The PROOF 301 trial was a phase III randomized study designed to evaluate whether the FGFR inhibitor infigratinib could improve outcomes compared with standard gemcitabine plus cisplatin as first-line treatment in patients with advanced cholangiocarcinoma harboring FGFR2 gene fusions or rearrangements.
Conducted across 120 sites with 1127 patients pre-screened, the study ultimately enrolled only 48 patients due to the rarity of this biomarker-defined population and significant screening challenges. While the trial was terminated early and could not provide definitive comparative conclusions, it offers important insights into the clinical activity of infigratinib—showing higher response rates but similar progression-free survival compared with chemotherapy—as well as the broader difficulties of conducting large randomized trials in rare, molecularly selected cancers.
Title: Phase III trial of infigratinib versus gemcitabine/cisplatin in adults with advanced cholangiocarcinoma with FGFR2 gene fusion or rearrangement: results and reflections on early termination of PROOF 301
Authors: G. K. Abou-Alfa, I. Borbath4, S. Roychowdhury, L. Goyal6,7, A. Lamarca8, T. Macarulla9, R. T. Shroff10, D.-Y. Oh, C. Tamas, D. M. Savastano, D. F. van Veenhuyzen, C. Xu, E. Freas, J. Solanas, M. M. Javle
Background
Advanced cholangiocarcinoma is associated with poor outcomes, with most patients presenting with unresectable or metastatic disease. For over a decade, gemcitabine plus cisplatin has been the standard first-line treatment, achieving a median overall survival of 11.7 months in the ABC-02 trial. More recently, immunotherapy combinations such as durvalumab or pembrolizumab added to chemotherapy have modestly improved survival.
Molecular profiling has identified actionable alterations in cholangiocarcinoma, particularly FGFR2 fusions or rearrangements, reported in approximately 9–16% of intrahepatic cases. Infigratinib, an oral FGFR1–3 inhibitor, demonstrated clinically meaningful activity in the phase II setting, with an overall response rate (ORR) of 23%, disease control rate of 84%, and median progression-free survival (PFS) of 7.3 months. These results led to accelerated approval in previously treated FGFR2-rearranged disease. The phase III PROOF 301 trial was designed to evaluate whether infigratinib could improve outcomes when used in the first-line setting.
Methods
PROOF 301 was a multicenter, open-label, randomized phase III trial enrolling adults with unresectable or metastatic cholangiocarcinoma harboring FGFR2 gene fusions or rearrangements. FGFR2 status was determined using local or central next-generation sequencing assays.
Eligible patients had ECOG performance status 0–1 and adequate organ function. Prior systemic therapy for advanced disease was not allowed, except limited prior exposure (≤1 cycle of gemcitabine-based therapy after protocol amendment). The primary endpoint was PFS assessed by blinded independent central review (BICR). Secondary endpoints included overall survival (OS), investigator-assessed PFS, ORR, disease control rate, duration of response (DoR), and safety.
Study Design
Patients were randomized in a 2:1 ratio to receive either infigratinib or gemcitabine plus cisplatin. Infigratinib was administered at 125 mg orally on days 1–21 of a 28-day cycle. Chemotherapy consisted of gemcitabine 1000 mg/m² plus cisplatin 25 mg/m² on days 1 and 8 of a 21-day cycle.
The trial aimed to enroll approximately 300 patients to detect a PFS improvement (hazard ratio 0.67). However, between June 2019 and October 2022, 1127 patients were pre-screened, and only 48 (4.3%) were randomized (29 to infigratinib, 19 to chemotherapy). FGFR2 rearrangements were identified in only 6.8% of screened patients (77/1127). Due to poor accrual, the study was terminated early and lacked statistical power for definitive comparisons.
Results
Baseline characteristics were balanced. Median age was 58.5 years, and 91.7% of patients had stage IV disease. Most tumors were intrahepatic (93.8%), and liver metastases were present in 77.1%.
Efficacy
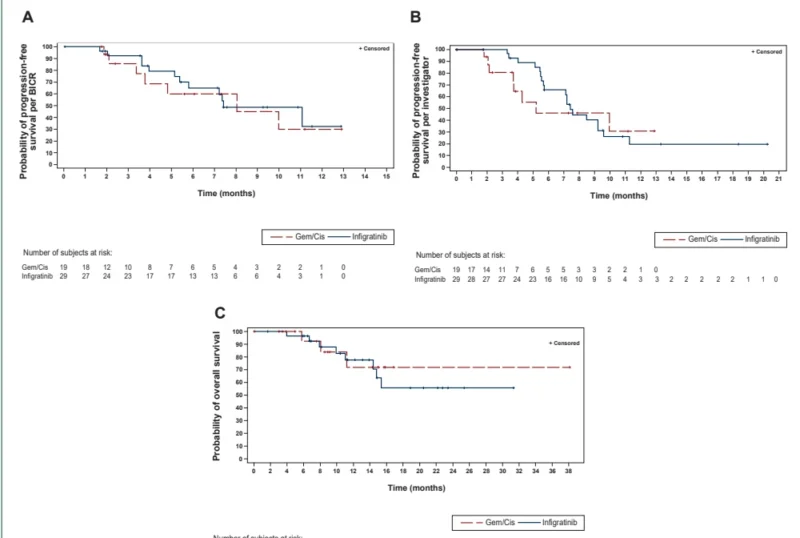
Median PFS by BICR was 7.4 months with infigratinib versus 8.0 months with chemotherapy. Six-month PFS rates were 64.9% and 59.9%, and 12-month rates were 32.5% and 29.9%, respectively.
Despite similar PFS, response rates favored infigratinib. ORR by BICR was 37.9% versus 15.8%, with all responses being partial. Disease control rates were 86.2% with infigratinib and 73.7% with chemotherapy. Median DoR was 5.6 months for infigratinib and not evaluable for chemotherapy.
Investigator-assessed outcomes showed a median PFS of 7.4 months with infigratinib versus 5.2 months with chemotherapy. ORR was 31.0% versus 10.5%, and disease control rate was 96.6% versus 73.7%, again favoring infigratinib.
Overall survival data were immature. Median OS was not reached in either arm. Estimated 12-month OS rates were 77.7% for infigratinib and 71.9% for chemotherapy.
Safety
All patients experienced treatment-emergent adverse events. Grade 3–4 events occurred in 79.3% of patients receiving infigratinib compared with 58.8% with chemotherapy. Serious adverse events occurred in 34.5% of infigratinib-treated patients.
Common grade 3–4 toxicities with infigratinib included elevated ALT (10.3%), elevated AST (10.3%), and palmar-plantar erythrodysesthesia (10.3%). In the chemotherapy arm, neutropenia (29.4%) and anemia (11.8%) were most frequent.
Hyperphosphatemia was the most common treatment-related adverse event with infigratinib (82.8%), followed by fatigue, diarrhea, dry eye, and stomatitis. Treatment discontinuation due to adverse events occurred in 24.1% of infigratinib-treated patients and 23.5% of chemotherapy-treated patients.
Key Findings
PROOF 301 was unable to meet its primary objective due to early termination and insufficient enrollment. Although PFS was similar between arms by central review, infigratinib demonstrated higher response rates and disease control compared with chemotherapy. These findings are consistent with prior phase II data but remain descriptive.
A critical observation was the extremely low enrollment efficiency. Over 1100 patients were screened to randomize only 48, largely due to the low prevalence of FGFR2 alterations (6.8%) and screening challenges. Additionally, evolving standards of care, including immunotherapy combinations, likely reduced the attractiveness of the control arm.
Conclusion
The PROOF 301 trial provides important insights despite its early termination. Infigratinib showed meaningful antitumor activity but did not demonstrate a clear PFS advantage over chemotherapy in this underpowered study. More importantly, the trial underscores the major logistical and methodological challenges of studying targeted therapies in rare molecular subgroups.
Future progress in FGFR2-rearranged cholangiocarcinoma will depend not only on effective drugs but also on innovative clinical trial designs and improved molecular screening strategies.


