For patients with locally recurrent inoperable or metastatic triple-negative breast cancer, first-line treatment decisions remain difficult when immunotherapy is not an option. This is still the reality for a large proportion of patients. Despite progress in PD-L1-positive disease and in selected biomarker-defined settings, chemotherapy continues to be the mainstay for many women with advanced TNBC, usually with limited durability and modest response rates (Punie et al., 2025; Huppert et al., 2022; Li et al., 2019).
The phase III TROPION-Breast02 trial now adds an important new option to that space. In this randomized international study, datopotamab deruxtecan (Dato-DXd) demonstrated statistically significant improvements in both progression-free survival and overall survival compared with investigator’s choice of chemotherapy in previously untreated advanced TNBC for whom immunotherapy was not an option (Dent et al.).
Why This Study Matters
Triple-negative breast cancer accounts for around 10% to 20% of breast cancers and is still associated with early relapse, visceral spread, brain metastases, and short survival once metastatic disease develops (Howlader et al., 2018; O’Reilly et al., 2021; Baranova et al., 2022). Although checkpoint inhibitors have changed outcomes for some patients, this benefit is not universal. In the TROPION-Breast02 paper, the authors note that approximately 70% of patients with metastatic TNBC are not candidates for immunotherapy, leaving chemotherapy as the default first-line approach for most of this population (Punie et al., 2025).
That is exactly the group addressed here.
Dato-DXd is a TROP2-directed antibody-drug conjugate composed of a humanized anti-TROP2 monoclonal antibody linked to a topoisomerase I inhibitor payload through a tumor-selective cleavable linker. Earlier studies had already shown activity in pretreated metastatic TNBC and HR-positive/HER2-negative disease, but TROPION-Breast02 asked a more ambitious question: could Dato-DXd outperform standard chemotherapy in the first-line setting for advanced TNBC when immunotherapy is not used (Okajima et al., 2021; Bardia et al., 2024; Dent et al., 2023)?
Trial Design at a Glance
TROPION-Breast02 was a randomized, open-label, phase III trial that enrolled patients with previously untreated, locally recurrent inoperable or metastatic TNBC. Patients had to have measurable disease and ECOG performance status 0 or 1. The study also included patients with PD-L1-low tumors, as well as selected patients with PD-L1-high tumors who could not receive PD-(L)1 therapy because of prior use in early disease, comorbidities, or lack of regulatory access.
A total of 644 patients were randomized 1:1 to receive either Dato-DXd 6 mg/kg intravenously every 3 weeks or investigator’s choice of chemotherapy. The chemotherapy options reflected prior taxane exposure and disease-free interval and included nab-paclitaxel, paclitaxel, eribulin, carboplatin, or capecitabine. Randomization was stratified by geographic region, disease-free interval, and PD-L1 status.
The dual primary endpoints were PFS by blinded independent central review and overall survival.
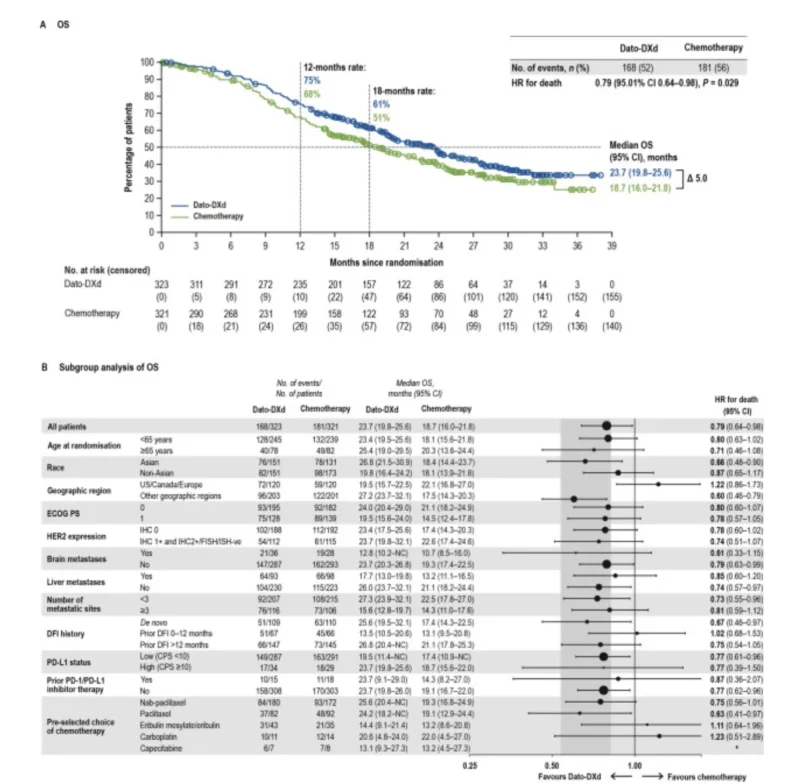
A Clear PFS Win for Dato-DXd
The most immediate efficacy signal in TROPION-Breast02 was the magnitude of the PFS benefit.
Median PFS was 10.8 months with Dato-DXd versus 5.6 months with chemotherapy, corresponding to a 43% reduction in the risk of progression or death with Dato-DXd (HR 0.57; 99% CI 0.44–0.73; P < 0.0001). The separation of the Kaplan–Meier curves was early and sustained. At 12 months, the PFS rate was 46% with Dato-DXd versus 26% with chemotherapy, and at 18 months it was 33% versus 17%.
This was not a result driven by one narrow subgroup. According to the study, the PFS advantage was consistent across all prespecified patient subgroups, including PD-L1 status, geographic region, disease-free interval, HER2-low versus HER2-0 expression, liver metastases, brain metastases, and number of metastatic sites.
In OncoDaily terms, that matters. It means this was not just statistically significant. It looked clinically broad.
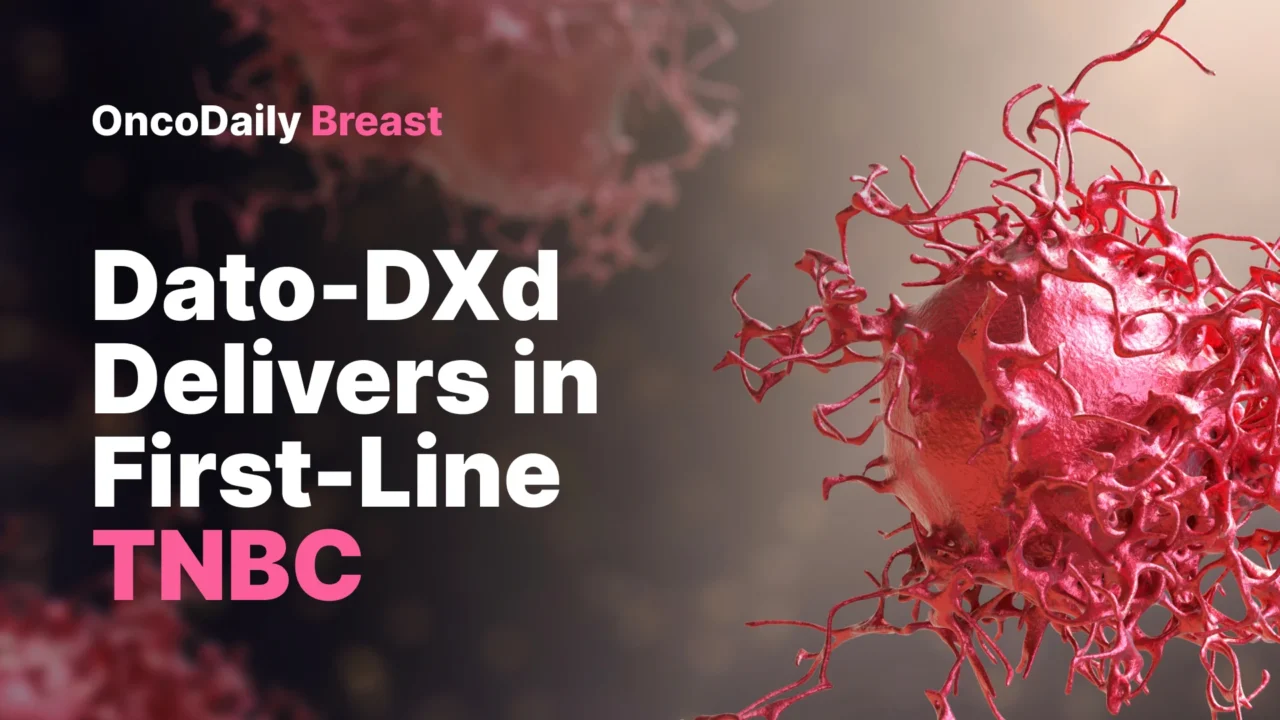
Overall Survival Also Improved
The second major result was that Dato-DXd also improved overall survival, which is what makes this trial especially important.
Median OS was 23.7 months with Dato-DXd compared with 18.7 months with chemotherapy, for an absolute difference of 5 months. The hazard ratio for death was 0.79 (95.01% CI 0.64–0.98; P = 0.029), meeting statistical significance.
The 12-month OS rate was 75% with Dato-DXd versus 68% with chemotherapy, and the 18-month rate was 61% versus 51%.
That is a meaningful result in first-line metastatic TNBC, especially in a population with limited therapeutic alternatives.
Response Rates and Durability Favored the ADC
The benefit was not limited to time-to-event endpoints. Dato-DXd also produced a much higher response rate than chemotherapy.
The confirmed objective response rate was 63% with Dato-DXd versus 29% with chemotherapy. Complete responses were seen in 9% of patients receiving Dato-DXd compared with 2% in the chemotherapy arm. Disease control at 12 weeks was also higher, 84% versus 60%.
Just as important, responses lasted longer. Median duration of response was 12.3 months with Dato-DXd and 7.1 months with chemotherapy.
This combination of higher response rate plus longer durability helps explain why the survival curves separated the way they did.
Who Was Included?
One strength of TROPION-Breast02 is that the enrolled population looked clinically relevant.
About one-third of patients had de novo metastatic disease. Brain metastases were present in 11% of the Dato-DXd arm and 9% of the chemotherapy arm. Liver metastases were seen in roughly 29% to 31%. Around 15% to 16% had a disease-free interval of 0 to 6 months, a group known to have particularly poor outcomes. Prior taxane exposure was common, and most patients had already received systemic therapy in the neoadjuvant or adjuvant setting.
This was not a trial built around unusually favorable biology. It included patients clinicians recognize from daily practice.
The Subgroup Story Deserves Attention
Although the overall results were clearly positive, the subgroup analyses add nuance.
The PFS benefit remained consistent across all prespecified subgroups, which strengthens the main efficacy message. The OS benefit also generally favored Dato-DXd, but not every subgroup showed the same pattern. The paper notes that the United States/Canada/Europe subgroup had an exploratory OS hazard ratio above 1, as did some small subgroups defined by disease-free interval or prespecified chemotherapy choice.
The authors were appropriately cautious here. These subgroup analyses were descriptive, not adjusted for multiplicity, and some had small numbers with wide confidence intervals. They also identified imbalances in poor prognostic features within the United States/Canada/Europe subgroup, with more patients in the Dato-DXd arm having multiple metastatic sites, liver metastases, and brain metastases. In addition, subsequent ADC use was more frequent in the chemotherapy arm in that region. When the prespecified IPCW method was applied to adjust for these factors, the OS hazard ratio in that subgroup shifted to 0.90, suggesting confounding rather than a true reversal of treatment effect.
That is exactly the kind of detail that matters when interpreting a major phase III trial.
Safety Was Familiar, but the Pattern Was Different From Chemotherapy
The safety profile of Dato-DXd in TROPION-Breast02 was consistent with what has already been seen with this ADC.
Any treatment-related adverse event was reported in 93% of patients receiving Dato-DXd and 83% receiving chemotherapy. Grade 3 or higher treatment-related events occurred in 33% and 29%, respectively. Serious treatment-related adverse events were reported in 9% versus 8%. Importantly, treatment discontinuation due to treatment-related adverse events was lower with Dato-DXd, 4% versus 7%, and there were no treatment-related deaths in either arm.
The most common Dato-DXd toxicities were stomatitis, nausea, and alopecia. Stomatitis occurred in 57%, nausea in 45%, and alopecia in 41%. Ocular surface events were also notable, including dry eye and keratitis, which are now recognized parts of the Dato-DXd toxicity profile.
Most oral mucositis and ocular events were low grade, and many improved with management and dose modification. No patients discontinued Dato-DXd because of oral mucositis or stomatitis, while only 1% discontinued due to ocular surface events.
Interstitial lung disease or pneumonitis remains a toxicity of special interest with deruxtecan-based ADCs. In this study, 9 patients (3%) in the Dato-DXd arm had adjudicated drug-related ILD/pneumonitis events. Most were grade 1 or 2, but one patient had a fatal event described in the adjudication dataset, although the death itself was assessed by the investigator as related to breast cancer. This remains an important safety consideration in clinical use.
Patient-Reported Outcomes Also Leaned Toward Dato-DXd
An underappreciated part of the trial is the patient-reported outcomes dataset.
Time to deterioration in pain, physical functioning, global health status/quality of life, and breast and arm symptoms was numerically in favor of Dato-DXd. In a disease where first-line treatment is often palliative and symptom control matters greatly, that direction of effect supports the clinical relevance of the efficacy benefit.
This is particularly meaningful because patients on Dato-DXd stayed on treatment much longer than patients on chemotherapy. Median treatment duration was 8.5 months with Dato-DXd versus 4.1 months with chemotherapy, yet discontinuation from toxicity remained lower.
Where This Fits in the Evolving TNBC Landscape
TROPION-Breast02 arrives in a rapidly changing first-line TNBC setting. Immunotherapy remains essential for eligible PD-L1-positive patients, and PARP inhibitors still matter for selected germline BRCA-mutated disease. But outside those groups, clinicians have long relied on chemotherapy with limited expectations.
This trial suggests that TROP2-directed ADCs are moving earlier in the treatment course. The paper also references the phase III ASCENT-03 study of sacituzumab govitecan, which showed improved PFS but not a clear OS advantage at the time of the primary analysis. TROPION-Breast02 now provides a phase III dataset in which a TROP2 ADC improved both PFS and OS against chemotherapy in the first-line setting.
That does not automatically settle sequencing questions between different ADCs. It does, however, make one point much clearer: the first-line treatment standard for patients with advanced TNBC who cannot receive immunotherapy is no longer a chemotherapy-only conversation.
The Bottom Line
TROPION-Breast02 is a clinically important trial.
In patients with previously untreated, locally recurrent inoperable or metastatic TNBC for whom immunotherapy was not an option, datopotamab deruxtecan improved progression-free survival, overall survival, objective response rate, and duration of response compared with chemotherapy. The safety profile was consistent with prior experience, with stomatitis, nausea, ocular events, and ILD remaining key areas to monitor, but discontinuation due to toxicity was lower than with chemotherapy.
For a population that has historically had few good first-line options, this is a meaningful advance.
You can read full article here