A new Cancer Discovery study offers a clearer explanation for why some HER2-positive breast cancers do not respond durably to HER2-targeted antibody-drug conjugates, even when HER2 remains a defining feature of the disease. Using newly developed preclinical models, the authors show that HER2-low subclones can survive HER2-targeted ADC therapy, drive recurrence, and cooperate with HER2-high cells during treatment evolution. The same study also identifies ABCC1 and USP9X as potential therapeutic targets that may help sensitize resistant disease to trastuzumab deruxtecan, or T-DXd (Goyette et al.).
This matters because HER2 heterogeneity is not a rare laboratory curiosity. The authors note that HER2 heterogeneous breast cancer, defined by ASCO/CAP criteria as tumors with ERBB2 amplification in more than 5% but less than 50% of cancer cells or with a HER2-negative area by FISH, may represent up to 40% of HER2-positive breast cancer cases. Prior clinical work has already linked this phenotype to worse outcomes and reduced pathological complete response after neoadjuvant HER2-directed treatment (Metzger Filho et al., 2021; Li et al., 2024). In the current paper, the investigators build on that clinical observation by asking a more practical question: what exactly allows these tumors to persist under treatment pressure?
Why HER2 Heterogeneity Remains a Clinical Challenge
HER2-targeted therapy changed the natural history of HER2-positive breast cancer, from trastuzumab and pertuzumab to small-molecule kinase inhibitors and more recently ADCs such as T-DM1 and T-DXd. Yet benefit is still uneven. The central argument of this study is that a tumor labeled HER2-positive may still contain biologically distinct cancer cell populations with very different vulnerabilities. In other words, a single biomarker label may hide important internal diversity.
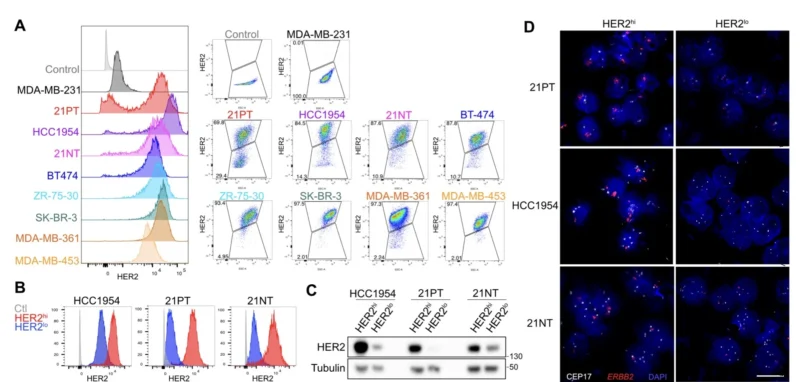
To study that problem, Goyette et al. generated paired HER2-high and HER2-low populations from human HER2-positive breast cancer cell lines derived from the same tumor background. These paired populations preserved stable differences in HER2 expression, and FISH confirmed that ERBB2 amplification was present in HER2-high cells but absent in HER2-low cells, closely mirroring the clinical definition of heterogeneous disease. At the molecular level, the main consistent difference between the paired populations centered on the ERBB2 amplicon, while HER2-low cells also appeared less HER2-like and more basal-like by transcriptomic profiling. Reanalysis of patient tumor data supported the same pattern, showing that HER2-low cells carried more basal and mesenchymal features than HER2-high counterparts (Perou et al., 2000; Janiszewska et al., 2021).
That observation is clinically meaningful. Basal-like or more plastic subpopulations may be less dependent on HER2 amplification itself, even if they remain part of a HER2-positive tumor. This creates the conditions for selective survival when a HER2-directed drug removes the most dependent clones first.
HER2-Low Cells Were Less Sensitive to ADCs but Not to HER2 Kinase Inhibitors
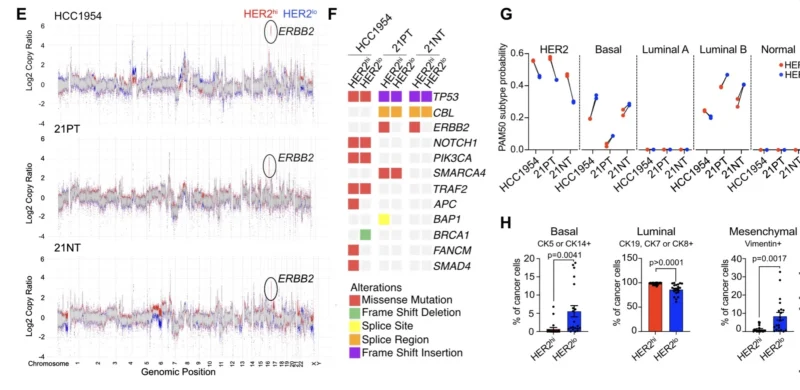
One of the most important findings in the paper is that HER2-low cells were consistently less sensitive to HER2-targeted ADCs, including T-DXd and T-DM1, than the matched HER2-high populations. By contrast, this resistance pattern did not hold for HER2 tyrosine kinase inhibitors such as neratinib and tucatinib. In fact, HER2-low cells remained sensitive to HER2 kinase inhibition, suggesting they still relied on HER-family signaling despite lacking ERBB2 amplification (Goyette et al.).
This distinction is one of the most useful translational messages from the study. It suggests that resistance in HER2 heterogeneous tumors is not simply a matter of generalized drug insensitivity. Rather, it is partly drug-class specific. ADCs depend on sufficient target expression, internalization, linker processing, and payload susceptibility. HER2 kinase inhibitors work through a different mechanism and may still suppress signaling in cells that express less HER2 on the surface.
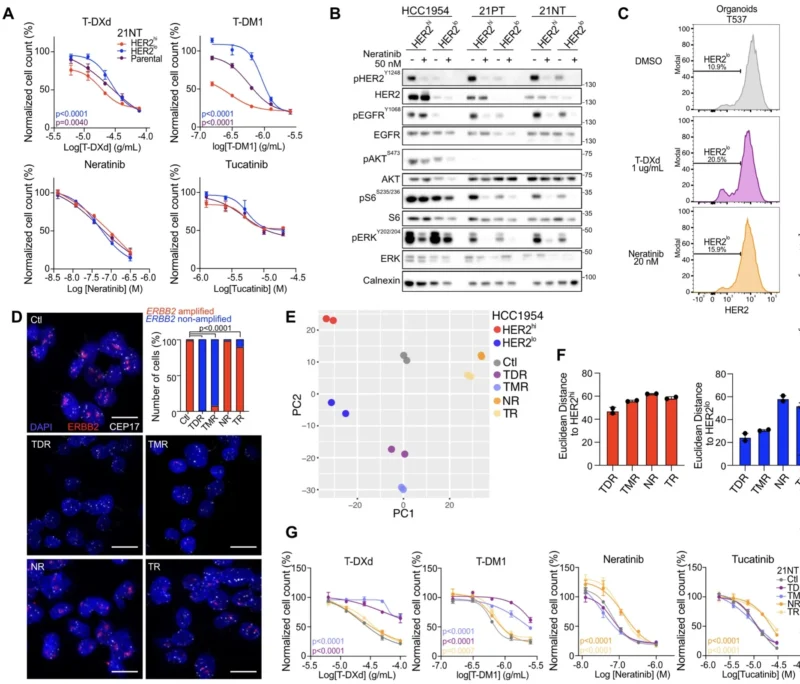
The authors strengthened this point in mixed-population experiments. When HER2-high and HER2-low cells were grown together and exposed to HER2-targeted agents over time, ADC treatment led to outgrowth of HER2-low cells, whereas TKI treatment did not produce the same selective dominance. Patient-derived organoids and organoids from a patient-derived xenograft showed the same directional pattern, supporting the relevance of the finding beyond standard cell lines.
Subclonal Cooperation Helps Explain Recurrence
The study goes further than showing differential drug sensitivity. It also addresses how subclones interact with one another. HER2-low cells were slower growing and were a minority population, yet they persisted. That raised the possibility that they were not merely passive survivors, but participants in a cooperative tumor ecosystem.
Indeed, when HER2-high and HER2-low cells were placed in co-culture, both populations grew better than they did alone. Transcriptomic and protein analyses suggested that co-culture promoted adaptive phenotypes, including changes linked to epithelial-to-mesenchymal transition and, in some settings, immune-related signaling and angiogenesis-associated factors. These data support the idea that HER2 heterogeneity is not just a diagnostic description, but a biologically active state in which subclones can alter one another’s phenotype and treatment response.
This concept became even more compelling in vivo. HER2-low cells alone were unable to initiate tumors in the mouse xenograft setting, but when mixed with HER2-high cells they were maintained within heterogeneous tumors. Short-term treatment with either neratinib or T-DXd reduced tumor size, but T-DXd was much more effective in homogeneous HER2-high tumors than in heterogeneous tumors. After T-DXd exposure, the residual disease in heterogeneous tumors was enriched for HER2-low cells. Later, when tumors recurred, they were again predominantly composed of HER2-high cells, suggesting that the HER2-low compartment may help sustain disease fitness during treatment and enable regrowth afterward.
That may be the most clinically provocative implication of the paper. It suggests that recurrence after T-DXd in heterogeneous tumors may not always arise because all HER2-high cells disappeared and new biology emerged. Instead, resistant HER2-low cells may serve as a reservoir that supports later re-expansion of HER2-high disease.
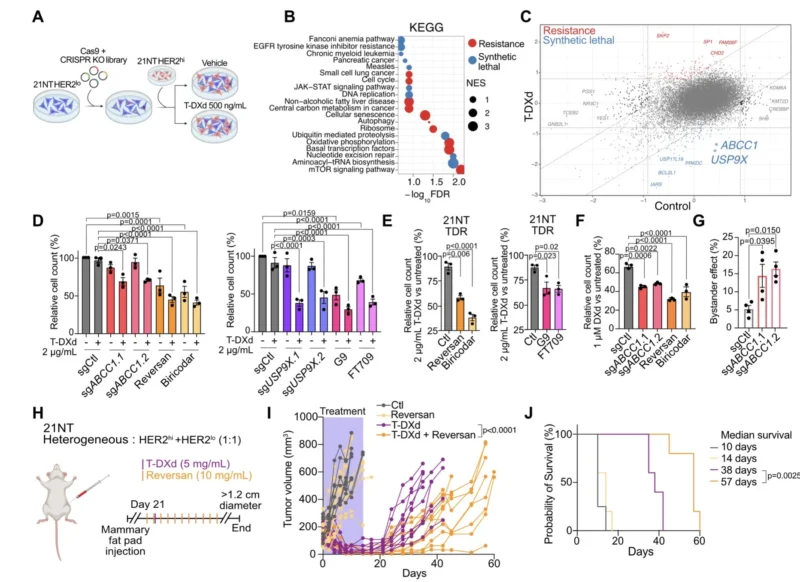
ABCC1 and USP9X Emerged as Strategies to Improve T-DXd Sensitivity
After establishing that HER2-low cells were a major source of ADC resistance, the authors performed a whole-genome CRISPR-Cas9 knockout screen in HER2-low cells grown in co-culture under T-DXd pressure. Among the synthetic lethal hits, two candidates stood out because pharmacologic tools already exist: ABCC1 and USP9X. Genetic or pharmacologic inhibition of either target increased sensitivity to T-DXd in HER2-low cells. The same approach also enhanced growth inhibition in T-DXd-resistant derivatives.
ABCC1 is a well-known multidrug resistance transporter. In this study, its inhibition appeared to improve T-DXd activity by increasing the effectiveness of the released payload, deruxtecan. Notably, blocking ABCC1 also increased the bystander effect of T-DXd in co-culture, an especially interesting point in a heterogeneous tumor where neighboring cells may express different levels of HER2. In mouse models, combining an ABCC1 inhibitor with T-DXd led to stronger tumor suppression and longer recurrence-free survival than T-DXd alone.
USP9X may be even more interesting mechanistically. The study shows that USP9X interacts with HER2 and helps regulate HER2 protein stability. Inhibiting USP9X increased HER2 ubiquitination and promoted more efficient trafficking of HER2 to lysosomes after T-DXd treatment. That matters because lysosomal delivery is essential for ADC processing and payload release. As a result, USP9X inhibition made HER2-low cells more sensitive not only to T-DXd but also to T-DM1. In vivo, combining a USP9X inhibitor with T-DXd improved tumor control and significantly prolonged recurrence-free survival. The authors also found that USP9X expression was higher in post-treatment tumors from patients who did not achieve pCR in the NCT02326974 clinical trial cohort, which adds clinical plausibility to the target (Metzger Filho et al., 2021; Li et al., 2024).
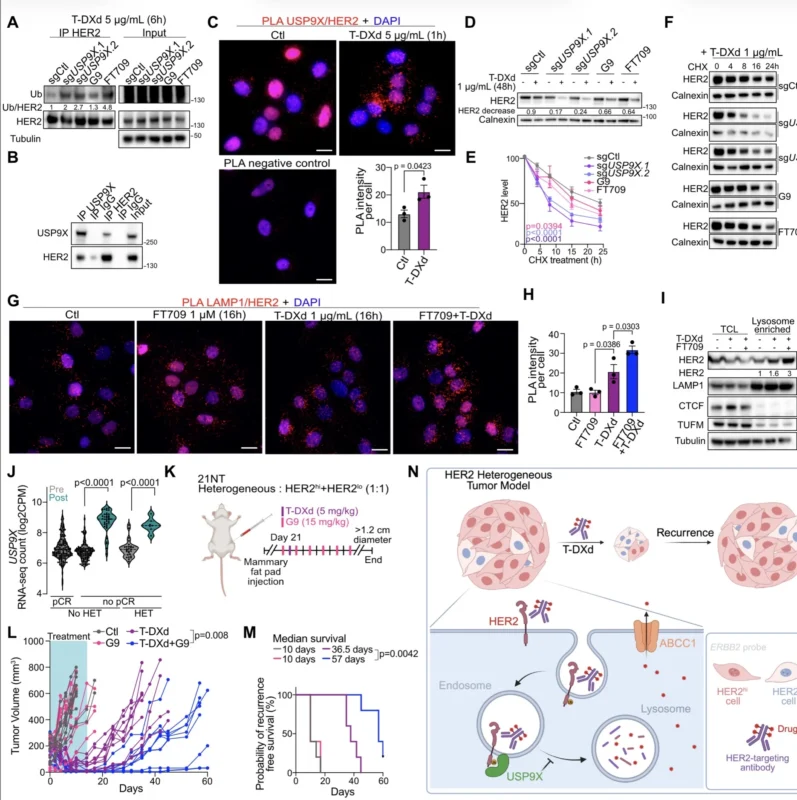
What This Could Mean for Treatment Sequencing
The discussion section places these findings in a broader clinical context. The authors argue that HER2 heterogeneity itself may function as a biomarker of relative resistance to HER2-targeted ADCs. They also note that HER2-low subclones remained sensitive to HER2 kinase inhibitors, supporting combination approaches that could hit both HER2-high and HER2-low compartments at the same time. In their models, combining T-DXd with neratinib was more effective than either agent alone. While toxicity may limit immediate frontline use of such strategies, the concept is biologically strong and deserves further exploration (Freedman et al., 2024; Li et al., 2020).
The paper also tempers enthusiasm appropriately. These are sophisticated preclinical systems, but they are still models. USP9X inhibitors remain early in development, and combinations that deepen ADC activity must be evaluated carefully for safety. Even so, the study succeeds in doing something valuable: it moves the field from a vague recognition that “heterogeneous tumors do worse” to a specific, testable biological explanation for why that happens.
A More Practical View of HER2-Positive Disease
For clinicians and researchers, the message is straightforward. Not every HER2-positive tumor should be expected to respond like a uniformly HER2-addicted tumor. Some contain a therapeutically important HER2-low compartment that can endure ADC exposure, shape recurrence, and potentially be targeted through alternative or combination strategies. Goyette et al. provide a persuasive preclinical framework showing that HER2 heterogeneity is both a mechanism of resistance and a therapeutic opportunity.
In breast cancer, that may influence how future trials stratify patients, how resistance biopsies are interpreted, and how combinations around T-DXd are designed. More broadly, the work may have implications beyond breast cancer, since HER2 heterogeneity has also been reported in gastric and endometrial tumors. The central lesson is that the presence of HER2 is not the whole story. The distribution of HER2 within a tumor may be just as important.
You Can Read Full Article Here