On April 1, 2026, Agenus Inc. announced that the first patient has been enrolled in the global Phase 3 BATTMAN (CO.33) trial (NCT07152821), evaluating the combination of botensilimab (BOT) and balstilimab (BAL) in patients with refractory, unresectable microsatellite stable (MSS) or mismatch repair proficient (pMMR) metastatic colorectal cancer.
Dr. Steven O’Day, Chief Medical Officer, Agenus, said:
“Enrollment of the first patient in the BATTMAN study marks a key milestone for Agenus and the BOT+BAL program. This study advances our goal of developing effective immunotherapies for patients who currently have few options. We’re grateful to our partners at CCTG, GI Cancer Trials in Australia, and PRODIGE and to the dedicated investigators, site staff, and patients driving this global effort.”

Steven O’Day
Trial Design and Conduct
The BATTMAN (CCTG CO.33) trial is a global, randomized, controlled Phase 3 study comparing botensilimab plus balstilimab versus best supportive care in this patient population. The trial is being conducted as an international cooperative group study led by the Canadian Cancer Trials Group (CCTG).
The study is planned to enroll approximately 830 patients across more than 100 sites in Canada, France, Australia, and New Zealand. Participating academic networks include CCTG, GI Cancer Trials in Australia, and France’s Partenariat de Recherche en Oncologie Digestive (PRODIGE) consortium, including Unicancer, GERCOR, and FFCD. The trial is designed as a registrational-enabling study to support potential regulatory submissions for the BOT+BAL combination.
Additional details from ClinicalTrials.gov (NCT07152821) further define the study as an interventional Phase 3 trial in patients with chemo-refractory, unresectable colorectal adenocarcinoma, with an estimated enrollment of 834 patients.
Key study timelines:
- Study start (actual): March 31, 2026
- Primary completion (estimated): December 2028
- Study completion (estimated): June 2029
The study is being conducted to determine whether patients with unresectable colorectal cancer treated with botensilimab and balstilimab live longer. It also evaluates whether quality of life improves, whether tumors slow in growth or shrink, and whether biomarkers in tumor tissue or blood can predict benefit. In addition, the study assesses safety and examines how long these immunotherapy drugs remain in the body.
Program Background
According to Agenus, colorectal cancer has become the leading cause of cancer-related death in adults under the age of 50, and MSS disease represents approximately 95% of metastatic colorectal cancer cases. This population has historically been resistant to immunotherapy.
The BATTMAN trial builds on earlier studies, including work conducted within the Canadian Cancer Trials Group, which suggested that doublet immunotherapy could extend survival even in tumors that are typically less responsive to immune-based treatments. The magnitude and durability of responses observed with botensilimab and balstilimab in earlier studies supported their evaluation in a Phase 3 trial.
Read more about BOT/BAL advances, the BATTMAN trial, FDA approval challenges, and key highlights from the Agenus stakeholder briefing on OncoDaily.
Clinical Development
Approximately 1,200 patients have been treated with botensilimab and/or balstilimab across Phase 1 and Phase 2 trials. Botensilimab, alone or in combination with balstilimab, has demonstrated clinical responses across nine metastatic, late-line cancers.
Balstilimab has been evaluated in more than 900 patients and has demonstrated clinical activity along with a favorable tolerability profile in several tumor types.
BOT/BAL in MSS mCRC
On September 9, 2025, Agenus Inc. (Nasdaq: AGEN) announced that its investigational immunotherapy combination, botensilimab (BOT) plus balstilimab (BAL), is now available to eligible patients with refractory microsatellite-stable (MSS) metastatic colorectal cancer (mCRC) through France’s compassionate access program (Accès Compassionnel, AAC).
The French National Agency for Medicines and Health Products Safety (ANSM) has approved hospital use of BOT/BAL for patients meeting specific criteria, including MSS status and no active liver metastases. Under this program, treatment is fully reimbursed by France’s Assurance Maladie, with hospitals compensated at the purchase price.
Garo Armen, PhD, Chairman & CEO of Agenus, said:

“This is a breakthrough for patients and their physicians. MSS colorectal cancer resists current immunotherapies, and treatment options after standard regimens are very limited. We are committed to supporting French centers to ensure a reliable supply and to collect high-quality real-world data.”
Read more about France granting reimbursed compassionate access for BOT/BAL in refractory MSS colorectal cancer on OncoDaily.
BOT/BAL in Ovarian and Sarcoma
France has expanded national compassionate access to the investigational immunotherapy combination botensilimab (BOT) plus balstilimab (BAL), extending fully reimbursed hospital-based use beyond refractory microsatellite-stable (MSS) colorectal cancer to include selected ovarian cancers and soft-tissue sarcomas.
On January 12, 2026, Agenus Inc. announced that France’s National Agency for Medicines and Health Products Safety (ANSM) approved an updated national protocol under the Autorisation d’Accès Compassionnel (AAC) framework. The revised authorization broadens eligibility for BOT/BAL to additional solid tumor indications with substantial unmet medical need after exhaustion of standard therapies.
Under the AAC program, treatment is fully reimbursed by France’s national health system (Assurance Maladie), with standardized hospital oversight and structured real-world data collection.
Read more about France expanding reimbursed compassionate access for botensilimab/balstilimab to ovarian cancer and soft-tissue sarcomas on OncoDaily.
Understanding Botensilimab and Balstilimab
Botensilimab (BOT) and balstilimab (BAL) are investigational immunotherapies developed by Agenus Inc., designed to enhance anti-tumor immune responses through complementary mechanisms.
Botensilimab is an Fc-enhanced anti–CTLA-4 antibody that stimulates both innate and adaptive immunity. It promotes immune activation by enhancing T-cell priming and expansion, reducing intratumoral regulatory T cells that suppress immune responses, and engaging myeloid cells to strengthen anti-tumor activity. In addition, it supports the development of long-term immune memory, which may contribute to sustained responses.
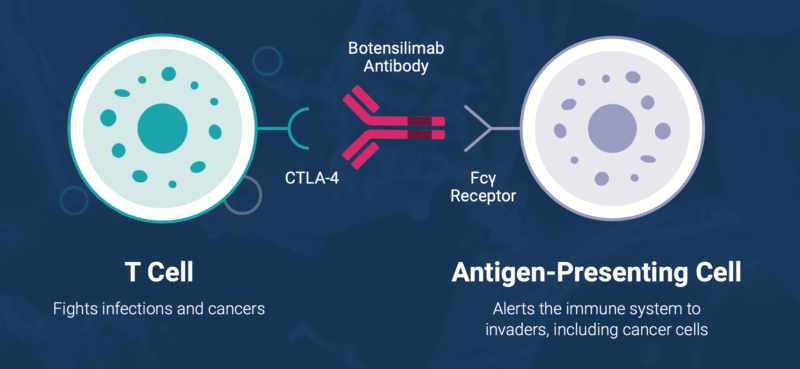
Balstilimab is a PD-1 inhibitor that blocks the interaction between PD-1 on T cells and its ligands PD-L1 and PD-L2. By inhibiting this pathway, it removes inhibitory signals that limit T-cell activity, thereby restoring the immune system’s ability to recognize and attack tumor cells.
Together, botensilimab and balstilimab are designed to generate a complementary and potentially synergistic immune response, with the goal of improving activity in tumors that are typically less responsive to standard immunotherapy approaches.
Expert Highlights
Dr. Chris O’Callaghan, DVM, PhD, Senior Investigator, Canadian Cancer Trials Group, said:
“Our collaboration with Agenus builds on years of cooperative-group research aimed at bringing immunotherapy benefits to patients with microsatellite-stable colorectal cancer—those historically left without effective options.”

Chris O’Callaghan
“Earlier CCTG studies suggested that doublet immunotherapy could extend survival even in cold tumors, and the magnitude and durability of responses seen with botensilimab and balstilimab in earlier studies warrant their investigation in a phase 3 trial.”
Dr. Jonathan Loree, MD, MSc, FRCPC, CO.33 Study Chair, said:
“The enthusiasm among investigators has been remarkable—within days of Health Canada submission, leading centers across Canada moved to open the study.”

Jonathan Loree
“We’re eager to advance this global effort and potentially transform outcomes for patients who have exhausted all other treatments.”
Key Takeaway
The initiation of the Phase 3 BATTMAN (CO.33) trial represents a key milestone in the development of botensilimab plus balstilimab for patients with MSS metastatic colorectal cancer, a population with limited treatment options and historically low responsiveness to immunotherapy.
The global scale of the study and rapid enrollment expectations reflect strong investigator and patient interest, supported in part by prior access through named patient programs and France’s AAC framework.
Full details are available in the Agenus press release.





