For six years, a single trial redefined how we treated HER2-positive breast cancer patients who didn’t achieve a pathologic complete response after neoadjuvant therapy. KATHERINE changed everything, and it did it so decisively that T-DM1 became untouchable.
Category 1. NCCN. Standard of care. Done.
Until now.
The results of DESTINY-Breast05 firmly placed T-DXd at the center of the post-neoadjuvant discussion in HER2-positive early breast cancer. Presented at the ESMO Presidential Symposium in October 2025 and later published in The New England Journal of Medicine in December 2025, the trial delivered one of the most important breast oncology readouts of the decade.
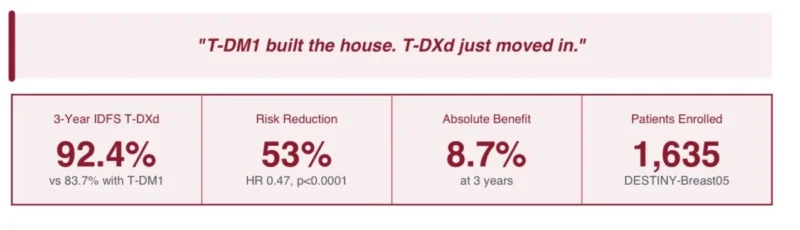
T-DXd reduced the risk of invasive disease recurrence or death by 53% versus T-DM1, achieved a nearly 9% absolute improvement in 3-year invasive disease-free survival (92.4% vs 83.7%), and showed an early but clinically notable signal for fewer brain metastasis events. With a hazard ratio of 0.47, the study crossed its prespecified interim boundary and established T-DXd as a major new standard-defining therapy for patients with high-risk residual HER2-positive disease after neoadjuvant treatment.
The Problem That Never Fully Went Away
To understand what DESTINY-Breast05 means, you have to start where the story actually begins not in 2025, but in 2018, when the primary results of KATHERINE were published.
KATHERINE was built on a sobering clinical reality: despite effective neoadjuvant HER2-directed therapy, approximately 45 to 66% of patients with HER2-positive early breast cancer still have residual invasive disease at surgery, depending on estrogen receptor status. These patients face substantially worse long-term outcomes than those who achieve pCR.
KATHERINE enrolled 1,486 patients with residual invasive disease after neoadjuvant taxane-based chemotherapy plus trastuzumab, randomizing them to adjuvant T-DM1 versus trastuzumab alone. The result was landmark: T-DM1 reduced the risk of invasive disease recurrence or death by 50% (HR 0.50; 95% CI 0.39–0.64; p<0.001), with 3-year IDFS of 88.3% versus 77.0% (N Engl J Med 2019;380:617–628).
The long-term follow-up, published in NEJM in January 2025 with a median follow-up of 8.4 years, confirmed what practitioners had hoped: the IDFS benefit was sustained (HR 0.54; 7-year IDFS 80.8% vs 67.1%), and T-DM1 now carried a significant overall survival advantage (HR 0.66; p=0.003), with OS of 89.1% vs 84.4% at 7 years (N Engl J Med 2025;392:249–257).
But KATHERINE also revealed something else, something hidden in its own success. The highest-risk patients: those with locally advanced (T4) disease or heavy nodal burden (N2–N3), still faced a roughly 25% recurrence rate at 3 years even with T-DM1. The drug worked remarkably well. And it still wasn’t enough.
That gap is precisely what DESTINY-Breast05 was designed to close.
What DESTINY-Breast05 Actually Did
The trial enrolled 1,635 patients, a higher-risk population than KATHERINE by design. It included twice as many patients with inoperable disease at baseline, nearly twice as many who were node-positive at surgery, and 80% of patients who had received dual
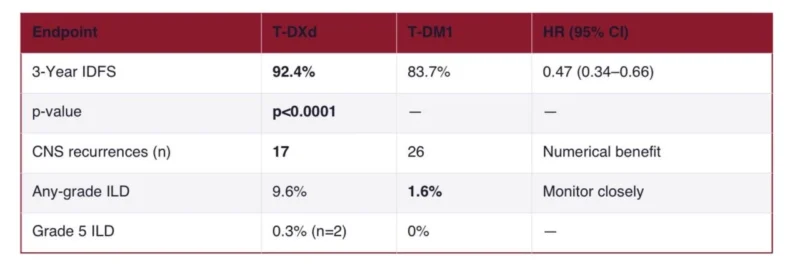
HER2-directed neoadjuvant therapy (trastuzumab + pertuzumab). These were not the easy cases. Patients were randomized 1:1 to T-DXd (5.4 mg/kg IV q3w) or T-DM1 (3.6 mg/kg IV q3w) for 14 cycles. The primary endpoint was invasive disease-free survival.At a median follow-up of approximately 30 months (data cutoff July 2, 2025), 3-year IDFS: 92.4% (T-DXd) vs 83.7% (T-DM1), HR 0.47 (95% CI 0.34–0.66; p<0.0001)
The hazard ratio of 0.47 crossed the pre-specified interim stopping boundary of 0.0183 with room to spare, a result the investigators described as easily beating the early reporting threshold.
The absolute IDFS difference of 8.7 percentage points, achieved in an already higher-risk population, translates to a number needed to treat of approximately 11, meaning for every 11 patients treated with T-DXd instead of T-DM1, one additional invasive disease event is prevented at 3 years. In a disease where recurrence often means incurable metastatic disease, that is not a statistical footnote.
The Details That Matter Clinically
Consistency across subgroups
The benefit of T-DXd over T-DM1 was observed regardless of hormone receptor status, HER2 IHC expression level (IHC 3+ or ISH+), prior neoadjuvant chemotherapy type (anthracycline-based or platinum-based), nodal status at surgery, region of accrual, and age. This is not a signal confined to a biological niche, it is broad, reproducible, and robust.
In patients with HER2 IHC 3+ disease (the dominant subgroup), 3-year IDFS was 91.8% vs 83.2% (HR 0.49). Among those who had received prior anthracyclines, the reduction in risk reached 55% (HR 0.45; 3-year IDFS 90.6% vs 80.3%). Even in the platinum-pretreated group, a 46% risk reduction was observed.
The CNS signal
Perhaps the most intriguing secondary observation: CNS recurrences occurred in 17 patients in the T-DXd arm versus 26 in the T-DM1 arm, nine fewer events. KATHERINE had notably failed to reduce CNS relapse risk with T-DM1 versus trastuzumab alone. Whether T-DXd can penetrate the blood-brain barrier sufficiently to prevent CNS seeding in early-stage disease remains an open question requiring longer follow-up, but the numerical trend is biologically plausible given T-DXd’s known activity in HER2+ brain metastases in the metastatic setting (DESTINY-Breast12).
Distant recurrence
The majority of IDFS events were distant recurrences, and T-DXd reduced these by approximately 50%, the same magnitude as the overall IDFS benefit. This matters because distant relapse in HER2+ breast cancer typically signals transition to a chronic, ultimately fatal disease state. Preventing distant recurrence is not managing cancer.
It is, in some patients, preventing death.
The Safety Conversation That Cannot Be Avoided
T-DXd’s efficacy data are extraordinary. Its toxicity profile demands honest discussion. Interstitial lung disease (ILD) remains the defining adverse event of concern. In DESTINY-Breast05, any-grade adjudicated drug-related ILD was observed in 9.6% of T-DXd-treated patients versus 1.6% in the T-DM1 arm. The majority of ILD events were Grade 1 (2.1%) or Grade 2 (6.7%). There were 2 Grade 5 (fatal) ILD events (0.3%) in the T-DXd arm.
Critically, radiotherapy timing, concurrent versus sequential with T-DXd, did not significantly affect ILD incidence or severity. This is practically important for a patient population where adjuvant radiation is common.The discussion at ESMO 2025 was framed well by Sara Tolaney: the benefits of T-DXd in high-risk patients “far outweigh the risks” provided appropriate monitoring and management protocols are followed. ILD in the T-DXd setting requires vigilance, early recognition, prompt dose interruption, and corticosteroid management, but it is manageable in the majority of patients.
The gastrointestinal and hematologic toxicity profile was otherwise expected and consistent with the known T-DXd label.
Who Should, and Should Not Receive T-DXd Post-Neoadjuvant Therapy
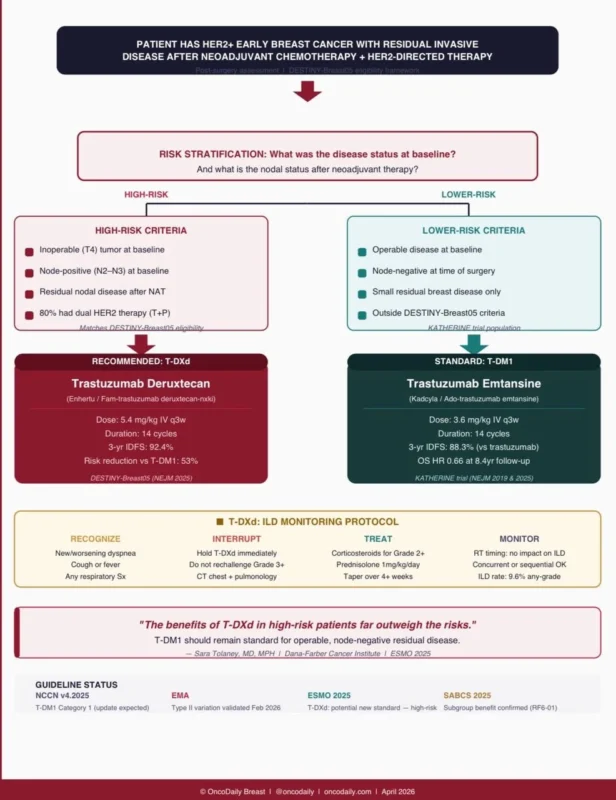
This is where clinical nuance matters most, and where DESTINY-Breast05 itself draws a line. Dr. Tolaney offered explicit guidance at ESMO: “T-DXd should be considered a standard therapy for patients who have residual node-positive disease or who have tumors that are inoperable at baseline.”
Conversely, she cautioned: “I would, however, caution against extending the use of T-DXd into those patients who fell outside the eligibility for this trial, as we have seen that patients who have lower-risk residual disease do very well with T-DM1.
T-DM1 should remain a standard of care for those patients who have operable disease at baseline and have tumors that are node-negative at the time of surgery.”
This is not a blanket replacement. DESTINY-Breast05 enriched for high-risk biology, and that is where the evidence, and arguably the benefit-risk calculus, sits most comfortably. For lower-risk residual disease (operable at baseline, node-negative after neoadjuvant therapy),T-DM1’s long-term IDFS and OS data from KATHERINE, reported at a median follow-up of 8.4 years.
The clinical question in 2026 is not “T-DXd or T-DM1?” in every case. It is: does this patient’s residual disease burden place them in the population where DESTINY-Breast05’s benefit applies?
The Regulatory and Guideline Landscape
At the time of ESMO 2025 presentation, NCCN guidelines (version 4.2025) still listed T-DM1 as the category 1 recommendation for post-neoadjuvant residual disease. Following simultaneous NEJM publication in December 2025 and updated data presented at SABCS 2025, the EMA validated the Type II variation application for T-DXd in this post-neoadjuvant setting in February 2026, a regulatory signal that formal approval and guideline integration is imminent.
The oncology community should anticipate updated NCCN and ESMO guideline language incorporating T-DXd as a preferred option for high-risk residual disease in HER2+ early breast cancer in the near term.
ADCs Are Moving Upstream Across HER2-Positive Breast Cancer
DESTINY-Breast05 is also part of a larger pattern. The story here is not only about one post-neoadjuvant trial. It is about the systematic movement of antibody-drug conjugates into earlier and more curative treatment settings.
In metastatic HER2-positive breast cancer, DESTINY-Breast03 already established trastuzumab deruxtecan as superior to T-DM1, with a long-term analysis showing median overall survival of 52.6 months versus 42.7 months and a hazard ratio for death of 0.73. That trial made it clear that T-DXd was not simply another HER2-directed option. It was a more effective ADC in a disease where HER2 biology remained central.
The field then moved even earlier. In first-line metastatic HER2-positive disease, DESTINY-Breast09 showed that trastuzumab deruxtecan plus pertuzumab led to a significantly lower risk of progression or death than standard THP, extending the ADC’s reach further forward in the disease course.
And in early-stage disease, DESTINY-Breast11 added neoadjuvant evidence. In that phase III study, neoadjuvant T-DXd followed by THP achieved a pathologic complete response rate of 67.3%, compared with 56.3% for standard dose-dense AC followed by THP. That does not yet settle the full neoadjuvant standard-of-care debate, but it makes the trajectory impossible to miss: the ADC is moving upstream, and it is doing so on the strength of randomized data.
What This Means for Daily Practice
For the practicing oncologist, DESTINY-Breast05 changes the post-neoadjuvant discussion in a very specific way. It does not mean that every patient with any residual HER2-positive disease should automatically receive T-DXd. It means that for patients whose disease matches the high-risk residual-disease definition used in the trial, the evidence now supports a more potent post-neoadjuvant strategy than the historical standard. That is the clinically important shift.
This also means T-DM1 is not obsolete. It remains supported by long-term survival data from KATHERINE and still has a central place in the HER2-positive early breast cancer story. But its role is becoming more precisely defined as T-DXd enters the post-neoadjuvant space for higher-risk residual disease. That is how standards evolve in modern oncology: not by erasing the previous standard, but by clarifying where the newer therapy delivers a stronger benefit.
The Bigger Message
The deeper significance of DESTINY-Breast05 is strategic. Oncology is no longer satisfied with proving that a drug works late. The ambition now is to bring the most effective biologically informed therapies into the part of the treatment pathway where cure is still realistic. In HER2-positive breast cancer, that strategy is now visible across the metastatic, post-neoadjuvant, and neoadjuvant settings. DESTINY-Breast05 is one of the clearest examples of that shift.
And that is why the right 2026 question is not a generic drug-comparison question. It is a risk-definition question. Which patients truly resemble the trial population? Which patients carry enough residual disease burden that the magnitude of T-DXd benefit justifies its use in the curative-intent setting? That is the question clinicians now have to answer case by case.

You Can Read All Destiny Breast Trials on OncoDaily


