CheckMate 743 is a pivotal phase III trial that established nivolumab plus ipilimumab as a first-line standard of care for unresectable malignant pleural mesothelioma, showing a sustained overall survival advantage over platinum–pemetrexed chemotherapy and reshaping treatment for a disease long defined by limited durable options; the newly reported 5-year follow-up confirms that this benefit persists with long-term survivors, durable responses, and no new safety signals, while exploratory biomarker analyses suggest baseline monocytic myeloid-derived suppressor cells (M-MDSCs) may help identify patients less likely to benefit from dual checkpoint blockade.
Background
Unresectable malignant pleural mesothelioma (PM) remains an aggressive thoracic malignancy with historically poor outcomes. For nearly two decades, platinum plus pemetrexed chemotherapy represented the standard first-line treatment, offering median overall survival (OS) of approximately 12–14 months.
The phase III CheckMate 743 trial previously demonstrated a significant survival advantage with first-line nivolumab plus ipilimumab compared with chemotherapy, leading to regulatory approval worldwide. The newly published 5-year follow-up in Journal of Clinical Oncology represents the longest reported follow-up for first-line immunotherapy in pleural mesothelioma and provides critical insight into durability of benefit, long-term safety, biomarker correlations, and treatment-switching–adjusted outcomes.
Methods
CheckMate 743 was a global, open-label, randomized phase III trial enrolling patients with unresectable pleural mesothelioma. Patients were randomized 1:1 to receive:
- Nivolumab 3 mg/kg every 2 weeks plus ipilimumab 1 mg/kg every 6 weeks, or
- Platinum (cisplatin or carboplatin) plus pemetrexed every 3 weeks for up to 6 cycles.The primary
The primary endpoint was overall survival (OS). Secondary endpoints included progression-free survival (PFS), objective response rate (ORR), duration of response (DOR), safety, and exploratory biomarker analyses.
The 5-year update included treatment-switching–adjusted analyses using inverse probability of censoring weighting (IPCW), as 24% of patients in the chemotherapy arm received subsequent immunotherapy.
Median follow-up at database lock (June 14, 2023) was 66.8 months, making this the most mature dataset available in this setting.
Study Design
A total of 605 patients were randomized:
- 303 to nivolumab plus ipilimumab
- 302 to chemotherapy
Baseline characteristics were balanced across arms
Importantly, 24% of patients in the chemotherapy arm received subsequent immunotherapy compared with 6% in the nivolumab/ipilimumab arm.
Exploratory analyses included evaluation of baseline monocytic myeloid-derived suppressor cells (M-MDSCs) as a potential biomarker of resistance.
Results
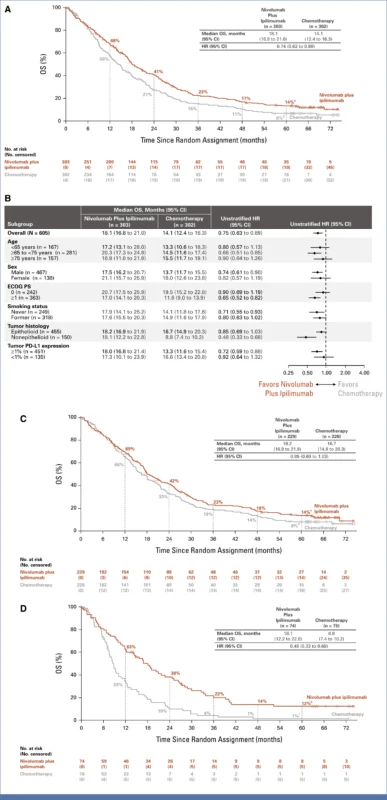
At a median follow-up of 66.8 months, nivolumab plus ipilimumab continued to demonstrate a statistically and clinically meaningful overall survival benefit compared with platinum–pemetrexed chemotherapy. The hazard ratio for overall survival was 0.74 (95% CI, 0.62–0.88), confirming a 26% relative reduction in the risk of death. The 5-year overall survival rate was 14% in the immunotherapy arm compared with 6% in the chemotherapy arm, representing more than a doubling of long-term survival.
When stratified by histology, important differences emerged. Among patients with epithelioid pleural mesothelioma, the 5-year overall survival rate was 14% with nivolumab plus ipilimumab versus 8% with chemotherapy, with a hazard ratio of 0.85 (95% CI, 0.69–1.03). In contrast, patients with nonepithelioid histology derived a markedly greater benefit from dual immune checkpoint blockade. In this subgroup, the 5-year overall survival rate was 12% with nivolumab plus ipilimumab compared with only 1% with chemotherapy, corresponding to a hazard ratio of 0.48 (95% CI, 0.33–0.68). This finding reinforces the particular clinical value of immunotherapy in a subgroup historically associated with poor outcomes and limited chemotherapy responsiveness.
Progression-free survival differences were less pronounced. The hazard ratio for progression-free survival in the overall population was 0.93 (95% CI, 0.77–1.13). However, at 5 years, 8% of patients treated with nivolumab plus ipilimumab remained progression-free, whereas no patients in the chemotherapy arm were progression-free at that time point, highlighting the durability of disease control achieved with immunotherapy.
Objective response rates were 39% in the nivolumab plus ipilimumab arm and 44% in the chemotherapy arm. Although the initial response rate was numerically lower with immunotherapy, duration of response strongly favored nivolumab plus ipilimumab. Among responders, 17% of patients in the immunotherapy group maintained ongoing responses at 5 years, while none of the chemotherapy responders had ongoing responses at that landmark. This durable tail of response represents a key distinction between immune checkpoint blockade and cytotoxic chemotherapy in mesothelioma.
Because 24% of patients in the chemotherapy arm subsequently received immunotherapy, a treatment-switching–adjusted analysis was conducted using inverse probability of censoring weighting. After adjustment, the median overall survival in the chemotherapy arm decreased to 12.1 months, and the hazard ratio comparing nivolumab plus ipilimumab with chemotherapy improved to 0.64 (95% CI, 0.53–0.78), indicating that crossover likely diluted the magnitude of survival benefit observed in the intention-to-treat analysis.
Exploratory biomarker analyses showed that in the M-MDSC–evaluable population, nivolumab plus ipilimumab improved overall survival compared with chemotherapy, with median overall survival of 19.6 months versus 14.9 months and a hazard ratio of 0.72 (95% CI, 0.59–0.88). Higher baseline monocytic myeloid-derived suppressor cell levels were associated with worse outcomes in patients treated with immunotherapy, with a hazard ratio for overall survival of 1.25 (95% CI, 1.09–1.43) per log increase, suggesting a potential resistance signal that warrants prospective validation.
No new safety signals were observed at 5 years, and the long-term safety profile remained consistent with previous reports, supporting the sustained tolerability of dual checkpoint blockade in this population.
Key Findings
- Nivolumab plus ipilimumab continues to demonstrate durable survival benefit at 5 years in unresectable pleural mesothelioma.
- Five-year survival more than doubled compared with chemotherapy (14% vs 6%).
- The greatest magnitude of benefit was observed in nonepithelioid mesothelioma (HR 0.48).
- Durability of response clearly favors immunotherapy, with 17% of responders maintaining response at 5 years.
- Treatment-switching–adjusted analyses strengthened the survival benefit (HR 0.64).
- High baseline M-MDSC levels may predict reduced benefit from dual checkpoint blockade.
- No new long-term safety concerns were identified.
Conclusion
The 5-year update from CheckMate 743 confirms nivolumab plus ipilimumab as a durable, long-term standard of care for first-line treatment of unresectable pleural mesothelioma. Survival benefit remains preserved across histologic subtypes and is particularly meaningful in nonepithelioid disease, a subgroup historically associated with poor outcomes.
Importantly, the magnitude of benefit increases after adjusting for treatment switching, strengthening confidence in dual checkpoint inhibition as the preferred first-line strategy.With 14% of patients alive at 5 years and durable responses maintained in a subset, this study establishes immunotherapy as a transformative advance in pleural mesothelioma and represents the longest follow-up dataset available in this setting to date.


