Metastatic uveal melanoma remains one of the most difficult diseases in oncology to treat. Unlike cutaneous melanoma, it has shown only limited sensitivity to immune checkpoint blockade, and once metastases develop, the liver becomes the dominant site of disease in the vast majority of patients.
For this reason, liver-directed strategies have become especially important in uveal melanoma. Among them, percutaneous hepatic perfusion (PHP) has emerged as one of the most active regional approaches, allowing high-dose melphalan to be delivered directly to the liver while minimizing systemic exposure. This can lead to meaningful control of hepatic metastases, but the limitation is clear: PHP primarily treats liver disease and does not adequately address extrahepatic progression.
At the same time, ipilimumab plus nivolumab has shown only modest activity in uveal melanoma when used alone. Still, there has been increasing interest in whether regional liver therapy might enhance the effectiveness of immunotherapy, possibly by increasing tumor antigen release and improving immune activation.
The CHOPIN trial was designed to test exactly this concept: can combining PHP with ipilimumab and nivolumabimprove outcomes compared with PHP alone in patients with liver-dominant metastatic uveal melanoma?
Study Design
CHOPIN was an investigator-initiated, single-center, open-label, randomized phase 2 trial.
The study enrolled 76 patients with unresectable liver-only or liver-dominant metastatic uveal melanoma, good performance status, and no prior systemic therapy. Patients were randomly assigned in a 1:1 ratio to one of two treatment strategies:
- Percutaneous hepatic perfusion alone
- Percutaneous hepatic perfusion combined with ipilimumab and nivolumab
All patients were scheduled to receive two PHP procedures, performed in week 1 and week 7, using melphalan.
Patients in the combination arm also received:
- Ipilimumab 1 mg/kg
- Nivolumab 3 mg/kg
These were given every 3 weeks for four cycles, without maintenance therapy.
- The primary endpoint was 1-year progression-free survival.
- Secondary endpoints included overall survival, objective response rate, and safety.
- Median follow-up was 24.9 months.
Results
Progression-Free Survival
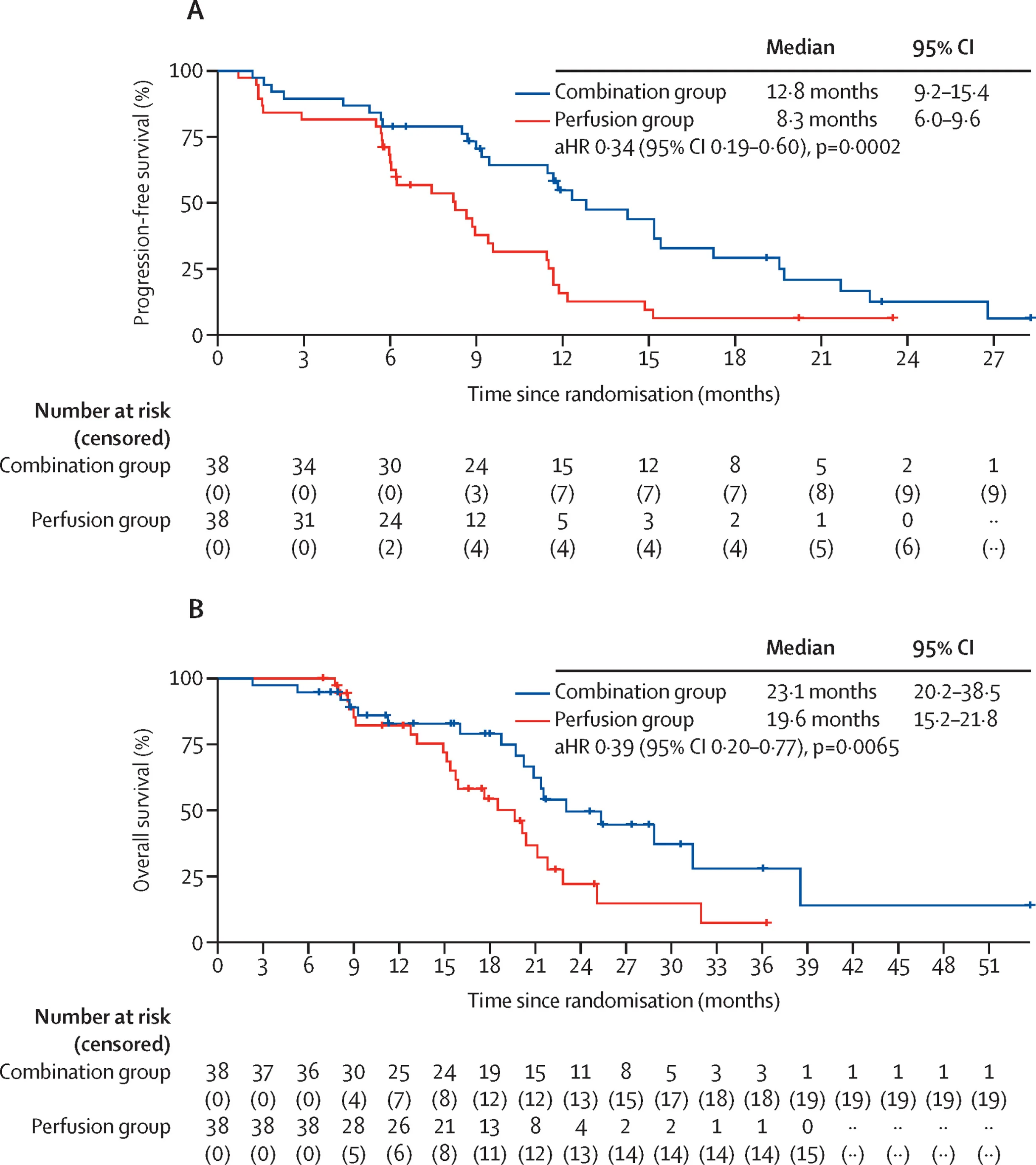
The primary endpoint clearly favored the combination strategy.
At 1 year, progression-free survival was 54.7% in the PHP + ipilimumab/nivolumab arm, compared with only 15.8%in the PHP-alone arm.
This translated into a 66% relative reduction in the risk of progression or death:
- HR 0.34
- 95% CI 0.19–0.60
- P = 0.0002Median progression-free survival was also longer with the combination:
- 12.8 months with PHP + immunotherapy
- 8.3 months with PHP alone
Interestingly, post-hoc analyses suggested even greater separation in hepatic progression-free survival, supporting the idea that the combination improved intrahepatic disease control in particular.
Overall Survival
Although the trial was not powered primarily for overall survival, the OS signal was also encouraging.
Median overall survival was:
- 23.1 months in the combination arm
- 19.6 months in the PHP-alone arm
The adjusted hazard ratio for death was:
- HR 0.39
- 95% CI 0.20–0.77
At 2 years, survival rates were:
- 49.6% with PHP + ipilimumab/nivolumab
- 22.1% with PHP alone
So while these OS results should be interpreted with appropriate caution, they strongly support the possibility that the addition of checkpoint inhibition is doing more than simply delaying radiographic progression.
Response Rate
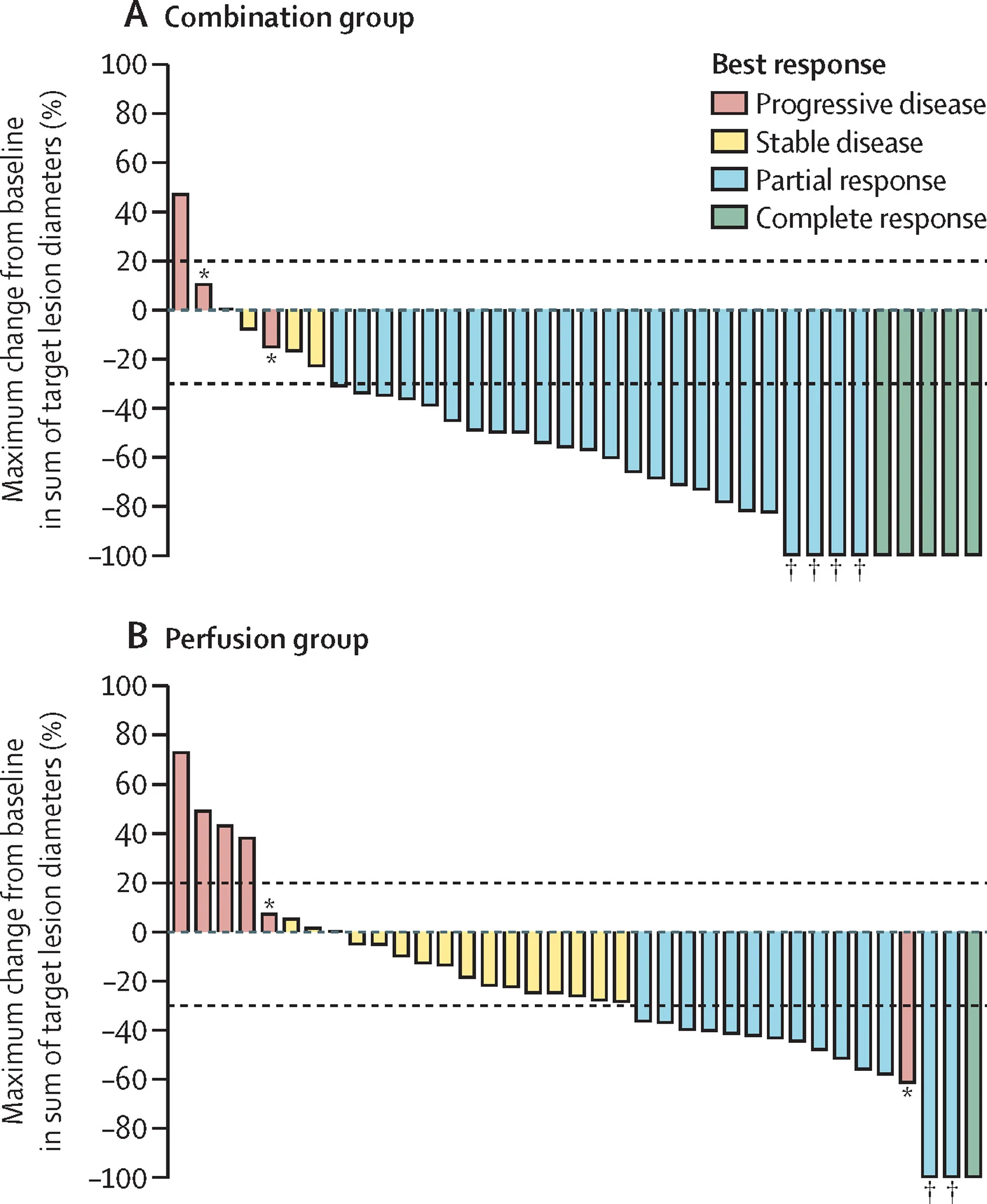
Tumor responses were also substantially higher with the combined approach.
The objective response rate was:
- 76.3% with PHP + immunotherapy
- 39.5% with PHP alone
In the combination arm, responses included:
- 5 complete responses
- 24 partial responses
In the perfusion-only arm, there were:
- 1 complete response
- 14 partial responses
This is a striking difference, and it reinforces the impression that regional therapy and immunotherapy may be acting synergistically.
Safety
As expected, the improved efficacy came with increased toxicity.
Grade 3–4 treatment-related adverse events occurred in:
- 82% of patients in the combination arm
- 41% of patients in the PHP-alone arm
The most common severe adverse events were:
- Thrombocytopenia
- Leukopenia
- Elevated γ-glutamyl transferase
- Anemia
Importantly, the combination arm experienced not only more immunotherapy-related toxicities, but also a higher rate of severe perfusion-related adverse events, suggesting that immune activation may amplify some of the hematologic and hepatic effects of PHP.
One treatment-related death occurred in the combination group due to triple M syndrome — the concurrent occurrence of myasthenia gravis, myositis, and myocarditis.
Despite this, most toxicities were described as self-limiting or manageable with supportive care, and no new unexpected safety signals were identified.
Why This Trial Matters
What makes CHOPIN important is not only that it improved progression-free survival, but that it supports a new therapeutic concept in metastatic uveal melanoma.
Historically, treatment options in this disease have been divided into:
- regional liver control, which is often effective but local,
- and systemic immunotherapy, which is conceptually attractive but usually weak in this disease.
CHOPIN suggests that these two strategies may be more powerful together than separately.
The biological logic is compelling: high-dose regional melphalan may increase tumor cell death, neoantigen release, and immune visibility, potentially making checkpoint inhibition more effective in a disease that is otherwise relatively immunologically silent.
Key Takeaway Messages
- CHOPIN is the first randomized trial to show a clear benefit from combining percutaneous hepatic perfusion with dual checkpoint blockade in metastatic uveal melanoma.
- The combination significantly improved 1-year progression-free survival compared with PHP alone.
- Objective response rates were substantially higher with the combined regimen.
- A favorable overall survival signal was also observed, although this requires cautious interpretation.
- Toxicity increased considerably, but was generally manageable in a specialized setting.
- These findings support a new treatment paradigm in which regional hepatic therapy and systemic immunotherapy are integrated rather than used separately.
Read All Article Here

You Can Also Read About KIMMTRAK (Tebentafusp) uses in Cancer: Dosages, Indications, Expectations and more