AXINET Trial (GETNE-1107) is an international, randomized, double-blind, placebo-controlled phase III trial that tested whether adding the VEGFR inhibitor axitinib to long-acting octreotide (LAR) could improve outcomes for patients with unresectable or metastatic, grade 1–2 extrapancreatic neuroendocrine tumors (epNETs) after progression.
Built on the strong biologic rationale that epNETs are highly vascular and angiogenesis-driven, the study compared axitinib plus octreotide LAR versus placebo plus octreotide LAR, aiming to extend disease control while maintaining manageable toxicity in a population where treatment options have historically been limited and highly dependent on tumor site and tempo.
Background
Extrapancreatic neuroendocrine tumors (epNETs) are typically well-differentiated (grade 1–2) malignancies that often grow slowly but can be difficult to control once metastatic. A defining biologic feature of many NETs is high vascularity, supported by pro-angiogenic signaling (including VEGF/VEGFR pathways). This created strong rationale to test anti-angiogenic tyrosine kinase inhibitors (TKIs) in epNETs, especially because historical systemic options were limited when this study was planned.
AXINET (GETNE-1107) evaluated whether adding axitinib—a selective VEGFR-1/2/3 inhibitor—to octreotide long-acting release (LAR) could improve tumor control and clinical outcomes in patients with advanced grade 1–2 epNETs.
Methods
AXINET was an international, randomized, double-blind, placebo-controlled phase II/III trial conducted across 27 hospitals in Spain, Italy, and Germany and sponsored by GETNE. Adults (≥18 years) with unresectable locally advanced or metastatic, histologically confirmed grade 1–2 epNETs (Ki-67 ≤20%) were eligible if they had progressive disease within 12 months prior to enrollment and measurable disease by RECIST 1.1. Patients could have received up to two prior systemic treatment lines, with prior somatostatin analogs (SSAs) allowed, but prior angiogenesis inhibitors were excluded. Key exclusions included uncontrolled hypertension, serious cardiovascular comorbidities, intracranial metastases, and factors limiting protocol compliance or interpretation.
Patients underwent regular clinical and laboratory monitoring every 4 weeks, and imaging (CT or MRI) every 12 weeks until progression or initiation of another anticancer therapy. Tumor response and progression were assessed locally by investigators using RECIST 1.1, and imaging was also evaluated retrospectively by a blinded independent central review (BICR) in most patients.
Study Design
Patients were randomized 1:1 to receive:
- Axitinib 5 mg orally twice daily + octreotide LAR 30 mg intramuscularly every 28 days, or
- Placebo orally twice daily + octreotide LAR 30 mg every 28 days
Treatment continued until disease progression, unacceptable toxicity, or withdrawal of consent. Dose interruptions and reductions were permitted (down to 2 mg twice daily). Crossover was not allowed.
Randomization was stratified by:
- Primary tumor site (GI vs other)
- Ki-67 index (≤5% vs >5%)
- Time from diagnosis to trial inclusion (≤12 months vs >12 months).
The primary endpoint was investigator-assessed progression-free survival (PFS). Secondary endpoints included BICR-assessed PFS, time-to-progression, objective response rate (ORR), duration of response, biochemical response (CgA/5-HIAA in those elevated at baseline), overall survival (OS), and safety.
The phase III component was powered to detect a target hazard ratio (HR) of 0.60 for investigator-assessed PFS, but actual control-arm PFS proved longer than anticipated, which the authors discuss as a factor that likely reduced power.
Results of AXINET Trial
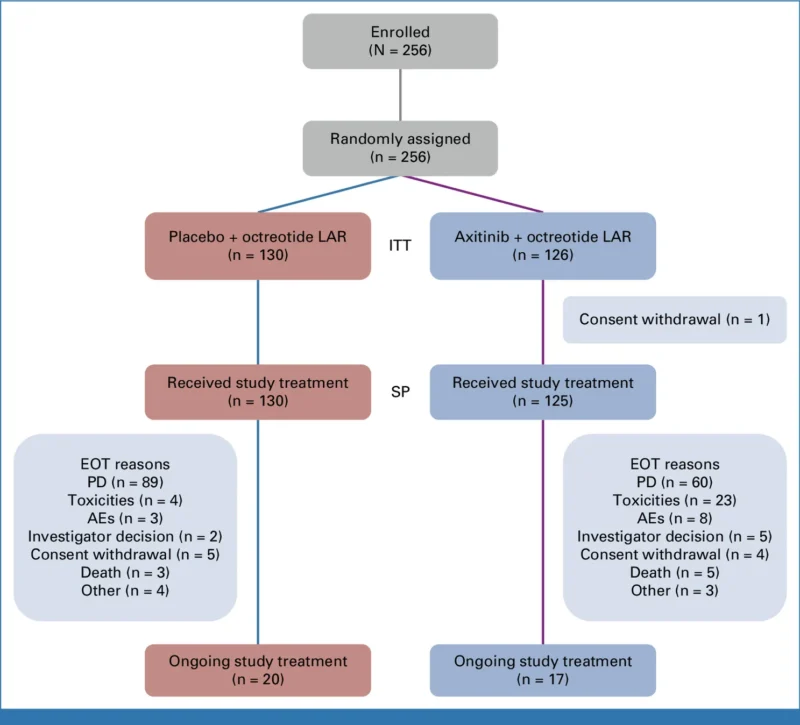
Between October 2011 and May 2019, 256 patients were randomized: 126 to axitinib and 130 to placebo (intention-to-treat population). One patient in the axitinib arm withdrew consent before receiving treatment and was excluded from safety analyses.
Key disease features reflected a typical epNET population: most tumors were grade 2 (69.1%), and the most common primaries were small intestine (49.6%), lung (28.1%), and colorectal (9.4%). Liver metastases were present in ~88% of patients (111/126 axitinib; 114/130 placebo). Importantly, 44.9% were treatment-naïve, with the remainder having received one or more prior systemic lines.
Efficacy: investigator vs central review
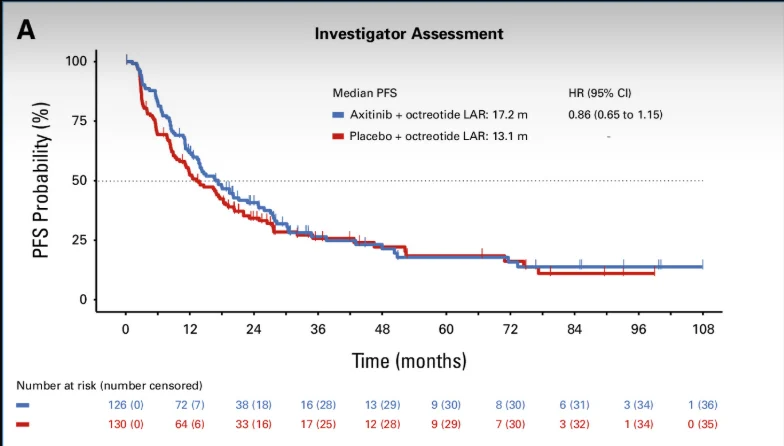
At the July 2020 cutoff, investigator-assessed PFS events occurred in 70.6% (axitinib) vs 73.1% (placebo). Median investigator-assessed PFS was:
- 17.2 months with axitinib (95% CI, 13.6–24.7)
- 13.1 months with placebo (95% CI, 10.9–18.6)
This corresponded to HR 0.86 (95% CI, 0.65–1.15; P = .324), meaning the primary endpoint was not met.
However, central imaging review told a different story. BICR assessment was available in 243/256 patients (94.9%). With BICR, PFS events occurred in 72.2% (axitinib) vs 81.5% (placebo), and median BICR-assessed PFS was:
- 16.6 months with axitinib (95% CI, 13.5–24.2)
- 9.9 months with placebo (95% CI, 8.2–13.9)
This yielded HR 0.71 (95% CI, 0.54–0.94; P = .017), demonstrating a statistically significant improvement in PFS by central blinded review.
The authors highlight that discrepancies between investigator and central reads are not unusual in NET trials, especially with extensive liver involvement. Hypervascular liver metastases can be sensitive to contrast timing, and anti-VEGF agents may cause necrosis/cystic change without major size reduction—features not perfectly captured by RECIST and potentially interpreted differently by local vs blinded central reviewers.
Tumor response
Axitinib produced higher response rates by both assessments:
- Investigator-assessed ORR: 17.5% vs 4.6% (P = .001)
- BICR-assessed ORR: 12.8% vs 3.2% (P = .005)
By investigator assessment, complete responses were rare but occurred in both groups (1.6% axitinib vs 0.8% placebo), while partial responses were more frequent with axitinib (15.9% vs 3.8%). Any tumor shrinkage was reported in 64.3% of axitinib-treated patients vs 46.2% with placebo.
Biochemical response (CgA and 5-HIAA among those elevated at baseline) did not significantly differ between arms.
Overall survival
OS was not mature at the primary endpoint cutoff (reported 116 deaths; 45.3% events), and follow-up was ongoing, so AXINET does not provide definitive OS conclusions.
Key findings
- AXINET enrolled 256 patients with advanced grade 1–2 epNETs (most with liver metastases ~88%).
- The primary endpoint (investigator-assessed PFS) was not met despite a numerical median PFS difference (17.2 vs 13.1 months; HR 0.86; P = .324).
- Blinded independent central review showed a significant PFS benefit (16.6 vs 9.9 months; HR 0.71; P = .017).
- Objective response rates were consistently higher with axitinib (17.5% vs 4.6% by investigators; 12.8% vs 3.2% by BICR).
- Toxicities reflected expected VEGFR inhibition effects, led by hypertension and diarrhea.
Conclusion
AXINET (GETNE-1107) tested a biologically rational strategy—VEGFR-targeted inhibition with axitinib layered onto octreotide LAR—in a large, randomized, double-blind epNET population accrued over many years. While investigator-assessed PFS did not reach statistical significance, blinded independent central review demonstrated a significant PFS improvement, and response rates were meaningfully higher with axitinib by both investigator and central assessment.
The toxicity profile was predictable and manageable, dominated by hypertension and gastrointestinal effects typical of VEGFR blockade, with higher rates of treatment interruptions and dose reductions than placebo. Overall, AXINET adds important evidence that anti-angiogenic TKIs can provide clinically relevant disease control in extrapancreatic NETs, while underscoring why central radiology review and careful interpretation of imaging endpoints are especially critical in this setting.


