Triple-negative breast cancer remains one of the most difficult subtypes to treat, not because progress has been absent, but because progress has been uneven. Immunotherapy and antibody-drug conjugates have changed outcomes for selected patients, yet a true biomarker-driven strategy for metastatic TNBC is still far less developed than in other breast cancer subtypes. That is why the TNBC cohort of the SUMMIT basket trial deserves attention. It focused on a very small but clinically important subgroup: patients with HER2-mutant metastatic triple-negative breast cancer, a population estimated to represent only 1% to 3% of TNBC cases, but one that may carry a real therapeutic vulnerability.
In the final analysis of this cohort, neratinib, given alone or with trastuzumab, showed meaningful antitumor activity in heavily pretreated HER2-mutant metastatic TNBC. The responses were not universal, and the study was not randomized, but the signal was real enough to support the inclusion of neratinib-based combinations in the NCCN Guidelines for patients with hormone receptor-positive or hormone receptor-negative metastatic breast cancer harboring activating HER2 mutations (Jhaveri et al.).
Read About TNBC on OncoDaily
Why HER2 Mutations Matter in TNBC
HER2-mutant disease is not the same as HER2-positive disease by immunohistochemistry or amplification testing. These are tumors that remain HER2-negative by conventional pathology, yet carry activating ERBB2 mutations that may drive tumor growth. In metastatic breast cancer, HER2 mutations are more commonly discussed in hormone receptor-positive disease, but they are also found in TNBC, where they may offer one of the few identifiable molecular targets in an otherwise treatment-resistant setting (Bose et al.; Jiang et al.).
That distinction is critical. These patients would not usually be identified as candidates for standard HER2-directed therapy based on routine HER2 overexpression or amplification testing alone. The SUMMIT data therefore speak not only to treatment efficacy, but also to the growing importance of genomic testing in metastatic TNBC.
How The SUMMIT TNBC Cohort Was Designed
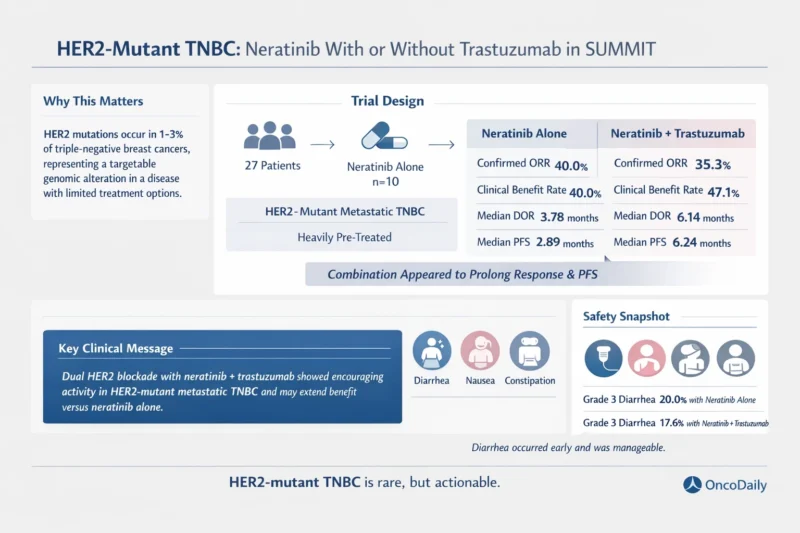
The SUMMIT study was an open-label, multicohort, phase II basket trial evaluating neratinib-based treatment in patients with HER2-mutant solid tumors. Within the TNBC cohort, patients were enrolled sequentially into two groups. The first group received neratinib monotherapy at 240 mg orally daily. The second group received neratinib plus trastuzumab, with neratinib at the same dose and trastuzumab given intravenously every three weeks.
A total of 27 patients with activating HER2-mutant metastatic TNBC were enrolled between July 2014 and September 2021. Ten patients received neratinib alone, and seventeen received the combination of neratinib plus trastuzumab. These were not untreated patients. The cohort was heavily pretreated, with a median of 2.5 prior metastatic regimens in the monotherapy group and 2.0 prior metastatic regimens in the combination group. Most patients had visceral disease, and all had documented hormone receptor-negative, HER2-negative disease at enrollment by standard clinical testing (Jhaveri et al.).
What Mutations Were Seen?
The genomic profile of this cohort was diverse, but several patterns stood out. Across the 27 patients, investigators reported 12 unique HER2 mutations. Most patients had a single missense mutation in the kinase domain, including variants such as V777L, L755S, and D769Y. A smaller subset had HER2 exon 20 insertions, while a few carried extracellular domain mutations such as S310F or S310Y. Two patients had dual HER2 mutations.
Importantly, no baseline tumors showed HER2 copy number amplification, reinforcing that this was a truly HER2-mutant, non-amplified population. Co-alterations were common, especially TP53, PIK3CA, and ARID1A, but none clearly eliminated the possibility of benefit. That matters, because one of the recurrent questions in HER2-mutant disease is whether co-mutations blunt dependence on HER2 signaling. In this dataset, responses still occurred despite frequent genomic complexity (Jhaveri et al.).
The Efficacy Signal
The trial was small, so every efficacy conclusion has to be interpreted carefully. Still, the results are clinically meaningful.
The primary endpoint, objective response rate at first post-baseline assessment, was 50.0% with neratinib alone and 29.4% with neratinib plus trastuzumab. That number, however, does not tell the whole story. When looking at confirmed ORR at any assessment, the results were 40.0% for neratinib monotherapy and 35.3% for the combination. Clinical benefit rate was 40.0% with monotherapy and 47.1% with the combination.
The more informative difference came from the durability metrics. Median duration of response was 3.78 months with neratinib alone and 6.14 months with neratinib plus trastuzumab. Median progression-free survival was 2.89 months with monotherapy and 6.24 months with the combination (Jhaveri et al.).
This is the key clinical message. Although the confirmed response rate was not dramatically higher with trastuzumab added, the combination appeared to deepen and prolong benefit. The authors are careful not to overstate this, because the cohorts were sequential and non-randomized. Still, in practice, a signal for longer disease control in HER2-mutant TNBC is not easy to dismiss.
Which Patients Seemed To Benefit Most?
One of the more interesting observations was that responses appeared particularly notable in tumors with HER2 exon 20 insertions, although responses were also seen in patients with kinase domain missense mutations. Among the 12 patients who achieved a clinical response, six had exon 20 insertions and five had single kinase domain mutations. Responses were seen across ductal and lobular histologies, which is also relevant given the known enrichment of HER2 mutations in some lobular subtypes.
The paper also notes that in the combination cohort, responses appeared numerically higher in patients who had received endocrine therapy earlier in the course of their disease, though that observation is exploratory and based on very small numbers. Prior HER2-directed therapy was rare and limited to the neoadjuvant or adjuvant setting, so these data mainly reflect activity in tumors that had not already been heavily exposed to HER2 blockade (Jhaveri et al.).
What The ctDNA Analysis Adds
The most valuable part of this paper may be its translational analysis. Serial ctDNA sequencing showed a pattern that fits clinical intuition but is still important to document: HER2 mutation variant allele frequencies fell during treatment in patients who responded or had stable disease, then rose again at progression.
That dynamic makes HER2-mutant TNBC look like a disease that can be genuinely HER2-driven, at least for a period of time. In five patients with a clinical response and serial plasma samples, HER2 VAFs decreased on treatment and re-emerged at progression. Similar declines were seen in some patients with stable disease. In contrast, patients with progressive disease had little or no meaningful reduction in HER2 VAF (Jhaveri et al.).
The resistance story was also revealing. Investigators identified emergent on-pathway and off-pathway alterations, including ERBB3, PIK3CA, MTOR, KRAS, and TP53. One patient treated with neratinib developed the known HER2 T798I gatekeeper mutation, which had been described before. In the combination cohort, however, no additional HER2 alterations were detected upon progression in this small dataset, suggesting that dual HER2 targeting may shift the biology of acquired resistance even if it does not fully prevent it (Jhaveri et al.).

Safety And Tolerability
No neratinib study can be discussed without addressing diarrhea, and this cohort was no exception. The most common treatment-emergent adverse events were diarrhea, nausea, and constipation. Any-grade diarrhea occurred in 80.0% of patients treated with neratinib alone and in 100.0% of those receiving neratinib plus trastuzumab. Grade 3 diarrhea occurred in 20.0% and 17.6%, respectively.
The reassuring point is that severe diarrhea occurred early, with a median onset of 7 days, and was generally short-lived, with a cumulative median duration of 2 days. Dose interruptions and reductions were used when needed, but none of the patients discontinued treatment because of diarrhea. One patient discontinued the combination because of a seizure related to brain metastases, not to study therapy (Jhaveri et al.).
So the toxicity profile was real, but familiar and manageable. That matters because in a biomarker-defined subgroup as small as HER2-mutant TNBC, a clinically useful regimen has to be feasible outside of an idealized trial setting.
What This Means For Practice
The SUMMIT TNBC cohort is not a practice-changing phase III study. It is a small, non-randomized basket-trial analysis. But that does not make it unimportant. In metastatic TNBC, where therapeutic options are often broad rather than biologically tailored, even modestly sized data can matter when they identify a real target and a real signal of activity.
The key point is not that neratinib plus trastuzumab solves HER2-mutant TNBC. It does not. The key point is that it offers a targeted option for a genomically defined subgroup that previously had little guidance beyond standard chemotherapy and newer broadly active agents. Based on these data and earlier SUMMIT results in hormone receptor-positive disease, neratinib-based combinations are now reflected in NCCN guidance for metastatic breast cancer with activating HER2 mutations (Jhaveri et al.).
That also has implications for testing. If HER2 mutations can define a treatable subset in TNBC, then genomic profiling should not be viewed as optional academic detail. It becomes part of therapeutic decision-making.
The Larger Context
The paper also places neratinib-based treatment in a rapidly evolving HER2-low and HER2-altered landscape. The authors mention the success of trastuzumab deruxtecan in HER2-low metastatic breast cancer and discuss ongoing questions about whether HER2-mutant, HER2-low, hormone receptor-negative disease may one day benefit from sequencing or combination strategies involving both kinase inhibition and antibody-drug conjugates. That question remains open, but the field is clearly moving toward more precise targeting of HER2 biology beyond amplification alone (Modi et al.; Arshad et al.; Li et al.).
Final Perspective
For a rare subgroup, these are meaningful data. In HER2-mutant metastatic triple-negative breast cancer, neratinib alone produced confirmed responses, and the addition of trastuzumab appeared to extend disease control, with a median PFS of 6.24 months and a median duration of response of 6.14 months in the combination cohort. The ctDNA analysis strengthens the biological argument that these tumors can be HER2-driven and shows plausible mechanisms of acquired resistance. The toxicity profile was consistent with prior neratinib experience and largely manageable with prophylaxis and supportive care (Jhaveri et al.).
In TNBC, where treatment often feels empiric even when effective, that kind of biomarker-defined signal matters. HER2-mutant disease remains uncommon, but it is no longer invisible. And once a subgroup becomes visible, the standard of care usually begins to change.
Read full article here


