Immunotherapy transformed the treatment landscape of advanced non-small-cell lung cancer (NSCLC), but it also created a new challenge for clinical research: how can investigators test multiple promising combinations quickly enough to keep pace with a rapidly changing standard of care? The phase II FRACTION-Lung trial was built to answer exactly that question.
Reported by He and colleagues in ESMO Open, FRACTION-Lung trial evaluated nivolumab alone and in combination with dasatinib, ipilimumab, relatlimab, or linrodostat across multiple patient subsets with advanced NSCLC, using an adaptive platform design intended to identify signals of activity efficiently and allow ineffective strategies to be deprioritized early (He et al., 2026). Although the study ultimately showed limited efficacy and was discontinued early, it remains clinically important. Not because it established a new treatment standard, but because it offers a valuable window into how immunotherapy platform trials function in real time, where they succeed, and where they run into the realities of fast-moving thoracic oncology.
For readers, FRACTION-Lung trial is interesting on two levels. The first is therapeutic: how did the different nivolumab-based combinations perform in immunotherapy-naive and previously treated patients? The second is strategic: what does this study teach us about platform trial methodology in lung cancer?
Read About Non-Small Cell Lung Cancer on OncoDaily
Why FRACTION-Lung Mattered At The Time It Was Designed
When FRACTION-Lung trial was launched in 2016, immunotherapy was already beginning to reshape metastatic NSCLC, but the field had not yet stabilized. Anti-PD-1 and anti-PD-L1 therapy had entered practice, but many major combination standards had not yet fully matured. This created a fertile moment for signal-seeking studies exploring ways to deepen or extend immune responses.
The FRACTION program was designed as a rolling, adaptive, phase II platform study across tumor types. In the lung cohort, patients were assigned to different treatment tracks based on prior exposure to immunotherapy, PD-L1 status, and prior lines of therapy. The goal was not to provide a definitive head-to-head comparison between regimens, but to rapidly screen multiple nivolumab-based combinations and determine which, if any, deserved further development (He et al., 2026).
This was an ambitious and rational concept. Traditional drug development is often too slow for immuno-oncology, especially when multiple mechanisms are competing for attention at once. A platform design can, in theory, add new arms, close ineffective ones, and redistribute patients more efficiently than running multiple separate studies. FRACTION-Lung trial also included a particularly patient-centered feature: those who progressed on one arm could be re-randomized into another arm within the same trial if they remained eligible.
That feature deserves recognition. In advanced lung cancer, where patients often move rapidly through lines of therapy, a design that allows continued participation without requiring entry into an entirely new study is both practical and humane.
The Biological Logic Behind The Regimens
The combinations selected for FRACTION-Lung trial reflect the scientific thinking of the time.
Nivolumab plus ipilimumab was based on the now familiar idea of dual immune checkpoint blockade, targeting PD-1 and CTLA-4 to enhance T-cell priming and effector activity. Long before this became an established option in NSCLC, the combination had already shown meaningful benefit in melanoma, making it one of the most biologically credible strategies to test in lung cancer (He et al., 2026).
Nivolumab plus relatlimab targeted PD-1 and LAG-3, another inhibitory pathway involved in T-cell exhaustion. This was a forward-looking choice. At the time, LAG-3 was an emerging target, and the rationale for combining it with PD-1 inhibition was strong, even if clinical validation in thoracic oncology was still immature.
Nivolumab plus linrodostat explored inhibition of IDO1, a pathway that had generated major enthusiasm as a means of reversing tumor-associated immune suppression. In retrospect, this arm also reflects a broader lesson in immuno-oncology: even mechanistically elegant ideas can fail to translate into meaningful clinical benefit.
Nivolumab plus dasatinib may appear less intuitive on first glance, but it was supported by preclinical evidence suggesting immunomodulatory effects and possible reshaping of the tumor microenvironment. Dasatinib’s role as a multi-kinase inhibitor made it an intriguing partner for PD-1 blockade, especially in a signal-seeking setting where unconventional immune combinations were worth testing.
Seen together, these arms illustrate a key strength of platform trials: they allow multiple biological hypotheses to be tested under one operational umbrella.
Study Population And Trial Structure
FRACTION-Lung trial enrolled 295 treated patients with advanced NSCLC and Eastern Cooperative Oncology Group performance status 0 or 1. The study included both patients without prior immunotherapy and those previously exposed to immunotherapy. Some patients were re-randomized after progression, reinforcing the adaptive nature of the design (He et al., 2026).
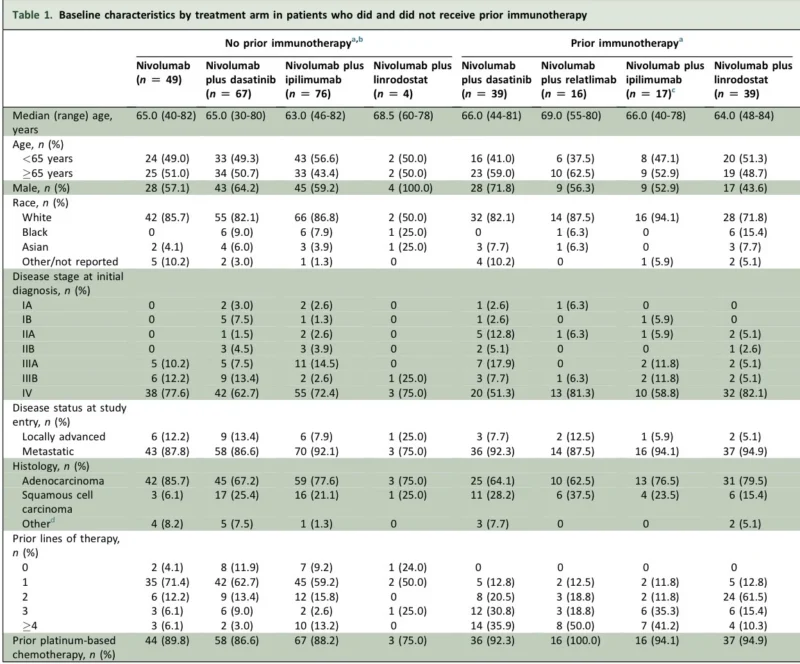
Patients without prior immunotherapy were enrolled into tracks defined partly by PD-L1 status and prior chemotherapy exposure, while those with prior immunotherapy entered later tracks examining combinations in a more resistant disease setting. Baseline characteristics were broadly similar across arms, with most patients having metastatic disease at study entry, adenocarcinoma histology, and prior platinum-based chemotherapy.
One point that stands out is how heavily pretreated many patients were, particularly in the prior-immunotherapy cohorts. This matters clinically. It is difficult to rescue robust efficacy in highly selected resistant populations, particularly with relatively small sample sizes and in a disease as biologically heterogeneous as advanced NSCLC.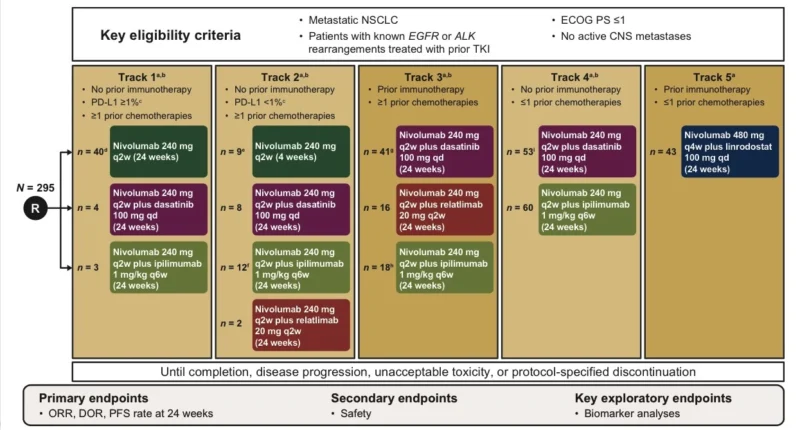
Efficacy Signals Were Modest Across Most Arms
The central clinical finding of FRACTION-Lung trial is straightforward: efficacy was limited.
Among patients without prior immunotherapy, objective response rates ranged from 0% in several arms to 25% in very small cohorts treated with nivolumab plus ipilimumab or nivolumab plus dasatinib. In the more informative larger groups, nivolumab plus ipilimumab produced a 20.0% ORR in track 4, while nivolumab monotherapy showed a 17.5% ORR in PD-L1-positive track 1 and no responses in PD-L1-negative track 2 (He et al., 2026).
Among patients with prior immunotherapy, activity was even more modest. ORRs ranged from 2.3% with nivolumab plus linrodostat to 5.6% with nivolumab plus ipilimumab. Median duration of response could not be meaningfully estimated because responses were too few.
These results immediately place FRACTION-Lung trial in context. This was not a study that identified a breakthrough salvage strategy. It was a study that showed how difficult it is to generate robust efficacy in advanced NSCLC once resistance has emerged, and how many theoretically attractive combinations fail to deliver clinically meaningful response rates.
The progression-free survival data tell a similar story. In immunotherapy-naive patients, 24-week PFS rates ranged from 30.2% with nivolumab monotherapy to 45.5% with nivolumab plus ipilimumab in a very small track 2 subgroup and 39.1% in the larger track 4 nivolumab-plus-ipilimumab cohort. In previously treated patients, 24-week PFS rates were only 19.1% with nivolumab plus dasatinib and 11.1% with nivolumab plus linrodostat (He et al., 2026).
These numbers are not practice-changing. But they are informative. They suggest that among the tested strategies, nivolumab plus ipilimumab had the most consistent signal in immunotherapy-naive disease, while the other combinations offered little evidence of meaningful rescue benefit after prior checkpoint therapy.
Nivolumab Plus Ipilimumab Emerged As The Most Credible Combination
If one regimen in FRACTION-Lung trial looks most clinically relevant in hindsight, it is nivolumab plus ipilimumab.
That is not only because it produced some of the better efficacy signals in the trial, but also because the broader field eventually validated this combination in registrational studies. By the time FRACTION-Lung trial was progressing, CheckMate 227 had already helped establish nivolumab plus ipilimumab as an important option in metastatic NSCLC, and this evolving external evidence contributed to the eventual discontinuation of the study (He et al., 2026).
This is one of the most interesting aspects of FRACTION-Lung trial. The trial was not terminated because of a major safety disaster. It was overtaken by progress. In a rapidly moving therapeutic environment, platform trials must compete not only with biological uncertainty but with the speed of external clinical development. A combination can go from experimental to standard-of-care before a phase II signal-seeking platform has fully matured.
That is both a challenge and a compliment to the field. It means thoracic oncology is advancing quickly. But it also means adaptive platforms need to be even more agile if they are to remain relevant.
Why The Other Combinations Fell Short
The limited performance of the non-ipilimumab combinations is also clinically revealing.
The relatlimab arms were too small to support strong conclusions, which is frustrating because the biology remains interesting. LAG-3 is now a validated target in other tumors, but FRACTION-Lung trial could not adequately determine whether nivolumab plus relatlimab had meaningful activity in advanced NSCLC. This is not proof of failure as much as proof of underpowered exploration.
The linrodostat results were disappointing, particularly in patients with prior immunotherapy, where ORR was only 2.3%. This aligns with a broader historical pattern: the IDO1 pathway generated substantial enthusiasm, but clinical development in solid tumors has been repeatedly undermined by weak efficacy signals despite elegant biology.
The dasatinib combination produced scattered signals but no convincing pattern of durable benefit. The pleural effusion signal and the overall modest efficacy make it difficult to argue for further prioritization in NSCLC based on these data alone.
In that sense, FRACTION-Lung trial performed exactly the job a platform trial is supposed to do. It separated regimens with limited future promise from those more worthy of continued attention.
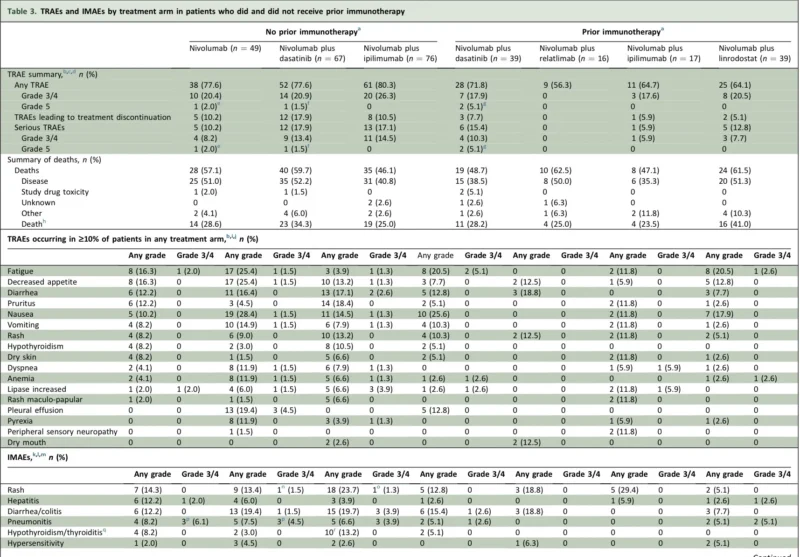
Safety Was Manageable But Not Trivial
Safety findings were generally consistent with the known profiles of checkpoint inhibitors and their combinations. No major unexpected safety signals emerged, but toxicities were not negligible.
Among immunotherapy-naive patients, any-grade treatment-related adverse events occurred in roughly 78% to 80% across arms. Grade 3 or 4 treatment-related events occurred in about 20% to 26%, and treatment discontinuation due to toxicity ranged from 10.2% to 17.9% (He et al., 2026). In previously treated patients, toxicity rates were somewhat lower overall, but still meaningful.
The most common treatment-related events included fatigue, nausea, diarrhea, and decreased appetite. Pleural effusion was seen only in the dasatinib-containing arms, which is notable because it reinforces the specific toxicity footprint that kinase inhibitor combinations can bring into immunotherapy regimens.
Immune-mediated adverse events were also in line with expectations, including rash, diarrhea or colitis, hepatitis, hypothyroidism, and pneumonitis. Four deaths were attributed to study drug toxicity, including pneumonitis and respiratory failure, underscoring that even exploratory immunotherapy trials require close safety vigilance (He et al., 2026).
For practicing oncologists, the takeaway is familiar but important: the threshold for accepting toxicity in a combination regimen must always be matched by a credible efficacy signal. In FRACTION-Lung trial, that tradeoff was not favorable for most of the tested regimens.
Biomarker Work Was Interesting But Limited By Scale
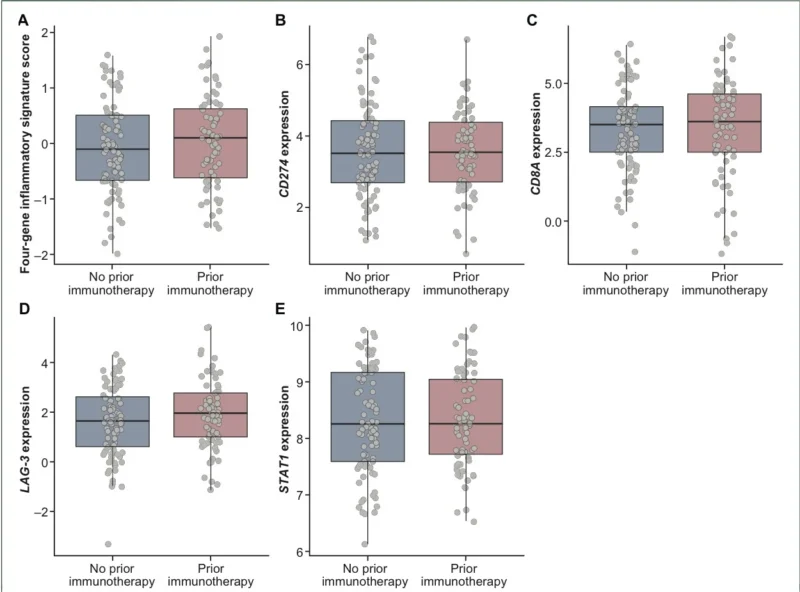
The biomarker component of FRACTION-Lung trial was exploratory, and the study’s small numbers prevented definitive conclusions. Still, the translational work offers some useful observations.
Tumor samples showed variability in CD8, Ki-67/CD8, and LAG-3 expression, especially among patients previously treated with immunotherapy. Peripheral blood flow cytometry suggested lower frequencies of peripheral PD-1-positive effector memory cells in previously treated patients, while some broader T-cell subset distributions were similar between cohorts (He et al., 2026).
The inflammatory four-gene signature did not show a clear difference between immunotherapy-naive and previously treated groups. This is not necessarily surprising. In NSCLC, resistance biology is unlikely to be captured by a single compact inflammatory signature, especially in small heterogeneous cohorts treated across multiple different arms.
Still, the translational effort was worth including. One of the key purposes of platform trials is not only to detect clinical signals but to generate hypotheses about which patients may benefit from which strategies. FRACTION-Lung could not fully deliver on that goal, but it pointed in the right direction.
The Most Important Result May Be Methodological
Paradoxically, the greatest value of FRACTION-Lung trial may not be the efficacy data at all. It may be the methodological lesson.
This study showed that adaptive platform designs are feasible in advanced NSCLC. They can enroll biologically distinct subgroups, test multiple regimens under one umbrella, permit re-randomization, and produce clinically interpretable screening data. That is a real success.
At the same time, FRACTION-Lung trial also exposed the vulnerabilities of this model. Small sample sizes within individual arms limit interpretability. Changing standards of care can rapidly make tracks obsolete. Early termination can leave promising hypotheses unresolved. And in a disease as dynamic as advanced NSCLC, the pace of external approvals may outstrip the pace of internal platform learning.
These are not arguments against platform trials. They are arguments for building smarter ones. Future lung cancer platforms will need more integrated biomarker selection, faster adaptation rules, stronger statistical planning for evolving benchmarks, and perhaps tighter alignment with registrational development pathways.
Final Perspective
FRACTION-Lung trial did not identify a new standard-of-care regimen for advanced NSCLC. Most of the tested nivolumab-based combinations showed limited efficacy, especially after prior immunotherapy, and the study was ultimately overtaken by a rapidly evolving treatment landscape. But that does not make it a failed trial.
It was a useful trial. It demonstrated that adaptive platform studies can operate in thoracic oncology. It provided an early testing ground for several immunotherapy combinations. It reinforced the difficulty of overcoming checkpoint resistance with empiric add-on strategies. And it highlighted nivolumab plus ipilimumab as the only regimen among those tested with a signal that ultimately aligned with broader phase III validation (He et al., 2026).
Clinically, it reminds us that not every rational immunotherapy combination will translate into meaningful benefit. Strategically, it shows that the future of lung cancer research depends not only on better drugs, but also on better ways to test them.
Full article available on ESMO Open