Cervical cancer remains a major global health challenge, with more than 650,000 new cases diagnosed annually. For patients with locally advanced disease who are not candidates for surgery, concurrent chemoradiotherapy with cisplatin is the established standard of care. Since the landmark trials of the 1990s demonstrated superior survival with combined therapy over radiotherapy alone, cisplatin has served as the cornerstone radiosensitizing agent.
Despite decades of clinical use, the optimal dose and schedule of cisplatin during radiotherapy remains uncertain. Two regimens have dominated clinical practice: weekly cisplatin at a dose of 40 mg/m² and tri-weekly cisplatin at a dose of 75 mg/m² administered every three weeks.
Weekly administration became widely adopted because of its perceived tolerability; however, robust phase III comparisons between these schedules have been lacking. The TACO trial (GCIG/KGOG 1027/THAI 2012) was designed to address this evidence gap by evaluating survival outcomes, toxicity, and quality of life associated with each regimen.
Study Design and Patient Population
This prospective, randomized phase III trial enrolled 314 patients with FIGO stage IIB–IVA cervical cancer between 2012 and 2019. Participants were randomly assigned in a 1:1 ratio to receive cisplatin concurrently with radiotherapy according to one of two schedules. Patients in the weekly arm received cisplatin at 40 mg/m² administered weekly for six cycles, while those in the tri-weekly arm received cisplatin at 75 mg/m² every three weeks for three cycles.
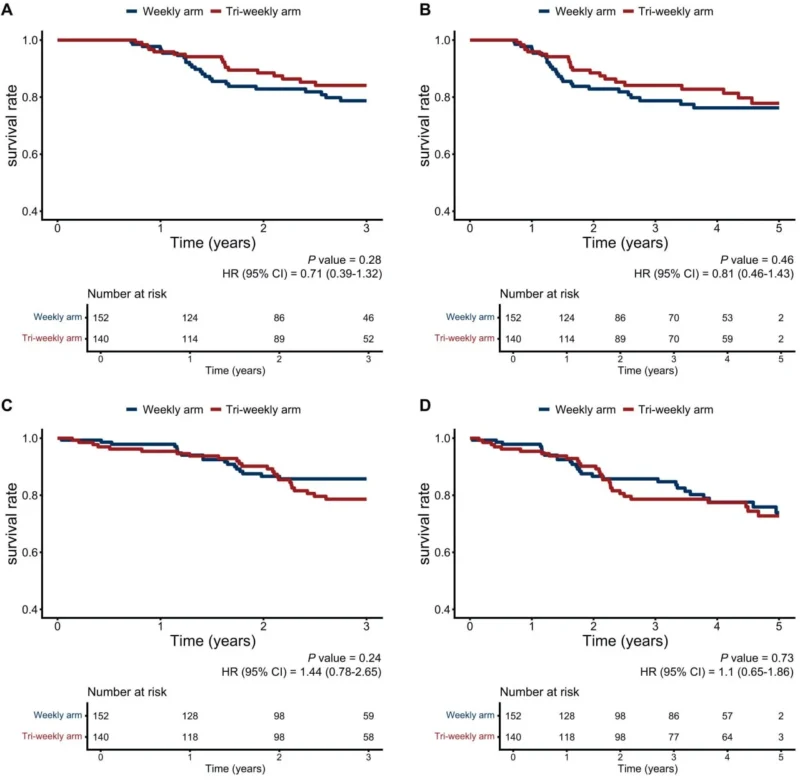
All patients underwent standard external beam radiotherapy followed by intracavitary brachytherapy. Eligibility criteria required adequate performance status, defined as an ECOG score of 0 to 2, along with sufficient hematologic, renal, and hepatic function. The primary endpoint of the study was three-year recurrence-free survival, while secondary endpoints included overall survival, toxicity profiles, treatment compliance, and patient-reported quality of life.
Treatment Compliance
Both treatment regimens demonstrated high completion rates, although important differences emerged in treatment delivery. Completion of planned chemotherapy cycles was achieved in 77.0 percent of patients in the weekly arm compared with 85.7 percent in the tri-weekly arm. Adequate compliance, defined as receiving at least four cycles in the weekly arm or at least two cycles in the tri-weekly arm, was similarly high in both groups, reaching 91.4 percent and 95.0 percent, respectively.
Chemotherapy delays occurred significantly more often in the weekly arm, affecting 27.0 percent of patients compared with 13.6 percent in the tri-weekly arm. Radiotherapy adherence, however, was comparable between groups. These findings suggest that the tri-weekly schedule may be easier to deliver consistently during concurrent chemoradiotherapy.
Survival Outcomes
The trial did not demonstrate superiority of either regimen with respect to disease control or survival. The three-year recurrence-free survival rate was 78.7 percent in the weekly arm and 84.1 percent in the tri-weekly arm, a difference that did not reach statistical significance. Similarly, five-year overall survival rates were nearly identical, at 73.9 percent for weekly cisplatin and 72.7 percent for tri-weekly cisplatin.
Rates of recurrence, persistent disease, and patterns of failure were also comparable between the two groups. These findings indicate that increasing peak cisplatin dose through tri-weekly administration does not translate into improved survival outcomes when the cumulative cisplatin dose exceeds approximately 200 mg/m², a threshold achieved by both regimens.
Toxicity Profile
Safety outcomes revealed clinically meaningful differences between the treatment schedules. Severe grade 3 and 4 toxicities occurred more frequently in the weekly arm, particularly hematologic adverse events. Grade 3–4 hematologic toxicity was observed in 49.3 percent of patients receiving weekly cisplatin compared with 30.4 percent of those receiving tri-weekly treatment, a highly significant difference.
Although severe gastrointestinal toxicity was somewhat more common in the tri-weekly arm, the overall safety profile favored the tri-weekly regimen. Lower rates of neutropenia and thrombocytopenia likely contributed to fewer treatment delays and improved tolerability.
Radiation Therapy Toxicity Myth-Busting
Quality of Life Outcomes
A notable strength of the TACO study was its comprehensive evaluation of patient-reported outcomes using validated instruments, including the EORTC QLQ-C30 and the cervical cancer–specific CX24 module. Overall, quality of life measures favored the tri-weekly regimen across several domains, including global health status, physical functioning, role functioning, and dyspnea.
Certain symptoms, such as appetite loss, diarrhea, and slightly worse cognitive scores, were more pronounced in the tri-weekly arm. Nevertheless, the overall pattern suggested a lower treatment burden and improved patient experience with tri-weekly dosing. These findings highlight the importance of incorporating patient-reported outcomes into treatment decisions, particularly when survival outcomes are similar.
Clinical Interpretation
The absence of a survival advantage with tri-weekly cisplatin challenges the assumption that higher peak drug concentrations necessarily improve outcomes during chemoradiotherapy. Instead, treatment efficacy appears to depend more on achieving an adequate cumulative dose and ensuring successful completion of therapy.
Both schedules provide comparable oncologic effectiveness, but tri-weekly dosing reduces severe hematologic toxicity and is associated with fewer treatment delays. The improved quality of life observed with tri-weekly administration further supports its clinical relevance. Given that cervical cancer disproportionately affects low- and middle-income regions, treatment strategies that minimize toxicity and healthcare utilization without compromising efficacy are particularly valuable.
Limitations and Future Directions
Although the TACO trial provides high-level evidence, several important questions remain. Long-term survivorship outcomes beyond five years have yet to be fully characterized, and cost-effectiveness across diverse healthcare systems requires further evaluation. Optimal patient selection for each regimen also remains unclear, as does the integration of cisplatin-based chemoradiotherapy with emerging treatments such as immunotherapy.
Future research may also explore whether biological factors, including human papillomavirus status or tumor radiosensitivity, influence responses to different cisplatin schedules.
The TACO phase III trial demonstrates that tri-weekly cisplatin at 75 mg/m² administered concurrently with radiotherapy is not superior to weekly cisplatin at 40 mg/m² in terms of survival for patients with locally advanced cervical cancer. However, the tri-weekly regimen offers meaningful advantages in safety and patient-reported quality of life.
These findings support tri-weekly cisplatin as a feasible and well-tolerated alternative to the traditional weekly regimen. Treatment decisions should therefore be individualized, taking into account efficacy, toxicity, logistical considerations, and patient preferences, particularly in settings where minimizing treatment burden is essential.
Optimal cisplatin scheduling in cervical cancer should be based not only on survival outcomes but also on toxicity and quality of life considerations. Weekly and tri-weekly regimens demonstrate comparable efficacy, while tri-weekly administration is associated with fewer severe hematologic toxicities, fewer treatment delays, and generally improved patient-reported outcomes.
Written by Nare Hovhannisyan, MD



