The week of May 4 to May 10 brings a strong selection of updates across GI oncology, highlighting progress in clinical development, translational research, biomarker-driven strategies, and evolving approaches to cancer care.
This week’s selection reflects major activity in pancreatic cancer, from the NEJM publication of daraxonrasib in previously treated RAS-mutated pancreatic cancer to new work exploring perioperative NALIRIFOX, liquid biopsy and tumor volumetry, and immune strategies targeting cDC2/CD4+ T-cell biology. Together, these updates show how precision medicine, immunology, and translational science continue to reshape the pancreatic cancer research landscape.
Important advances are also seen across colorectal, gastric, hepatocellular, and biliary tract cancers. New evidence highlights the global consistency of chemo-immunotherapy benefit in advanced gastric and GEJ cancer, the clinical activity of a c-Met–directed antibody-drug conjugate in metastatic colorectal cancer, and emerging insights into timing of immune checkpoint inhibitor administration in HCC. At the same time, liquid biopsy in biliary tract cancer and radiomics in HCC continue to expand the role of non-invasive and imaging-based tools in prognosis, monitoring, and treatment decision-making.
Together, these posts from May 4 to May 10 capture a week of meaningful progress across GI oncology, from first-in-human targeted therapy and perioperative treatment strategies to tumor biology, immunotherapy resistance, and the growing integration of biomarkers into clinical research and practice.
Wungki Park — Physician-Scientist | Translational Therapeutics Investigator | KRAS and DNA Repair Therapeutics in GI Cancers | Founder, sPARK Lab and GRIT | United States
“Honored to share our NEJM publication on daraxonrasib (RMC-6236), the first-in-human oral RAS(ON) multi-selective tri-complex inhibitor, in previously treated RAS-mutated pancreatic cancer.
More than 90% of pancreatic cancers harbor KRAS mutations, yet KRAS had long been considered “undruggable.” In this phase 1/2 study, daraxonrasib demonstrated encouraging clinical activity in refractory pancreatic cancer, including:
In second-line (2L) RAS G12-mutant PDAC
• ORR 35%• Median PFS 8.5 months
• Median OS 13.1 months
In third-line or above (3L+) RAS G12-mutant PDAC
• ORR: 21%• Median PFS 4.3 months
• Median OS 8.6 months
Strong preclinical science can translate for clinical impact and is foundational for first-in-human drug development.
Common toxicities included any %/ G3 %:
rash 96/34
stomatitis/mucositis 45/3
diarrhea 43/3
nausea 32/0
paronychia 15/0These toxicities require proactive monitoring supportive care and experience-based management
Deeply grateful to the patients, families, investigators, research teams and collaborators who made this work possible. Much more work ahead, including understanding resistance and developing smarter combinations but this represents an important step forward for RAS-driven cancers.”
Read about Daraxonrasib in Previously Treated RAS-Mutated Pancreatic Cancer on OncoDaily.
Florian Lordick, MD, FESMO — Oncologist, Professor of Medicine at University of Leipzig, Head of Medical Oncology, Director of the Comprehensive Cancer Center Central Germany | Germany
“A major question in global oncology is whether the benefits of immune checkpoint inhibition are truly consistent across different world regions.
Our new meta-analysis, now published in Gastro Hep Advances, evaluated more than 6,700 patients from seven randomized phase III trials of first-line PD-(L)1 inhibitor plus chemotherapy in advanced gastric and GEJ cancer.
The key finding:
Survival benefit was remarkably consistent in both Asian and non-Asian patient populations.This is important because gastric cancer biology, epidemiology, treatment pathways, and access to care differ substantially across regions. The data support that the benefit of chemo-immunotherapy is biology-driven rather than geography-driven.
Beyond efficacy, these findings may also help strengthen confidence in the global applicability of multiregional clinical trials and contribute to reducing disparities in access to innovative therapies worldwide.
Grateful to all co-authors and collaborators for this truly international effort. Taroh Satoh Matthew Strickland Efrat Dotan Samuel Klempner”
Michael Burns — Senior Medical Director, Late Development, Oncology at Pfizer | United States
“Nice to see this publication in Journal of Clinical Oncology reporting clinical data for telisotuzumab adizutecan (Temab-A; ABBV-400) in colorectal cancer.
Congratulations to AbbVie, the investigators, the teams, and the patients who made this work possible.
Scientific Takeaway: Temab-A, a c-Met–directed antibody–drug conjugate, demonstrated antitumor activity with a manageable safety profile in heavily pretreated metastatic CRC.”
Victor Robic — PhD Student in Oncology at Gustave Roussy, IHU PRISM, and BioMaps | France
“I am delighted to share the publication of our study “Integrating Tumor Volumetry and Liquid Biopsy: a New Model to Define Prognosis in Advanced Pancreatic Ductal Adenocarcinoma” in the European Journal of Cancer.
We evaluated whether the combination of CT and liquid biopsy improved the prognostic assessment of patients with advanced pancreatic cancer in our cohort.
A big thank you to Maxime Collin for this great teamwork, as well as to Nathalie LASSAU and Alice Boilève for their trust and for this collaboration between the imaging and medical oncology departments.”
Arndt Vogel — Senior Consultant and Professor of Gastrointestinal Oncology at Hannover Medical School | Germany
“Time-of- day of first checkpoint inhibitor dose influences clinical outcomes and immune responses in hepatocellular carcinoma
- timing of the first ICI infusion imprints a distinct early immunological program
- easy to implement”
Enes Erul — Oncology Fellow at Ankara University | Turkey
“Happy to share that our review on liquid biopsy in biliary tract cancer is now online in Expert Review of Molecular Diagnostics. Taylor & Francis Group Taylor & Francis Research Insights
- This topic is especially important because BTC is often difficult to biopsy, yet molecular profiling is increasingly central to treatment decisions.
- In the paper, we tried to bring together the current evidence on ctDNA, bile-based approaches, MRD, treatment monitoring, and resistance mechanisms while also being realistic about the limitations.
- For me, the key message is simple: liquid biopsy is not here to replace tissue, but it can be a very valuable companion when tissue is limited, when rapid molecular information is needed, or when we want to understand how the disease changes over time.
Many thanks to all my mentors Yüksel ÜRÜN and co-authors.”
Roberto Cannella — Radiologist, Researcher and Assistant Professor in Diagnostic Radiology at University of Palermo | Italy
“Happy to share our new article: Reproducibility of segmentation and radiomic features in contrast-enhanced CT images of early, intermediate and advanced HCC patients: a multicenter study published in European Journal of Radiology!
Radiomic feature reproducibility depends on HCC stage and region.
Early HCC lesions yield more reproducible features compared to advanced tumours.
Manual revision enhances concordance, particularly for lesion regions, but defining peritumoral tissue remains challenging.”
Victoire Cardot-Ruffino — Postdoctoral Fellow at Institut Curie | France
“Pancreatic cancer doesn’t respond to immunotherapy …
but maybe we’ve been targeting the wrong cells !
Our study just released in Cancer Cell shows that activating cDC2s to prime CD4+ T cells can clear checkpoint blockade-resistant tumors in mice, bypassing the conventional cDC1/CD8+ T cell pathway entirely. cDC2s outnumber cDC1s 10-to-1 in PDAC patients and they persist after chemotherapy. That’s an underexplored opportunity !
This work was a big part of my postdoc journey in Stephanie Dougan’s lab at Dana-Farber Cancer Institute and Harvard Medical School. Grateful to have been part of it !”
Davide Melisi — Associate Professor of Medical Oncology at University of Verona | Italy
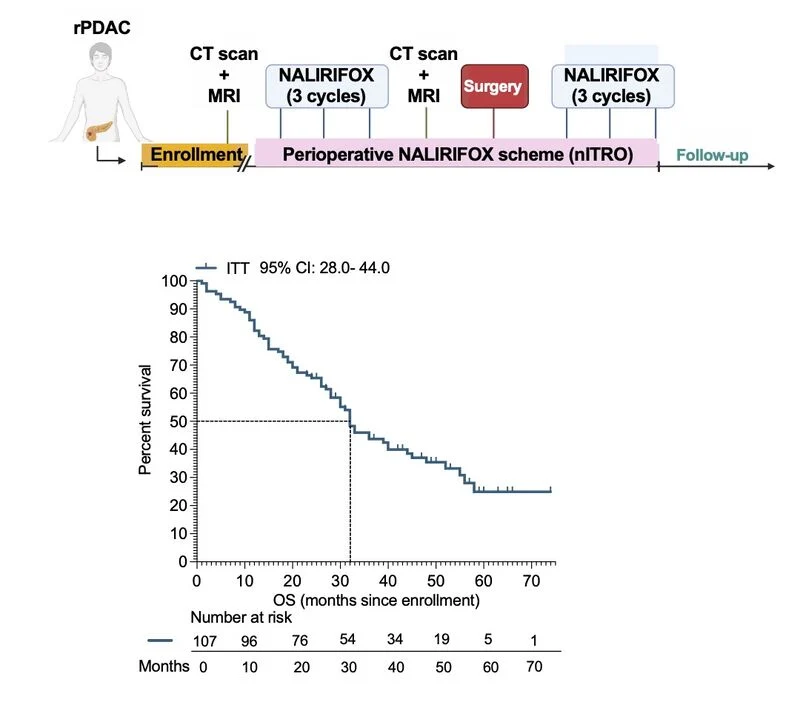
“New publication out now — and an important step forward in resectable pancreatic cancer.
In this updated analysis of the phase II nITRO trial, we explored the role of perioperative NALIRIFOX—not only confirming its clinical activity, but also digging deeper into the biology behind patient outcomes.
Why this matters:
• Median OS reached 32 months in the ITT population — 48 months in resected patients
• NALIRIFOX confirmed as an active perioperative strategy with meaningful long-term outcomes
• Biology is key:
→ High TNF-α = poorer response, lower resection rates, shorter survival
→ Germline DDR/tumor suppressor alterations = improved outcomes
• The real signal: patients with low TNF-α + g.Deficient profile achieved the best outcomes
→ mOS up to 55 months
→ 100% resection rate
• DPYD variants helped identify patients at risk of severe toxicity → opening the door to smarter dose personalizationTake-home:
NALIRIFOX isn’t just active—it’s a platform for precision perioperative therapy, where inflammation, genomics, and pharmacogenomics converge to guide decisions.”
Melanie Heinlein — PhD Scientist | Cross-Functional Collaborator in Industry & Academia | Clinical Science | United States
“Excited to finally share our paper published in #NatureCancer and with it, the end of a pretty incredible chapter at Genentech and my time in the US.
In this work, we developed an immunocompetent mouse model of metastatic colorectal cancer and found that metastatic dissemination is driven by a high-MAPK, low-WNT cell state. We also showed that inhibiting mutant KRAS can reverse this metastatic program and significantly reduce metastasis formation, highlighting just how plastic and adaptable these tumor cells are during disease progression.
This project has been years in the making and would not have been possible without an amazing group of collaborators, mentors, and friends. I’m incredibly grateful to everyone who contributed along the way.
A special thank you to Frederic de Sauvage for giving me the opportunity and freedom to truly follow the science.I feel incredibly grateful for this journey with its amazing science, the mentorship, the friendships, and all the opportunities to learn and grow along the way.”
Find out 10 Must-Read Posts in GI Oncology from April 27 to May 3 on OncoDaily.




