On April 17, Agenus Inc. announced findings from an investigator-initiated Phase II trial conducted at Memorial Sloan Kettering Cancer Center, evaluating a novel combination of botensilimab (BOT), balstilimab (BAL), and MiNK Therapeutics’ allo-iNKT cell therapy, agenT-797, in patients with advanced gastroesophageal adenocarcinoma that had progressed on prior PD-1-based treatment. The data were presented at the 2026 American Association for Cancer Research (AACR) Annual Meeting in San Diego.
Dhan Chand, Ph.D., Vice President of Research at Agenus, said:
“These findings illustrate the mechanistic synergy of agenT-797 with botensilimab and balstilimab in this PD-1 refractory setting,”

“The induction approach promoted significant intratumoral infiltration of T cells and dendritic cells, the formation of organized tertiary lymphoid structures in on-treatment biopsy tissue from a patient with durable benefit, and activation of peripheral CD4 and CD8 T-cell populations. These changes are consistent with immune priming and tumor immune reprogramming, providing a biological rationale for the improved progression-free survival observed with the induction strategy.”
Understanding BOT/BAL
Botensilimab (BOT) and balstilimab (BAL) are investigational immunotherapies developed by Agenus Inc., designed to enhance anti-tumor immune responses through complementary mechanisms.
Botensilimab is an Fc-enhanced anti–CTLA-4 antibody that activates both innate and adaptive immunity. It promotes anti-tumor activity by improving T-cell priming and expansion, reducing immunosuppressive regulatory T cells within the tumor microenvironment, and engaging myeloid cells. It may also support the development of long-term immune memory, potentially contributing to more durable responses.
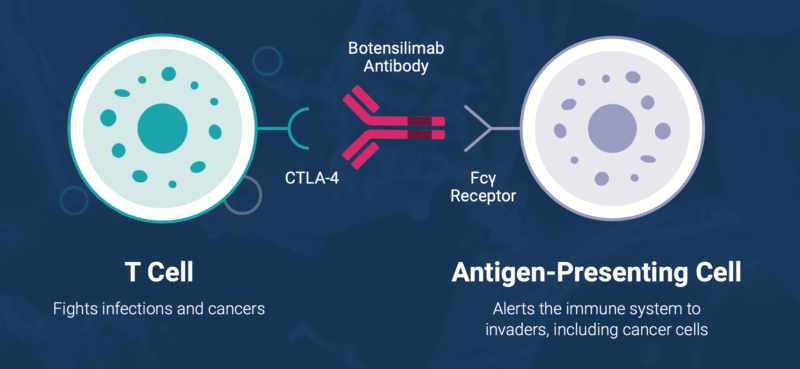
Balstilimab is a PD-1 inhibitor that blocks the interaction between PD-1 on T cells and its ligands PD-L1 and PD-L2. By inhibiting this pathway, it removes inhibitory signals that restrain T-cell function, helping to restore the immune system’s ability to recognize and attack cancer cells.
Together, botensilimab and balstilimab are intended to produce a complementary and potentially synergistic immune response, with the aim of improving outcomes in tumors that are typically less responsive to conventional immunotherapy.
Background and Study Rationale
Patients with advanced gastroesophageal adenocarcinoma who progress after frontline therapy and become refractory to PD-1 blockade face very limited treatment options. This study explores a multi-agent immunotherapy strategy designed to enhance immune activation and reprogram the tumour microenvironment. The regimen combined botensilimab, balstilimab, agenT-797, as well as ramucirumab and paclitaxel. The trial was designed not only to evaluate efficacy, but also to investigate the role of immune priming and treatment sequencing.
Methods
This investigator-initiated Phase II trial was conducted at Memorial Sloan Kettering Cancer Center and enrolled patients with advanced gastroesophageal adenocarcinoma whose disease had progressed after frontline therapy.
The study (n=17) was designed to explore immune priming and treatment sequencing. Patients received induction with agenT-797, either alone or in combination with BOT and BAL, followed by the full regimen including ramucirumab and paclitaxel, or initiated the full combination regimen without induction. Longitudinal biomarker sampling was performed throughout the study to evaluate immune activation and tumor microenvironment changes over time.
Presentation at AACR 2026
The results were presented on April 20, 2026, at the AACR Annual Meeting 2026 in San Diego, during the Phase II and Phase III Clinical Trials session.
The abstract, titled “A phase II study of agenT-797, botensilimab (BOT) and balstilimab (BAL) in PD-1 refractory gastroesophageal cancer (GEC)”, was presented by Samuel L. Cytryn, M.D., gastrointestinal medical oncologist at Memorial Sloan Kettering Cancer Center. It was included in Poster Section 52 (Abstract CT166) during the session scheduled from 2:00–5:00 PM PT / 5:00–8:00 PM EDT.
Key Findings
According to Agenus, the combination achieved a disease control rate (DCR) of 77% across all treated patients, with a subset experiencing long-term survival beyond 20 months.
The induction strategy was associated with improved outcomes. Median progression-free survival (PFS) was 6.9 months in the induction cohort compared with 3.5 months in patients treated without induction (HR 0.19; p=0.015). Median overall survival (OS) was 9.5 months versus 5.2 months, respectively. Additionally, 43% of patients in the induction cohort were alive at both 12 and 18 months, compared with 20% and 0% in the non-induction cohort.
The study did not meet its primary endpoint of objective response rate (ORR). However, the observed disease control and the durability of survival in a subset of patients support further investigation of this approach in this refractory population, where long-term outcomes remain limited. Agenus noted that durability and survival may be the most clinically relevant endpoints in this PD-1 refractory population.
You can also read about Phase 3 BATTMAN Trial for pMMR Metastatic Colorectal Cancer on OncoDaily.
Translational Insights and Mechanism
Correlative analyses showed that treatment with BOT, BAL, and agenT-797 was associated with significant intratumoral infiltration of T cells and dendritic cells, as well as activation of peripheral CD4 and CD8 T-cell populations. The formation of organized tertiary lymphoid structures was also observed in tumor tissue from a patient with durable benefit.
These findings are consistent with immune priming and tumor immune reprogramming, providing a biological rationale for the improved progression-free survival observed with the induction strategy.
Botensilimab is a human Fc-enhanced multifunctional anti-CTLA-4 antibody designed to stimulate both innate and adaptive immune responses. Balstilimab is a fully human monoclonal IgG4 antibody targeting PD-1. AgenT-797, developed through MiNK Therapeutics, is described as an allo-iNKT cell therapy intended to further enhance immune activation.
Safety Profile
The safety profile was consistent with what has been previously reported for each component agent. The most frequently observed treatment-emergent adverse events included fatigue, fever, diarrhea, anorexia, nausea, and mucositis. Immune-mediated side effects encompassed dermatitis, colitis, gastritis, enteritis, hepatitis, and hypothyroidism.
Conclusion
This early-phase study evaluates a novel combination strategy integrating checkpoint inhibition, cellular therapy and chemotherapy in PD-1 refractory gastroesophageal cancer. While objective responses were limited, the high disease control rate and signals of prolonged survival—particularly with an induction approach—highlight the potential importance of immune priming and treatment sequencing.
Further analyses of the full biospecimen dataset are ongoing and are expected to provide additional insights into immune mechanisms, optimal sequencing strategies and potential biomarkers to better identify patients most likely to benefit.
Full details are available in the Agenus press release.
You can also read about 10 Updates in Gastroesophageal cancer on OncoDaily.




