Endometrial cancer is the most common gynecologic malignancy in developed countries, with rising incidence driven by aging populations, obesity, and metabolic disorders. Traditionally managed with surgery, radiotherapy, chemotherapy, and hormonal therapy, the disease is now understood as a biologically heterogeneous entity shaped by hormonal imbalance, molecular alterations, and immune dynamics.
Advances in genomic profiling, particularly through The Cancer Genome Atlas (TCGA) and the ProMisE classification, have redefined risk stratification and therapeutic decision-making. In parallel, immunotherapy—especially immune checkpoint inhibitors—has emerged as a transformative treatment option, demonstrating significant clinical benefit in selected patient populations such as those with mismatch repair-deficient (dMMR) or microsatellite instability-high (MSI-H) tumors.
This review provides a comprehensive overview of the epidemiology, hormonal and molecular background, current treatment strategies, and the evolving role of immunotherapy in endometrial cancer, highlighting key clinical data and future directions toward more personalized care.
Title: Does immunotherapy hold great promise in endometrial cancer care?
Authors: Malgorzata Braszka, Hanna Chowaniec, Martyna Borowczyk, Ewa Dwojak, Maria Stępień, Antonina Ślubowska, Magda Mielczarek
Background
Endometrial cancer is the most common gynecologic malignancy in many high-income countries and ranks among the most frequently diagnosed cancers in women worldwide. Its incidence varies substantially across regions, with the highest rates reported in North America and other developed settings, where obesity, longer life expectancy, and metabolic disease contribute strongly to disease burden. The peak incidence occurs between 55 and 59 years of age, and only about 1% to 8% of cases are diagnosed in women younger than 40 years.
The review highlights that endometrial cancer is not only a hormonally driven disease but also a biologically heterogeneous malignancy shaped by molecular subtype, immune microenvironment, obesity-related inflammation, and endocrine signaling. These features are now redefining treatment, especially with the integration of immunotherapy and molecularly guided strategies.
Methods
This article is a narrative review of the current endometrial cancer treatment landscape, with particular focus on immunotherapy. The authors summarize epidemiologic data, hormonal and metabolic drivers of disease, molecular classification systems, currently approved treatments, and emerging therapeutic platforms. The review also examines the tumor microenvironment, including tumour-infiltrating lymphocytes, T-cell exhaustion, tumor-associated macrophages, and regulatory T cells, to explain why immunotherapy works well in some patients but not in others. In addition to approved checkpoint inhibitors, the paper discusses investigational approaches such as cancer vaccines, oncolytic viruses, adoptive cellular therapies, and bispecific antibodies, as well as ongoing international clinical trials.
Study Design
This is not a single clinical trial but a broad state-of-the-art review article. Its structure moves from disease epidemiology and hormonal biology to molecular classification, current treatment standards, approved immune checkpoint inhibitors, and future research directions. The article incorporates evidence from major molecular profiling efforts such as The Cancer Genome Atlas, which evaluated 373 tumors and established four molecular subgroups: POLE ultramutated, microsatellite instability hypermutated, copy number-low, and copy number-high or serous-like disease.
The review also discusses the clinically validated ProMisE classifier, which stratifies tumors into POLE-mutated, mismatch repair-deficient, p53 wild-type, and p53-abnormal categories. Through this framework, the article connects underlying biology with current and future therapeutic decisions.
Results
The review shows that endometrial cancer remains primarily managed with surgery, radiotherapy, chemotherapy, and hormone therapy, but immunotherapy has clearly changed the treatment landscape, especially in advanced and recurrent disease. Surgery remains the global cornerstone of treatment, commonly total hysterectomy with bilateral salpingo-oophorectomy, with lymph node staging depending on risk. Carboplatin plus paclitaxel continues to serve as the chemotherapy backbone, while radiation is used in selected postoperative settings.
A major result of the review is the strong link between hormonal imbalance and tumor development. Oestrogen stimulates endometrial proliferation, while progesterone counteracts this effect. Excess oestrogen exposure, especially in obesity, polycystic ovary syndrome, anovulation, and postmenopausal peripheral aromatization, supports hyperplasia and malignant transformation. The review emphasizes that obesity is not only a metabolic risk factor but also an immunologic one, because adipose tissue promotes insulin resistance, higher IGF-1 activity, inflammatory cytokine production, and further oestrogen synthesis.
The article also details how molecular classification now drives prognosis and treatment. POLE-mutated and dMMR or MSI-H tumors are especially relevant because their hypermutated state often creates an immune-responsive phenotype. By contrast, copy number-high or p53-abnormal tumors are generally more aggressive. The older Bokhman division into Type I and Type II disease is discussed, but the review notes that this classification is now seen as overly simplified compared with modern genomic profiling.
Among immune checkpoint inhibitors, pembrolizumab, dostarlimab, and durvalumab are presented as key approved agents.
Pembrolizumab monotherapy was approved for advanced or metastatic dMMR or MSI-H endometrial cancer after progression on prior systemic therapy, based on KEYNOTE-158. In KEYNOTE-868/NRG-GY018, progression-free survival in the dMMR cohort reached 74% in the pembrolizumab arm compared with 38% in the placebo arm.
Pembrolizumab plus lenvatinib also showed important activity in previously treated advanced disease. In KEYNOTE-146, the overall response rate was 39.8%, median duration of response was 22.9 months, median progression-free survival was 7.4 months, and median overall survival was 17.7 months. In KEYNOTE-775, this combination improved outcomes over chemotherapy in the pMMR population, with median progression-free survival of 6.6 versus 3.8 months and median overall survival of 17.4 versus 12.0 months.
Dostarlimab is another major advance. In the phase 3 RUBY study, dostarlimab plus chemotherapy followed by maintenance dostarlimab reduced the risk of progression or death by 72% in the dMMR or MSI-H population and by 36% in the overall population. Durvalumab is also highlighted as an important agent. In the DUO-E study, adding durvalumab to chemotherapy significantly lowered the risk of progression or death in patients with primary advanced or recurrent endometrial cancer.
The review also covers investigational checkpoint inhibitors. Nivolumab monotherapy showed activity in dMMR, MSI-H, and hypermutated tumors, although around 60% of patients either failed to respond or progressed within 6 months. In a phase 2 trial, nivolumab plus cabozantinib achieved a median progression-free survival of 5.3 months versus 1.9 months with nivolumab alone. Atezolizumab also showed meaningful benefit in the AtTEnd study. In the dMMR subgroup, median progression-free survival was not reached with atezolizumab plus chemotherapy versus 6.9 months with placebo plus chemotherapy, with a hazard ratio of 0.36. In the overall population, median progression-free survival was 10.1 versus 8.9 months.
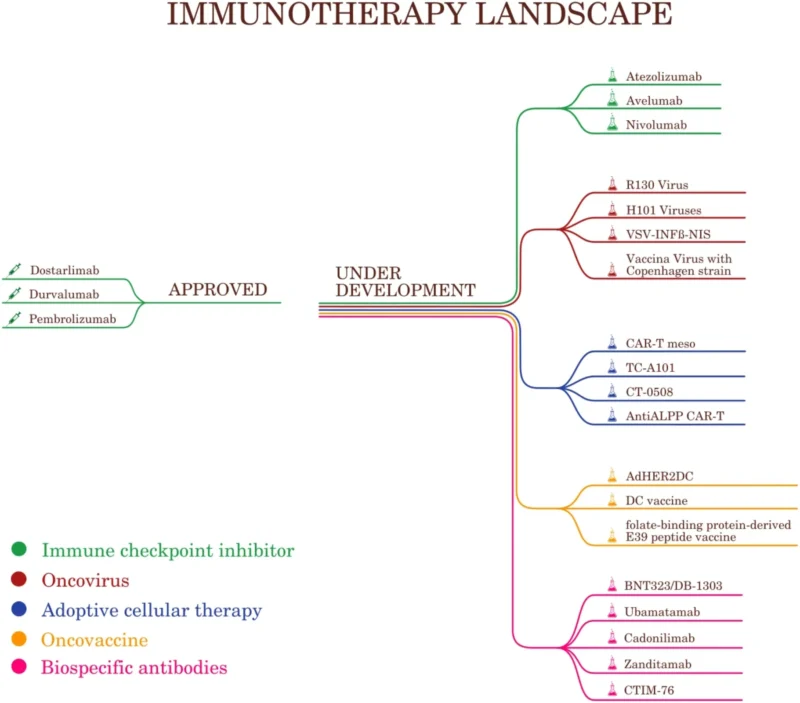
Beyond checkpoint inhibition, the review describes a broad investigational immunotherapy pipeline. Therapeutic vaccines, especially dendritic-cell-based strategies and folate-binding protein peptide vaccines, are being studied for recurrence prevention and disease control. Oncolytic viruses such as vaccinia virus, VSV-IFNβ-NIS, R130 virus, and H101 are under evaluation, although clinical data remain early. Adoptive cellular therapies, including HER2-directed macrophage CAR approaches and ALPP-targeted CAR-T cells, represent another promising area but remain investigational. Bispecific antibodies are also emerging. BNT323/DB-1303, a HER2-targeting agent, received Breakthrough Therapy designation from the FDA in 2023 for advanced endometrial cancer after immune checkpoint inhibitor failure.
The article finally underscores that the tumor microenvironment is central to treatment response. High oestrogen states may upregulate PD-L1, while loss of progesterone receptor signaling, M2-polarized macrophages, regulatory T cells, chronic inflammation, and metabolic dysfunction all promote immune escape. The prognostic role of tumor-infiltrating lymphocytes is complex, with some studies linking CD8-positive infiltration to better outcomes, especially in certain molecular contexts, but not uniformly across all histologies and grades.
Key Findings
This review makes it clear that endometrial cancer should no longer be viewed as a single disease. It is a molecularly diverse malignancy in which hormone signaling, obesity, immune regulation, and genomic subtype all affect prognosis and therapeutic response. Immunotherapy is now an established part of treatment, especially for dMMR and MSI-H disease, and checkpoint inhibitors have moved into earlier-line settings in combination with chemotherapy. Pembrolizumab, dostarlimab, and durvalumab represent the most important regulatory advances, while atezolizumab, nivolumab-based combinations, vaccines, oncolytic viruses, adoptive cell therapies, and bispecific antibodies remain active areas of research.
Another key message is that the future of endometrial cancer therapy is increasingly personalized. Molecular classification systems such as TCGA and ProMisE are no longer purely academic; they are becoming clinically essential. The RAINBO platform further reflects this shift by testing risk-adapted adjuvant therapy across four molecularly defined groups, including p53-abnormal, dMMR, no specific molecular profile, and POLE-mutated disease.
Conclusion
This review presents endometrial cancer as a hormonally influenced, molecularly heterogeneous, and immunologically complex disease that is entering a new era of precision oncology. Traditional modalities such as surgery, radiotherapy, chemotherapy, and hormonal therapy remain fundamental, but they are now being complemented by immunotherapy and biomarker-driven treatment selection. The strongest progress has been seen with checkpoint inhibitors, especially in dMMR and MSI-H populations, where clinically meaningful improvements in progression-free survival and risk reduction have already been demonstrated.
Even so, the article also shows that the future of endometrial cancer care will depend not only on more drugs, but on better patient selection, deeper understanding of the tumor microenvironment, and broader access to clinical trials. Overall, the treatment landscape is moving steadily toward more individualized and biologically informed care.


