HER2-low breast cancer has changed one of the most familiar classifications in breast oncology. For many years, tumors that were not HER2-positive were generally managed as HER2-negative disease, with treatment decisions driven by hormone receptor status, prior therapy, disease tempo, and chemotherapy sensitivity. The development of trastuzumab deruxtecan has changed this framework by making low HER2 expression clinically actionable in metastatic breast cancer (Modi et al., 2022).
HER2-low is not a separate biological subtype in the same way as HER2-positive, triple-negative, or hormone receptor-positive breast cancer. It is better understood as a treatment-relevant biomarker that identifies patients whose tumors may be eligible for HER2-directed antibody-drug conjugate therapy. This distinction is important because an ER-positive/HER2-low tumor, a triple-negative/HER2-low tumor, and a HER2-ultralow tumor still require different treatment strategies based on the broader clinical context (Wolff et al., 2023).
HER2-Low And HER2-Ultralow: What The Terms Mean
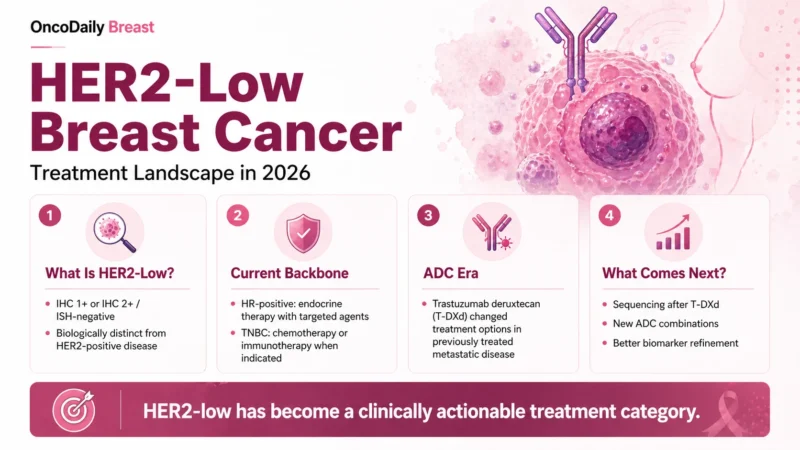
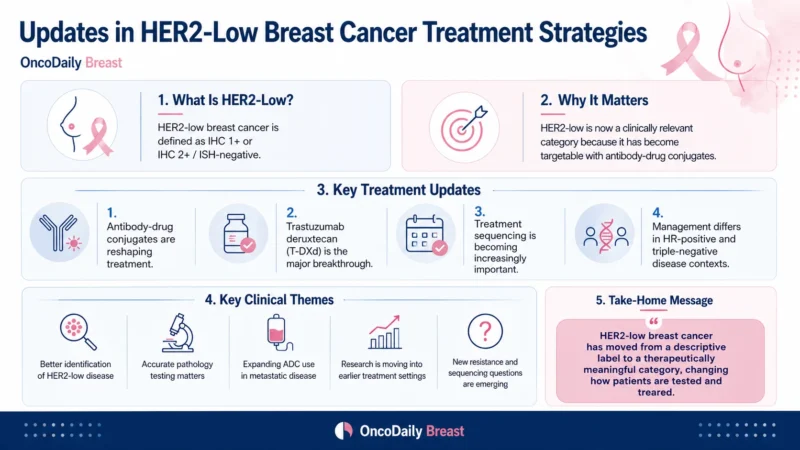
HER2-low breast cancer is generally defined as HER2 immunohistochemistry 1+ or HER2 IHC 2+ with negative in situ hybridization. HER2-ultralow refers to IHC 0 with membrane staining. This ultralow category became clinically relevant after the FDA approved fam-trastuzumab deruxtecan-nxki for unresectable or metastatic hormone receptor-positive HER2-low or HER2-ultralow breast cancer that progressed on one or more endocrine therapies in the metastatic setting (U.S. Food and Drug Administration [FDA], 2025).
The practical implication is simple but important: clinicians need the exact HER2 IHC score, not only the broad label “HER2-negative.” A report that only says HER2-negative may not provide enough information for modern metastatic treatment planning. The College of American Pathologists notes that metastatic breast cancer patients with HER2 IHC 1+ or 2+/ISH-negative results may be considered for trastuzumab deruxtecan if they meet other clinical eligibility criteria, and it recommends reporting the semi-quantitative HER2 IHC result to support treatment decisions (Wolff et al., 2023).
Testing Has Become Part Of Treatment Strategy
Accurate HER2 testing is now central to HER2-low treatment strategy. The challenge is that the difference between IHC 0 and IHC 1+ can be subtle, and the HER2-ultralow category makes even faint membrane staining clinically relevant in selected hormone receptor-positive metastatic cases. CAP emphasizes that HER2-low is not an official interpretive category in ASCO-CAP guidelines, but that the semi-quantitative score should be included in reports because it can identify patients who meet trial-based or label-based treatment criteria (Wolff et al., 2023).
In daily practice, this means that older pathology reports may need to be revisited when metastatic treatment decisions are being made. If the original report does not specify whether the tumor was IHC 0, 1+, or 2+/ISH-negative, pathology review or repeat HER2 testing may be useful. Testing metastatic tissue can also be informative when feasible, because HER2 expression may differ between primary and metastatic sites or change over time (Wolff et al., 2023).
DESTINY-Breast04 Established HER2-Low As Actionable
The major turning point for HER2-low disease was DESTINY-Breast04, a randomized phase 3 trial evaluating trastuzumab deruxtecan in patients with previously treated HER2-low advanced breast cancer. The study defined HER2-low disease as IHC 1+ or IHC 2+/ISH-negative and compared trastuzumab deruxtecan with physician’s choice chemotherapy (Modi et al., 2022).
DESTINY-Breast04 showed that trastuzumab deruxtecan improved both progression-free survival and overall survival compared with standard chemotherapy options in HER2-low metastatic breast cancer. This result transformed HER2-low from a descriptive pathology category into a treatment-relevant biomarker and established trastuzumab deruxtecan as an important option after prior chemotherapy in metastatic disease (Modi et al., 2022).
For clinicians, DESTINY-Breast04 changed the meaning of HER2-negative reporting. A tumor that is HER2 IHC 1+ or 2+/ISH-negative is still HER2-negative by traditional criteria, but it may also be HER2-low and therefore potentially eligible for trastuzumab deruxtecan in the appropriate metastatic setting (Modi et al., 2022; Wolff et al., 2023).
DESTINY-Breast06 Moved T-DXd Earlier In HR-Positive Disease
DESTINY-Breast06 expanded the HER2-low strategy into an earlier metastatic treatment setting for hormone receptor-positive disease. The FDA approval was based on DESTINY-Breast06, a randomized, multicenter, open-label trial that enrolled 866 patients with advanced or metastatic HR-positive breast cancer with HER2-low or HER2-ultralow expression. Patients had not received prior chemotherapy for advanced or metastatic disease (FDA, 2025).
Patients were randomized to fam-trastuzumab deruxtecan-nxki 5.4 mg/kg every three weeks or physician’s choice single-agent chemotherapy. In the HER2-low population, median progression-free survival was 13.2 months with trastuzumab deruxtecan and 8.1 months with chemotherapy, with a hazard ratio of 0.62. In the overall HER2-low and HER2-ultralow population, median PFS was also 13.2 versus 8.1 months, with a hazard ratio of 0.64. Overall survival data were immature at the time of the final PFS analysis (FDA, 2025).
This update is clinically important because trastuzumab deruxtecan is no longer limited to the post-chemotherapy HER2-low metastatic setting in HR-positive disease. It can now be considered after progression on one or more endocrine therapies in metastatic HR-positive HER2-low or HER2-ultralow breast cancer, before prior chemotherapy for advanced disease, when the patient meets eligibility criteria (FDA, 2025; Bardia et al., 2024).
Sequencing In HR-Positive HER2-Low Disease Is More Nuanced
In hormone receptor-positive metastatic breast cancer, HER2-low status does not replace endocrine-based treatment principles. Endocrine therapy combinations, CDK4/6 inhibitors, and biomarker-driven targeted therapies remain important when the disease is endocrine-sensitive and the patient does not require immediate cytoreduction.
The updated role of trastuzumab deruxtecan enters when endocrine-based treatment is no longer sufficient or appropriate. DESTINY-Breast06 supports trastuzumab deruxtecan after one or more endocrine therapies in metastatic HR-positive HER2-low or HER2-ultralow disease, while DESTINY-Breast04 supports its use in previously treated HER2-low metastatic disease after chemotherapy exposure (FDA, 2025; Modi et al., 2022).
The decision between another endocrine-targeted strategy and trastuzumab deruxtecan depends on endocrine sensitivity, prior CDK4/6 inhibitor exposure, tumor burden, symptoms, pace of progression, actionable molecular alterations, organ function, pulmonary risk, and patient goals. In slower endocrine-sensitive disease, another endocrine-based approach may remain reasonable. In more symptomatic or endocrine-resistant disease, trastuzumab deruxtecan may become more compelling if HER2-low or HER2-ultralow status is confirmed.
HER2-Low Triple-Negative Breast Cancer Requires A Different Strategy
HER2-low triple-negative breast cancer is not managed the same way as HR-positive HER2-low disease. In triple-negative breast cancer, first-line treatment decisions remain guided by PD-L1 status, germline BRCA status, prior therapy, disease burden, symptoms, and patient fitness. HER2-low status becomes particularly relevant when considering antibody-drug conjugate sequencing after standard earlier approaches.
DESTINY-Breast04 included patients with hormone receptor-negative HER2-low metastatic breast cancer, although this was a smaller cohort than the HR-positive population. The trial supported the broader concept that HER2-low expression can be therapeutically relevant beyond HR-positive disease, but treatment decisions in triple-negative disease still require careful sequencing with other available therapies (Modi et al., 2022).
Safety Remains Central To Treatment Selection
Trastuzumab deruxtecan is an active therapy, but safety monitoring is essential. Interstitial lung disease and pneumonitis remain among the most important risks. In DESTINY-Breast06, the FDA reported that common adverse reactions included hematologic laboratory abnormalities, nausea, fatigue, alopecia, liver enzyme elevations, diarrhea, vomiting, constipation, decreased appetite, COVID-19 infection, and musculoskeletal pain (FDA, 2025).
Pulmonary toxicity requires particular attention because early recognition and treatment interruption can be clinically important. Patients considered for trastuzumab deruxtecan need baseline respiratory assessment, education about symptoms such as new cough or dyspnea, and prompt evaluation of suspicious radiographic or clinical changes. Treatment selection should consider prior interstitial lung disease, prior pneumonitis, thoracic radiotherapy history, baseline pulmonary function, and the ability to monitor closely.
What Remains Unresolved
Several questions remain open. The first is pathology reproducibility. The distinction between IHC 0, HER2-ultralow, and IHC 1+ can be difficult, and CAP notes that HER2 IHC assays were originally designed to identify HER2 protein overexpression rather than to precisely distinguish very low levels of expression (Wolff et al., 2023).
The second unresolved issue is ADC sequencing. As more patients become eligible for antibody-drug conjugates, clinicians will need better evidence on how to sequence HER2-directed ADCs and TROP2-directed ADCs. Cross-trial comparisons should be avoided, and real-world sequencing must account for prior response, residual toxicities, marrow reserve, neuropathy, pulmonary risk, and patient preference.
The third question is whether HER2-low is stable over time. HER2 expression may vary across specimens, metastatic sites, and lines of treatment. When tissue is available and the treatment decision depends on HER2-low or HER2-ultralow status, repeat testing can be clinically meaningful, especially if the only available HER2 result is old or incomplete.
Clinical Takeaway
HER2-low breast cancer treatment has moved from a pathology observation to a major therapeutic strategy in metastatic disease. DESTINY-Breast04 established trastuzumab deruxtecan as an important option for previously treated HER2-low metastatic breast cancer, while DESTINY-Breast06 supported its earlier use in HR-positive HER2-low or HER2-ultralow metastatic breast cancer after endocrine therapy progression (Modi et al., 2022; FDA, 2025).
For clinical practice, the message is clear: do not rely only on the term “HER2-negative.” The exact HER2 IHC score and ISH result now matter. HER2-low and HER2-ultralow status should be interpreted within the full treatment sequence, not as a standalone subtype. The next stage of progress will depend on better testing standardization, safer ADC sequencing, and prospective data clarifying how HER2 expression changes over time and after therapy.
You Can Watch More on OncoDaily Youtube TV