Colorectal cancer remains a major global health burden and a leading cause of cancer-related mortality. Liver metastases occur in approximately half of patients during the disease course and represent a key determinant of survival. For selected patients with limited colorectal liver metastases (CRLM), curative-intent local therapies such as surgical resection or thermal ablation are recommended in current clinical guidelines.
Radiofrequency ablation (RFA) is widely used for small or unresectable liver metastases and offers the potential for local disease control with a minimally invasive approach. However, recurrence after ablation remains common, highlighting the need for improved biomarkers that can identify patients at higher risk of relapse and guide treatment decisions.
Circulating tumor DNA (ctDNA) has emerged as a promising liquid biopsy biomarker in colorectal cancer, particularly for detecting minimal residual disease and predicting recurrence after curative-intent treatment. Mutations in KRAS, one of the most frequently altered oncogenes in colorectal cancer, may provide additional prognostic information when detected through ctDNA analysis.
The study was published on March 9, 2026, in JCO Precision Oncology.
Title: Pretreatment KRAS-Mutant Circulating Tumor DNA Predicts Incidence and Patterns of Recurrence in Patients Undergoing Radiofrequency Ablation for Colorectal Liver Metastases: A Prospective Analysis.
Authors: Ke Lin, MB, Cailun Wang, MB, Bin Qiao, MD, Nan Zhang, MD, Daopeng Yang, MD, DongDong Jin, MMed, Cong Li, MB, Jingsen Lv, MB, Minghui He, MB, Zhao Lin, MB, Xiaohua Xie, MD, Xiaoyan Xie, MD, Bowen Zhuang, MD
Methods
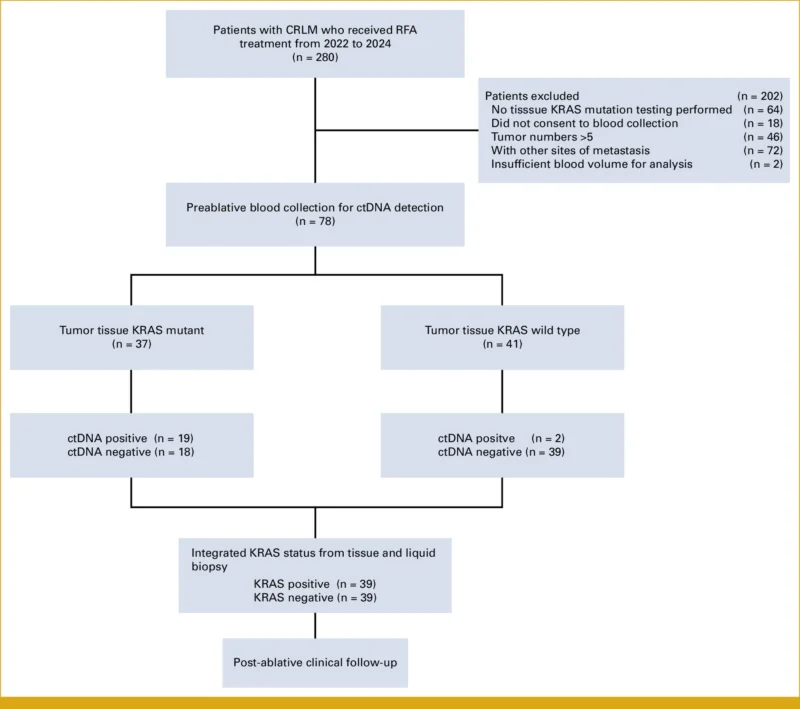
This prospective study included 78 patients with colorectal liver metastases who underwent curative-intent radiofrequency ablation (RFA). These patients were selected from a cohort of 280 individuals treated between January 2022 and December 2024, based on the availability of plasma samples and tissue specimens for genomic analysis.
Pretreatment plasma samples were analyzed using droplet digital polymerase chain reaction to detect KRAS mutations in circulating tumor DNA. KRAS status was also evaluated in tumor tissue, including primary tumor samples and liver biopsies when available.
Patients were followed radiologically using contrast-enhanced CT or MRI performed one month after RFA to confirm treatment success, followed by imaging every three months during the first two years and every six months thereafter.
Endpoints
The primary clinical endpoint was progression-free survival (PFS), defined as the time from RFA to the first imaging-confirmed recurrence at any site. Recurrence patterns were also evaluated. Associations between ctDNA status, clinicopathologic variables, and survival outcomes were assessed using Kaplan–Meier analysis and Cox proportional hazards models.
To explore biological mechanisms associated with ctDNA detection, tumor samples underwent transcriptomic profiling, with differential gene expression and pathway enrichment analyses performed using RNA sequencing data.
What is Radiofrequency Ablation?
Ablation therapy is a minimally invasive cancer treatment that destroys tumors directly in place rather than removing them surgically. Instead of cutting out the tumor, doctors use specialized probes to deliver energy into the cancerous tissue. This energy damages the cancer cells, causing them to break down and die while sparing as much surrounding healthy tissue as possible.
Several types of ablation therapy are used in clinical practice, depending on the type of energy applied to the tumor. The most commonly used approaches include:
- Radiofrequency ablation (RFA) – uses high-frequency electrical currents to generate heat and destroy cancer cells.
- Microwave ablation (MWA) – uses microwave energy to heat tumor tissue.
- Cryoablation – freezes cancer cells using extremely cold gases.
- Laser ablation – uses focused laser energy to heat and destroy tumor tissue.
- Irreversible electroporation (IRE) – uses electrical pulses to disrupt cancer cell membranes.
- High-intensity focused ultrasound (HIFU) – focuses ultrasound waves to heat and destroy tissue deep inside the body.
Among these techniques, radiofrequency ablation (RFA) is one of the most widely used and well-established methods. During the procedure, a needle-like probe is inserted directly into the tumor, usually guided by imaging such as CT or ultrasound. The probe delivers high-frequency electrical currents, which generate heat inside the tumor. This heat damages and destroys the cancer cells while minimizing injury to surrounding tissues.
RFA is commonly used to treat tumors in the liver, kidneys, and lungs, especially when surgery is not possible or when a minimally invasive approach is preferred. Because it can precisely target tumor tissue with relatively limited recovery time, RFA has become an important option in the management of several cancers, including colorectal liver metastases.
Read more about Ablation Therapy for Cancer: A Minimally Invasive Treatment Option on OncoDaily.
Results
A total of 78 patients with colorectal liver metastases were included in the analysis. The median age was 60 years (range 34–85), and 66.7% of patients had synchronous liver metastases. Neoadjuvant systemic therapy was administered in 56.4% of patients, while 80.8% received adjuvant chemotherapy following radiofrequency ablation.
Pretreatment ctDNA positivity was detected in 26.9% of patients (21/78). When combining tissue and plasma analyses, KRAS mutations were identified in 50% of the cohort. The concordance rate between tissue-based KRAS testing and ctDNA analysis was 74.4%.
Recurrence and Progression-Free Survival
During a median post-ablation follow-up of 654 days, 33 patients (42.3%) experienced disease recurrence.
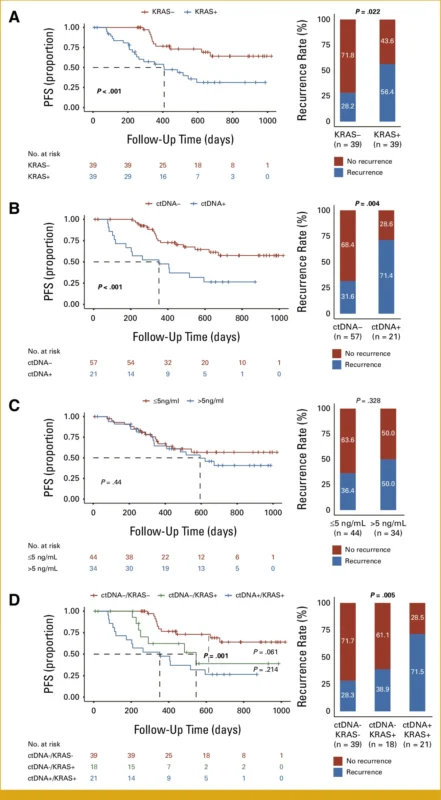
Pretreatment ctDNA detection was strongly associated with recurrence risk. 71.4% of patients with detectable ctDNA developed recurrence, compared with 31.6% of ctDNA-negative patients. KRAS-positive tumors were associated with higher recurrence rates than KRAS wild-type tumors, with recurrence observed in 56.4% versus 28.2% of patients, respectively.
Kaplan–Meier analysis demonstrated that ctDNA positivity was associated with significantly shorter progression-free survival. The median PFS was 352 days in ctDNA-positive patients, while the median PFS was not reached in ctDNA-negative patients.
Multivariable Cox regression analysis identified two independent predictors of recurrence: the presence of more than three hepatic lesions (adjusted HR 7.66, 95% CI 2.70–21.8; P < .001) and pretreatment ctDNA positivity (adjusted HR 3.20, 95% CI 1.60–6.42; P = .001).
When ctDNA and KRAS status were analyzed together, distinct prognostic groups emerged. Patients with ctDNA-negative/KRAS wild-type tumors had the lowest recurrence rate (28.2%), followed by ctDNA-negative/KRAS-positive cases (38.9%), whereas ctDNA-positive/KRAS-positive tumors had the highest recurrence risk (71.4%).
Patterns of Recurrence
The investigators also evaluated the site of relapse among patients who developed recurrence.
Among the 33 recurrent cases:
• 45.5% developed isolated liver metastases
• 36.4% developed isolated lung metastases
• 18.2% had metastases at other sites
Pretreatment ctDNA status was associated with distinct metastatic patterns. Patients with detectable ctDNA were significantly more likely to develop lung metastases, with 60% experiencing isolated pulmonary recurrence compared with 16.7% among ctDNA-negative patients. In contrast, liver-dominant recurrence was more common in ctDNA-negative patients.
Transcriptomic Findings
Transcriptomic profiling revealed 383 differentially expressed genes between ctDNA-positive and ctDNA-negative tumors.
Tumors from ctDNA-negative patients showed enrichment of immune-related pathways, including inflammatory response and interferon-gamma signaling. In contrast, ctDNA-positive tumors demonstrated enrichment of epithelial–mesenchymal transition and DNA repair pathways, which are commonly associated with aggressive tumor biology.
Notably, genes such as NNMT and TSPEAR, previously linked to tumor invasion and metastasis, were significantly overexpressed in the ctDNA-positive group.
Conclusion
This prospective analysis demonstrates that pretreatment KRAS-mutant circulating tumor DNA is a prognostic biomarker in patients with colorectal liver metastases undergoing radiofrequency ablation.
Detectable ctDNA before treatment was independently associated with higher recurrence risk, shorter progression-free survival, and a greater likelihood of lung-predominant metastatic relapse. Integrating ctDNA with KRAS mutation status further improved risk stratification.
These findings suggest that pretreatment ctDNA assessment may help refine patient selection for local ablative therapy, while also guiding surveillance strategies and systemic treatment considerations. Future studies incorporating longitudinal ctDNA monitoring and multicenter validation are needed to further refine ctDNA-guided treatment approaches in metastatic colorectal cancer.
Full article is available in JCO Precision Oncology.