The ROSELLA trial is a global phase III randomized study evaluating whether adding relacorilant, a selective glucocorticoid receptor antagonist, to nab-paclitaxel can improve outcomes in patients with platinum-resistant ovarian cancer—a setting with limited treatment options and poor survival. Conducted in 381 heavily pretreated patients across 14 countries, the trial demonstrated that the combination significantly prolonged overall survival compared with chemotherapy alone, reducing the risk of death by 35% and extending median survival from 11.9 to 16.0 months. Importantly, this benefit was achieved without the need for biomarker selection, supporting a broadly applicable treatment approach and introducing a novel mechanism to enhance chemotherapy sensitivity in ovarian cancer.
Title: Overall survival with relacorilant and nab-paclitaxel in patients with platinum-resistant ovarian cancer (ROSELLA): a phase 3 randomised controlled trial
Authors: Prof Domenica Lorusso, MD, Laurence Gladieff, MD, David M O’Malley, MD, Prof Jae-Weon Kim, MD, Gabriel Garbaos, MD, Prof Anna Fagotti, MD
Background
Platinum-resistant ovarian cancer remains one of the most challenging clinical scenarios in gynecologic oncology, with median overall survival typically ranging from 10 to 17 months despite multiple treatment options. Current therapies are largely limited to single-agent chemotherapy, with or without targeted agents such as bevacizumab, or biomarker-driven approaches including mirvetuximab soravtansine and trastuzumab deruxtecan in selected populations. However, these treatments are often associated with modest benefit and limited durability.
Relacorilant, a first-in-class selective glucocorticoid receptor antagonist, represents a novel therapeutic strategy. By inhibiting cortisol-mediated survival signaling in tumor cells, relacorilant enhances sensitivity to chemotherapy and promotes apoptosis. Early-phase studies demonstrated promising activity of relacorilant combined with nab-paclitaxel, leading to the development of the phase III ROSELLA trial, designed to evaluate its impact on survival outcomes in patients with platinum-resistant disease.
Methods
The ROSELLA trial was a global, randomized, controlled, open-label phase III study evaluating the efficacy and safety of relacorilant combined with nab-paclitaxel versus nab-paclitaxel alone.
Eligible patients were adult women (≥18 years) with platinum-resistant ovarian, fallopian tube, or primary peritoneal cancer, defined as disease progression within 6 months of the last platinum-based therapy. Patients had received 1 to 3 prior lines of systemic therapy, and all had prior exposure to bevacizumab.
Patients were randomized 1:1 to receive:
- Relacorilant (150 mg orally around chemotherapy days) + nab-paclitaxel (80 mg/m²)
- Nab-paclitaxel monotherapy (100 mg/m²)
The dual primary endpoints were:
- Progression-free survival (PFS), assessed by blinded independent central review
- Overall survival (OS)
Secondary and exploratory endpoints included safety, second progression-free survival (PFS2), and patient-reported outcomes.
Study Design
A total of 381 patients were enrolled between January 2023 and April 2024 across 117 sites in 14 countries, ensuring a diverse and representative population.
Patients were heavily pretreated:
- 44% had received three prior lines of therapy
- 61% had prior exposure to PARP inhibitors
- 100% had received bevacizumab
Randomization was stratified by geographic region and number of prior therapies. The study followed patients for survival and subsequent treatments, with a median follow-up of 24.8 months.
The trial used rigorous statistical methodology, including a stratified log-rank test and Cox regression modeling, ensuring robust and reliable survival analyses.
Results
The addition of relacorilant to nab-paclitaxel demonstrated statistically significant and clinically meaningful improvements in overall survival.
Median Overall Survival (OS):
- Combination: 16.0 months
- Monotherapy: 11.9 months
- Absolute gain: 4.1 months
- Hazard Ratio for Death: 0.65 (95% CI 0.51–0.83; p=0.0004)→ representing a 35% reduction in risk of death
- 18-Month Survival Rate: Combination: 46%, Monotherapy: 27%
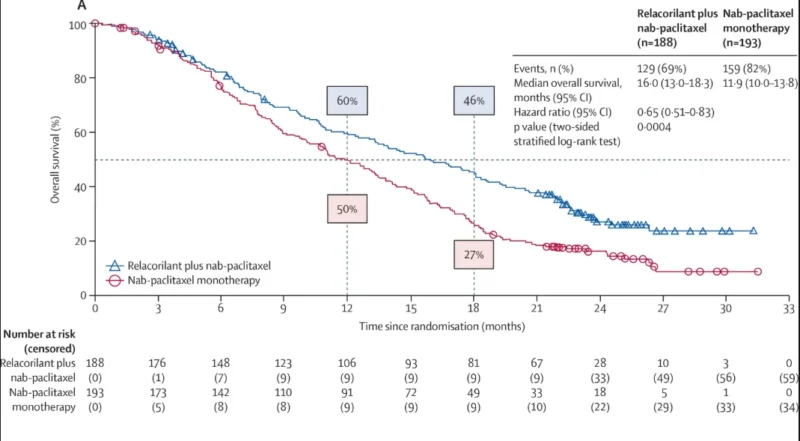
These results demonstrate a clear survival advantage, with Kaplan–Meier curves showing progressively widening separation over time, suggesting durable benefit.
Second progression-free survival (PFS2) improved significantly (HR 0.73; p=0.0037)
Benefit was consistent across all predefined subgroups, including heavily pretreated patients
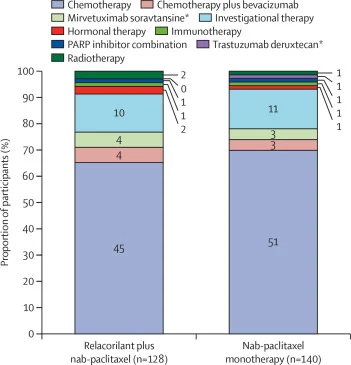
Importantly, subsequent therapies were balanced between groups, supporting that survival benefit was attributable to the study treatment rather than post-progression differences.
Key Findings
The ROSELLA trial provides several clinically important insights.The combination of relacorilant with nab-paclitaxel significantly improves survival outcomes in a population with otherwise limited treatment options. The 4.1-month median OS improvement is particularly meaningful in a disease setting where survival is often measured in months.
Unlike many modern therapies, this approach does not require biomarker selection, making it broadly applicable. This is especially relevant given that glucocorticoid receptor expression is present in over 95% of ovarian cancers, supporting an “all-comers” strategy.
The improvement in second progression-free survival indicates that the benefit extends beyond first progression, suggesting a lasting impact on disease biology and treatment trajectory.
Finally, these results validate glucocorticoid receptor antagonism as a novel and effective therapeutic mechanism in oncology.
Conclusion
The phase III ROSELLA trial establishes relacorilant plus nab-paclitaxel as a new, evidence-based treatment option for patients with platinum-resistant ovarian cancer. The combination delivers a statistically significant and clinically meaningful improvement in overall survival, with a manageable safety profile and no requirement for biomarker selection.
In a setting where therapeutic advances have historically been limited, these results represent a major step forward. The findings support the integration of relacorilant into clinical practice and justify further exploration of glucocorticoid receptor antagonists across other tumor types.


