The MONTBLANC trial has completed patient recruitment, marking an important milestone in the evaluation of a novel triple immunotherapy strategy for advanced hepatocellular carcinoma (HCC). The study investigates the combination of three mechanistically distinct agents — durvalumab, an anti–PD-L1 antibody; tremelimumab, an anti–CTLA-4 antibody; and bevacizumab, an anti-VEGF therapy — with the goal of enhancing anti-tumor immunity while targeting the tumor microenvironment.
With enrollment now complete, MONTBLANC moves closer to generating clinically meaningful efficacy and safety data for patients with unresectable HCC.
Background
Hepatocellular carcinoma (HCC) remains a major global oncology challenge and one of the leading causes of cancer-related mortality worldwide. A substantial proportion of patients are diagnosed with advanced or unresectable disease, when potentially curative approaches such as surgical resection, liver transplantation, or local ablation are no longer feasible. In this setting, systemic therapy plays a central role in disease control and survival prolongation.
Over the past several years, immune checkpoint inhibitor (ICI)-based combinations have reshaped the therapeutic landscape of advanced HCC. Strategies targeting PD-1/PD-L1, CTLA-4, and VEGF-mediated angiogenesis have demonstrated clinically meaningful activity and established new standards of care. However, durable benefit remains restricted to a subset of patients, and primary or acquired resistance continues to limit long-term outcomes.
These challenges underscore the need for rational combination strategies designed to enhance anti-tumor immunity, modify the tumor microenvironment, and improve the depth and durability of response in unresectable HCC.
About MONTBLANC Trial
The MONTBLANC study evaluates whether intensifying immunotherapy through a triple combination approach can improve outcomes compared with a more adaptive, response-guided strategy. Patients with unresectable HCC eligible for systemic treatment were enrolled and stratified into different therapeutic pathways.
MONTBLANC is a randomized, open-label, phase II clinical trial designed to compare two treatment strategies:
- In the upfront triple therapy arm, patients receive durvalumab, tremelimumab, and bevacizumab from treatment initiation, aiming to maximize early immune activation and tumor control.
- In the sequential treatment arm, patients begin with dual immunotherapy (durvalumab plus tremelimumab), with bevacizumab introduced later in cases of disease progression or insufficient radiologic response.
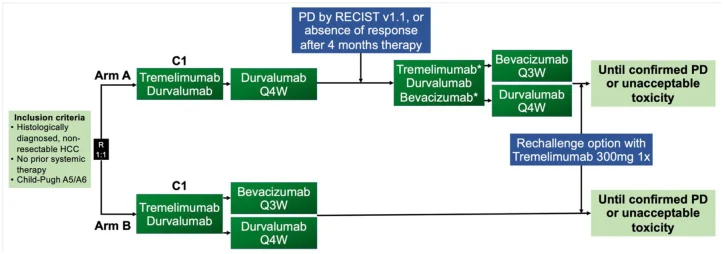
The primary endpoint is overall response rate (ORR), reflecting the proportion of patients achieving tumor shrinkage. Secondary endpoints include overall survival (OS), progression-free survival (PFS), safety, and patient-reported outcomes.
All procedures are conducted in accordance with international ethical standards, including Good Clinical Practice and the Declaration of Helsinki.
Trial Progress and Current Status
The MONTBLANC trial has now successfully completed patient recruitment, marking a critical milestone in its clinical development. Completion of enrollment ensures that the study population is fully accrued, allowing for robust evaluation of the predefined endpoints.
At this stage, no efficacy or survival data have been reported, as the trial is ongoing and patients continue to be followed for response and long-term outcomes. The focus now shifts toward data maturation, analysis, and eventual reporting of results.
Scientific Rationale
The rationale behind the MONTBLANC trial is grounded in the complementary mechanisms of its three therapeutic components.
Durvalumab blocks PD-L1, restoring T-cell–mediated anti-tumor activity. Tremelimumab targets CTLA-4, promoting T-cell priming and expansion. Bevacizumab inhibits VEGF, reducing tumor angiogenesis and improving immune cell infiltration into the tumor microenvironment.
By combining these mechanisms, the trial aims to overcome resistance to immunotherapy and enhance both the depth and durability of response in HCC. This integrated approach reflects a broader trend in oncology toward targeting multiple pathways simultaneously.
With patient recruitment now completed, the MONTBLANC trial enters a critical phase of follow-up and data maturation. The oncology community will closely monitor upcoming results to determine whether this triple immunotherapy strategy can meaningfully improve response rates, survival outcomes, and establish a new treatment approach in advanced hepatocellular carcinoma (HCC).


