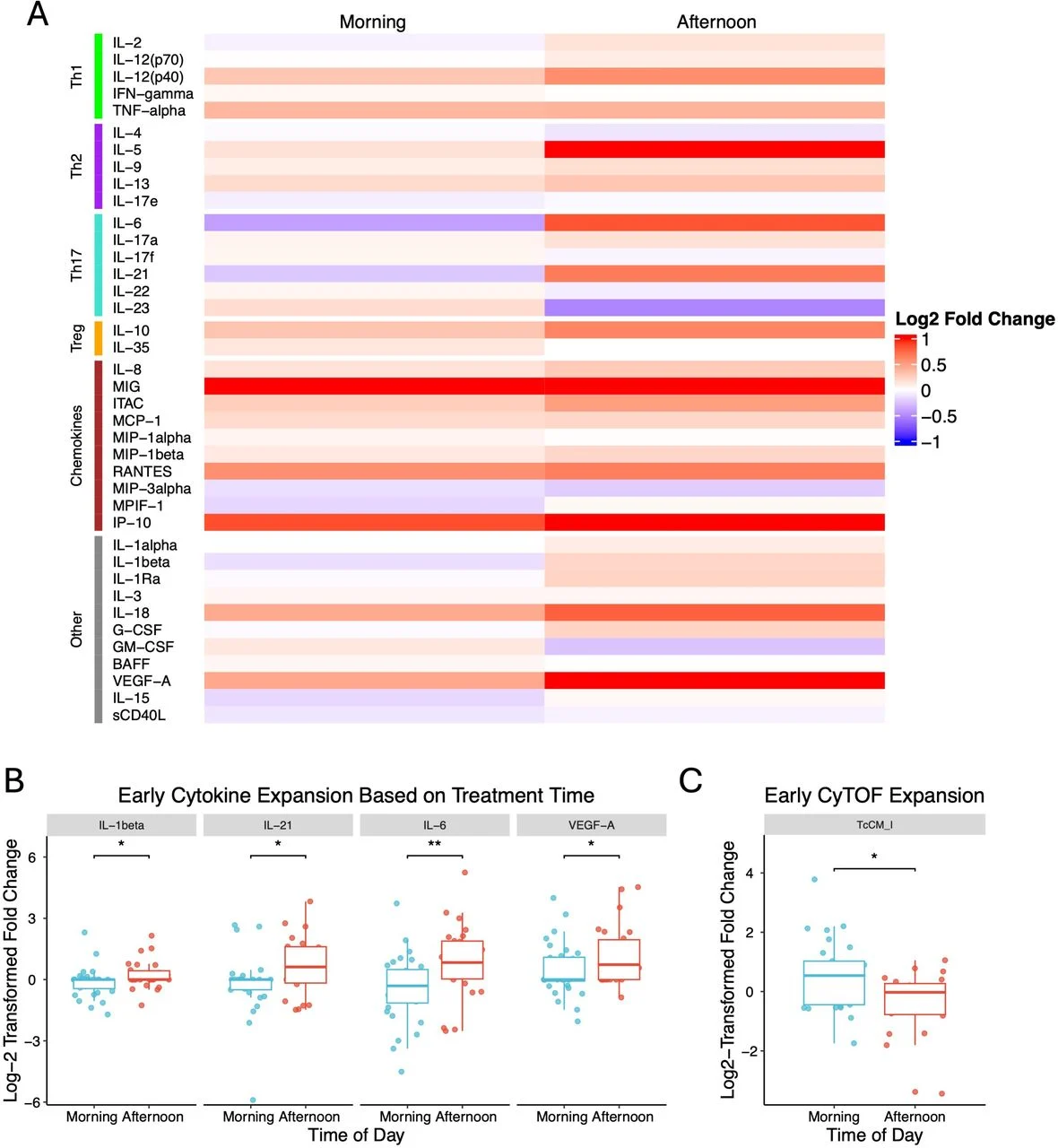
Clinical Implications
Although prospective randomized validation is still required, these findings raise the possibility that a simple scheduling intervention could meaningfully improve immunotherapy efficacy in HCC.
If confirmed, strategically prioritizing morning or late-morning first-dose ICI administration could represent a low-cost, non-pharmacologic optimization strategy capable of enhancing antitumor immune responses without introducing additional toxicity.
More broadly, the study reinforces the growing importance of circadian immunology within oncology and suggests that treatment timing itself may become an important component of precision immunotherapy in the future.
Read Full Article Here


