The long-term follow-up of the phase II GeparNuevo trial adds an important new layer to the discussion around immunotherapy in early triple-negative breast cancer. While the original study focused primarily on pathologic complete response, the newly reported analysis shows that adding durvalumab to neoadjuvant chemotherapy was associated with sustained improvements in long-term survival outcomes, even though checkpoint inhibition was not continued in the adjuvant setting. That is the part of the dataset that makes these findings especially thought-provoking for current practice (Loibl et al., 2026).
With a median follow-up of 86.4 months, compared with the previously reported 43.7 months, the durvalumab arm demonstrated significant benefit across several clinically meaningful endpoints. These included invasive disease-free survival, distant disease-free survival, and overall survival, suggesting that the impact of immunotherapy in this trial extended beyond short-term response and into durable long-term outcome (Loibl et al., 2026).
Why GeparNuevo Still Matters
Early triple-negative breast cancer has become one of the most dynamic spaces in breast oncology. The success of immune checkpoint inhibitors in the neoadjuvant setting, especially when combined with chemotherapy, has changed expectations around pathologic response and event-free survival. Yet an important question has remained unsettled: how much of the benefit comes from the neoadjuvant immune priming phase itself, and how much depends on continuing immunotherapy after surgery?
That is why GeparNuevo continues to attract attention. Unlike more recent treatment strategies that include adjuvant checkpoint inhibitor continuation, this trial evaluated durvalumab only during the neoadjuvant phase, together with chemotherapy. The fact that long-term benefit persisted without postoperative continuation makes the updated analysis clinically relevant far beyond the original study design (Loibl et al., 2026).
Study Design and Treatment Approach
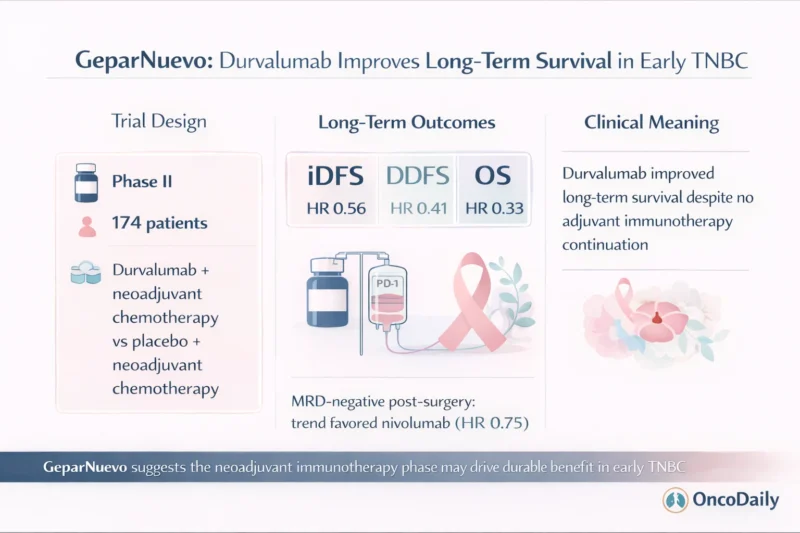
GeparNuevo was a phase II trial in patients with early triple-negative breast cancer, including tumors ranging from cT1b to cT4a-d. A total of 174 patients were randomly assigned to receive either durvalumab or placebo concurrently with neoadjuvant chemotherapy. The chemotherapy backbone consisted of weekly nab-paclitaxel, followed by dose-dense epirubicin and cyclophosphamide (Loibl et al., 2026).
The trial had originally been designed to examine whether the addition of durvalumab could improve pathologic complete response. The updated long-term report extends that question into survival, using STEEP-defined endpoints and stratifying all analyses by baseline stromal tumor-infiltrating lymphocytes, grouped as low, intermediate, or high (Loibl et al., 2026).
Long-Term Survival Benefit Becomes Clearer With Time
The most important message from this updated analysis is that the benefit of durvalumab became more convincing with longer follow-up. The study showed a statistically significant improvement in invasive disease-free survival, with a hazard ratio of 0.56 and a stratified log-rank P value of .0431. This indicates a meaningful reduction in invasive recurrence or death with neoadjuvant durvalumab compared with placebo (Loibl et al., 2026).
The effect was even more striking for distant disease-free survival, where the hazard ratio was 0.41 and the P value was .0069. In triple-negative breast cancer, distant relapse remains the event clinicians fear most because it is closely tied to long-term mortality. A hazard ratio of this magnitude suggests that the addition of durvalumab may have substantially reduced the risk of metastatic recurrence over time (Loibl et al., 2026).
Most notable of all, the trial also reported a significant improvement in overall survival, with a hazard ratio of 0.33 and a P value of .0085. For an early-stage triple-negative breast cancer study of this size, that is an especially important signal. It suggests that the benefit of neoadjuvant durvalumab was not limited to disease control measures alone but may translate into a real survival advantage (Loibl et al., 2026).
A Signal Stronger Than pCR Alone
One of the most interesting aspects of this report is that the authors emphasize survival improvement irrespective of the extent of pathologic response. This is important because neoadjuvant trials in triple-negative breast cancer are often interpreted heavily through the lens of pathologic complete response. pCR remains valuable, but GeparNuevo reminds us that it is not the whole story.
The updated data suggest that immunotherapy may shape long-term outcomes in ways that are not fully captured by the binary distinction between pCR and residual disease. In other words, some of the benefit of durvalumab may come from deeper biologic effects on micrometastatic disease or host immune control that continue to matter long after surgery and long after neoadjuvant treatment has ended (Loibl et al., 2026).
Nodal Status May Matter
In the exploratory subgroup analysis, patients with nodal involvement at baseline appeared to derive greater benefit in invasive disease-free survival from durvalumab. In this subgroup, the hazard ratio for iDFS was 0.33, with a P value of .01, and the interaction test was also statistically significant (Pinteraction = 0.045) (Loibl et al., 2026).
This finding should be interpreted carefully because it comes from an exploratory analysis, but it is clinically meaningful. Node-positive triple-negative breast cancer represents a group with higher baseline risk, and if the benefit of neoadjuvant durvalumab is indeed greater there, that could help refine how clinicians think about treatment intensity and patient selection. It also aligns with a broader principle seen across oncology: patients with higher-risk disease often have more to gain from effective systemic escalation.
Residual Disease and sTILs Add More Biological Detail
Another interesting part of the report concerns stromal tumor-infiltrating lymphocytes in residual disease. Among the 71 patients without pCR, residual disease sTILs could be assessed in 39. In a post hoc analysis, patients with high residual-disease sTILs (>10%) had an estimated 7-year invasive disease-free survival rate of 92.3%, compared with 51.4% in those with low sTILs (≤10%) (Loibl et al., 2026).
This is a particularly relevant observation because residual disease in triple-negative breast cancer is usually associated with poorer prognosis. The idea that the immune composition of residual disease may sharply differentiate long-term risk is biologically compelling. It suggests that not all residual disease is equal. Some tumors may remain biologically inflamed and under partial immune control, while others may reflect a more immune-depleted and aggressive phenotype.
Although the analysis was post hoc and based on a relatively small number of patients, it points toward an important future direction: residual disease after neoadjuvant therapy may need to be characterized not only by volume and pathology, but also by immune context.
The Most Practice-Shaping Implication
The line in this paper that will likely generate the most discussion is the authors’ conclusion that these results underscore the necessity to re-evaluate the adjuvant continuation of checkpoint inhibition (Loibl et al., 2026).
That does not mean adjuvant continuation should be abandoned. But it does mean the field may need to think more critically about how much benefit is truly being added by postoperative checkpoint inhibitor therapy when strong neoadjuvant immune priming has already occurred. GeparNuevo cannot answer that question definitively because it was not designed as a direct comparison of neoadjuvant-only versus perioperative continuation. Still, the durable survival benefit seen here without adjuvant immunotherapy makes the question unavoidable.
In a treatment era increasingly focused on optimizing intensity rather than simply adding more therapy, this is exactly the kind of signal that matters.
How These Results Fit Into the Broader TNBC Immunotherapy Story
The broader immunotherapy story in early triple-negative breast cancer has often been shaped by larger perioperative strategies that include both neoadjuvant and adjuvant phases. GeparNuevo now offers a different perspective. It suggests that a meaningful share of the long-term benefit may already be established during the neoadjuvant period itself.
That matters clinically for several reasons. First, it raises questions about overtreatment, especially when checkpoint inhibitors are associated with immune-related toxicity, cost, and prolonged treatment burden. Second, it reinforces the importance of the neoadjuvant window as a biologically active period where immune priming may have outsized long-term consequences. Third, it supports further study of biomarkers such as nodal status and residual-disease sTILs to identify which patients might need more therapy and which may not.

Limitations That Should Be Kept In Mind
As important as these results are, GeparNuevo remains a phase II trial with a relatively modest sample size of 174 patients. The subgroup analyses are exploratory, and the residual disease immune analyses are based on a limited subset. That means the findings should be viewed as highly informative and hypothesis generating, but not as the final word on postoperative checkpoint inhibitor strategy.
Still, the strength of the long-term endpoints, especially the overall survival signal, makes this trial unusually influential for its size.
The Clinical Takeaway
The long-term GeparNuevo analysis shows that adding durvalumab to neoadjuvant chemotherapy in early triple-negative breast cancer was associated with sustained improvement in invasive disease-free survival, distant disease-free survival, and overall survival, even without adjuvant continuation of checkpoint inhibition (Loibl et al., 2026).
That is the central message. This was not a transient pCR story. It became a long-term survival story.
Read full abstract here


