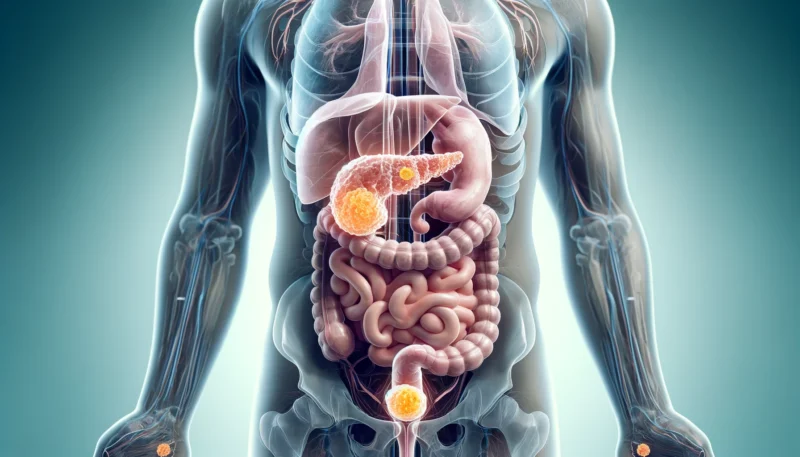
Neuroendocrine tumors (NETs) are a rare type of cancer that arises from specialized cells of the neuroendocrine system. These cells are found throughout the body and help regulate hormones, making NETs unique compared to other types of cancer. NETs can develop in different organs, but the most common sites include the lungs, pancreas, and gastrointestinal (GI) tract .
The Science of Neuroendocrine Tumors: What Causes These Rare Tumors?
Neuroendocrine tumors (NETs) are rare and complex cancers that originate from neuroendocrine cells, which have both nerve-like and hormone-producing properties. These tumors can develop in various organs, including the gastrointestinal tract, pancreas, lungs, and even the skin (as in Merkel cell carcinoma). The exact cause of NETs is not fully understood, but research suggests a combination of genetic, environmental, and hormonal factors contribute to their development.
The Role of Neuroendocrine Cells
Neuroendocrine cells are responsible for producing hormones and regulating bodily functions such as digestion, metabolism, and stress responses. When these cells undergo uncontrolled growth due to genetic mutations or environmental triggers, they can form tumors. Unlike many other cancers, NETs can be either slow-growing (indolent) or highly aggressive, making their behavior difficult to predict.
Genetic Mutations and Hereditary Risk
While most neuroendocrine tumors (NETs) arise sporadically, a subset of cases is associated with inherited genetic syndromes. These hereditary conditions significantly increase the lifetime risk of developing NETs and often present with tumors in multiple organ systems.
One of the most well-known syndromes is Multiple Endocrine Neoplasia Type 1 (MEN1), which is caused by a mutation in the MEN1 gene. This disorder predisposes individuals to tumors in endocrine glands, particularly the pancreas, pituitary, and parathyroid glands.Another condition, Von Hippel-Lindau Syndrome (VHL), involves mutations in the VHL gene and is linked to the development of pancreatic NETs and pheochromocytomas—neuroendocrine tumors of the adrenal glands.Tuberous Sclerosis Complex (TSC) is a genetic disorder characterized by the growth of benign tumors in multiple organs, including the brain, kidneys, and heart, and it also raises the risk for NETs in the pancreas and other sites.Lastly, Neurofibromatosis Type 1 (NF1) is a condition marked by nerve sheath tumors, skin changes, and bone abnormalities, and it has been associated with gastrointestinal NETs, especially in the small intestine and duodenum.
Environmental and Lifestyle Factors
While no direct lifestyle cause of NETs has been identified, some risk factors may contribute:
- Chronic inflammation: Long-term inflammation in the digestive system may increase the likelihood of developing gastrointestinal NETs.
- Exposure to toxins or radiation: Although not conclusively proven, some studies suggest that exposure to certain chemicals or radiation could trigger NET formation.
- Smoking and alcohol consumption: These are known risk factors for many cancers, including lung and pancreatic NETs.
Hormonal and Metabolic Influences
NETs are unique because they can actively produce hormones, leading to conditions such as carcinoid syndrome (flushing, diarrhea, and heart problems). Some researchers believe excessive hormonal stimulation may contribute to tumor growth, but this theory is still under investigation.
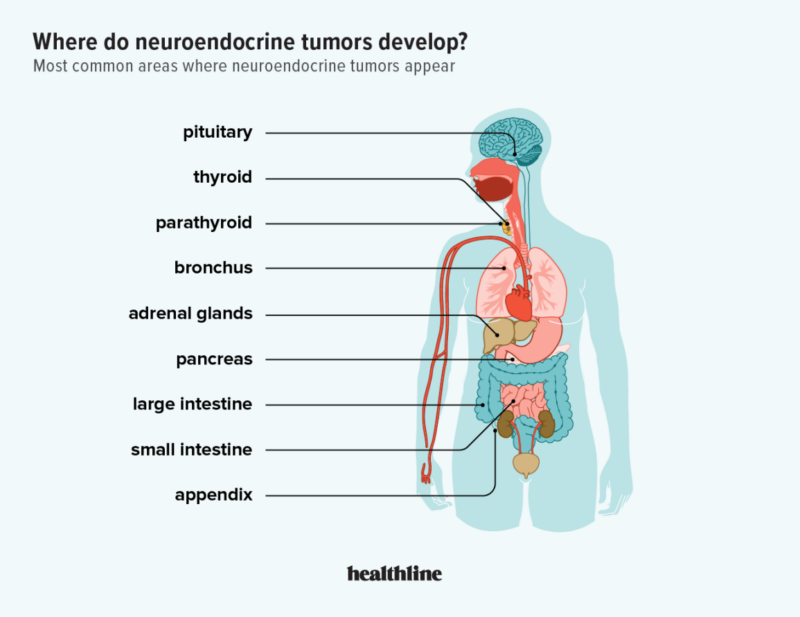
Types of Neuroendocrine Tumors
NETs are classified based on their location and their ability to produce hormones. The main types include:
Gastroenteropancreatic Neuroendocrine Tumors
Gastroenteropancreatic neuroendocrine tumors (GEP-NETs) are a diverse group of tumors that originate in the neuroendocrine cells of the gastrointestinal tract and pancreas. These tumors can vary significantly in behavior, location, and hormone production.
One subtype is pancreatic neuroendocrine tumors (pNETs), which may be classified as either functioning or non-functioning. Functioning pNETs secrete hormones such as insulin, gastrin, or glucagon, leading to distinct clinical syndromes, while non-functioning pNETs do not produce hormones at clinically detectable levels and often present later with mass-related symptoms.
Another major category includes carcinoid tumors, which are typically found in the small intestine, appendix, and rectum. These tumors often produce excess serotonin, leading to carcinoid syndrome in some cases—marked by flushing, diarrhea, and wheezing. Despite their relatively slow-growing nature, some GEP-NETs can metastasize and require long-term management.
Lung Neuroendocrine Tumors
Lung neuroendocrine tumors (NETs) represent a spectrum of malignancies that arise from neuroendocrine cells within the lungs. These include both indolent and highly aggressive subtypes.
Typical and atypical carcinoid tumors are considered the slower-growing forms of lung NETs. While generally less aggressive, they can occasionally secrete hormones such as serotonin or ACTH, leading to distinct paraneoplastic syndromes.In contrast, large-cell neuroendocrine carcinoma (LCNEC) is a high-grade tumor with a more aggressive clinical course. It tends to grow and spread more rapidly than typical carcinoids and often requires systemic chemotherapy similar to regimens used for other high-grade neuroendocrine cancers.
The most aggressive type in this group is small-cell lung cancer (SCLC). This form of lung NET grows quickly, metastasizes early, and is typically treated with a combination of chemotherapy and radiation. Despite initial responsiveness to treatment, recurrence is common and prognosis remains poor.
Other Rare NETs
Beyond the lungs, rare NETs can also arise in other locations. Thymic neuroendocrine tumors occur in the thymus gland, located in the upper chest, and are often aggressive, though uncommon. Adrenal NETs, including pheochromocytomas and paragangliomas, originate in or near the adrenal glands. These tumors can secrete excess catecholamines such as adrenaline, leading to symptoms like high blood pressure, palpitations, and sweating.
Symptoms of Neuroendocrine Tumors
NETs can be difficult to diagnose because they often grow slowly and cause nonspecific symptoms. The symptoms depend on the tumor’s location and whether it is producing hormones.
General Symptoms of NETs
- Fatigue
- Unexplained weight loss
- Abdominal pain or discomfort
- Flushing (redness of the skin, especially on the face and neck)
- Diarrhea
Hormone-Related Symptoms
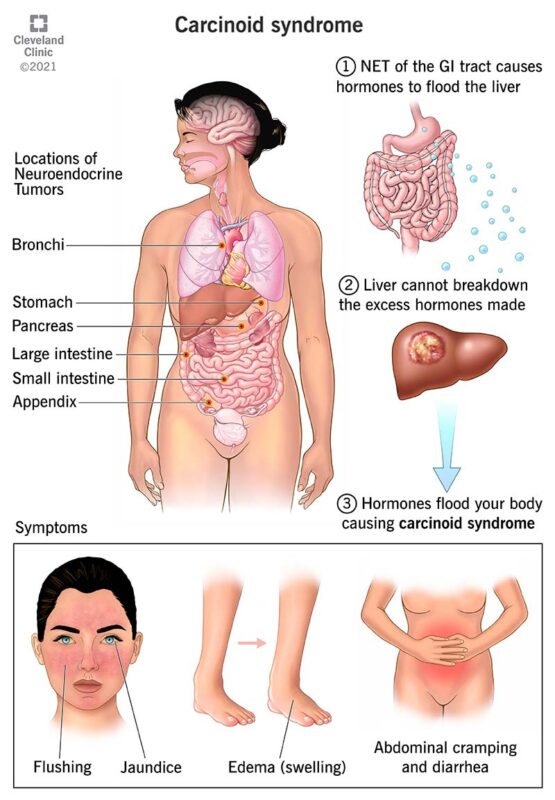
Functioning neuroendocrine tumors (NETs) are a subtype that produce excess hormones, leading to distinct clinical syndromes that can significantly affect quality of life and complicate diagnosis.
One of the most recognized is carcinoid syndrome, which results from elevated levels of serotonin. Patients with this syndrome often experience facial flushing, persistent diarrhea, wheezing, and in some cases, damage to heart valves—especially on the right side of the heart.
Another example is insulinoma, a type of pancreatic NET that secretes insulin inappropriately. This overproduction causes episodes of dangerously low blood sugar, or hypoglycemia, which can lead to confusion, fainting, and even seizures if left untreated.Gastrinoma, another hormone-producing NET, leads to excessive secretion of stomach acid. This results in recurrent peptic ulcers, abdominal pain, and acid reflux, a condition known clinically as Zollinger-Ellison syndrome.
Because the symptoms of functioning NETs often mimic those of more common gastrointestinal or endocrine disorders, they are frequently misdiagnosed. Many patients endure years of unresolved symptoms before receiving an accurate diagnosis and appropriate treatment.
Prognosis: What to Expect with NETs?
The prognosis for neuroendocrine tumors (NETs) depends on a variety of clinical and biological factors, with outcomes ranging from favorable in indolent cases to poor in aggressive forms.
One of the most important prognostic indicators is tumor grade. Low-grade NETs, which are slow-growing and well-differentiated, typically have a more favorable long-term outlook. In contrast, high-grade tumors such as poorly differentiated neuroendocrine carcinomas are more aggressive, associated with rapid progression and reduced survival.The stage at diagnosis also plays a crucial role. Patients diagnosed at an early stage, when the tumor is localized and potentially resectable, generally experience better survival outcomes. However, once the disease has metastasized—particularly to the liver or bone—the prognosis becomes more guarded.
Tumor location further influences prognosis. Pancreatic and gastrointestinal NETs often respond better to treatment and have longer survival rates, especially when diagnosed early. On the other hand, neuroendocrine tumors arising in the lungs, particularly large-cell and small-cell subtypes, tend to behave more aggressively and are associated with poorer outcomes.
Encouragingly, recent research shows that overall survival rates for NET patients have improved over the past decade. This is largely due to advances in imaging, earlier detection, and the growing availability of targeted therapies, including somatostatin analogs, PRRT, and novel systemic agents.(National Caner Institute 2024).
Diagnosis of Neuroendocrine Tumors
Neuroendocrine tumors (NETs) are often challenging to diagnose due to their slow growth and nonspecific symptoms. Many patients go undiagnosed for years, leading to delayed treatment. A combination of imaging tests, blood markers, and tissue biopsies is essential for an accurate diagnosis.
Initial Evaluation and Clinical Symptoms
Since NETs can develop in various organs (e.g., pancreas, intestines, lungs), symptoms vary widely. Physicians may suspect NETs if a patient experiences unexplained weight loss, flushing, persistent diarrhea, abdominal pain, or hormone-related symptoms like insulinoma (low blood sugar).
Imaging Tests for NET Detection
Imaging plays a crucial role in detecting and staging NETs:
- Computed Tomography (CT) Scan: Helps identify tumors and check for metastases.
- Magnetic Resonance Imaging (MRI): Especially useful for detecting liver metastases.
- Somatostatin Receptor Imaging (SRI): Uses radiolabeled tracers to detect somatostatin receptors on tumor cells.
- Gallium-68 DOTATATE PET/CT: The most sensitive test for detecting well-differentiated NETs.
- Octreotide Scintigraphy (OctreoScan): Less sensitive than Gallium-68 PET but still used in some cases.
- 18F-FDG PET Scan: Used for poorly differentiated, fast-growing NET.
Blood and Urine Biomarkers
Certain biochemical markers play a crucial role in the detection and monitoring of neuroendocrine tumors (NETs), though their specificity and usefulness can vary depending on the tumor type and activity.
Chromogranin A (CgA) is one of the most widely used general biomarkers for NETs. It is secreted by neuroendocrine cells and is often elevated in patients with well-differentiated tumors. However, elevated CgA levels are not exclusive to NETs and can also be seen in other conditions such as chronic atrophic gastritis, proton pump inhibitor use, and renal impairment, limiting its diagnostic specificity.
5-Hydroxyindoleacetic acid (5-HIAA) is the primary metabolite of serotonin and is measured in a 24-hour urine collection. It is particularly useful for diagnosing serotonin-producing carcinoid tumors, especially those of the midgut, and can help confirm the presence of carcinoid syndrome.
Neuron-specific enolase (NSE) may be elevated in more aggressive or poorly differentiated neuroendocrine carcinomas. While it is not typically used for initial diagnosis, it can be helpful in monitoring disease progression in high-grade NETs.
For pancreatic NETs (pNETs), specific hormone markers are used depending on the functional subtype. Insulin levels are elevated in insulinomas, often presenting with symptoms of hypoglycemia. Gastrin is markedly increased in patients with gastrinomas, contributing to acid overproduction and recurrent peptic ulcers. Glucagon, another pancreatic hormone, is elevated in glucagonomas and is associated with characteristic skin rashes, weight loss, and hyperglycemia.
Biopsy and Histopathology
Biopsy and histopathological analysis are essential steps in confirming the diagnosis of neuroendocrine tumors (NETs) and determining their grade. The type of biopsy performed depends on the tumor’s location and accessibility. Fine needle aspiration (FNA) is often used for lesions in the pancreas or lung, where a thin needle is guided by imaging to collect cells for examination. In many cases, a core needle biopsy is preferred because it provides a larger tissue sample, which is more suitable for accurately assessing tumor architecture and conducting grading.
One of the most important histologic tools in NET evaluation is the Ki-67 index. This index measures the percentage of tumor cells that are actively dividing, offering a quantifiable assessment of how quickly the tumor is growing. A low Ki-67 index indicates a slow-growing, well-differentiated tumor, while a high Ki-67 suggests a more aggressive, high-grade neuroendocrine carcinoma. This grading directly influences treatment decisions and prognosis, making biopsy and histopathology central to the clinical management of NETs.
Molecular and Genetic Testing
Molecular and genetic testing has become an essential tool in the personalized management of neuroendocrine tumors (NETs), particularly when considering prognosis, familial syndromes, and targeted therapies. For example, mutations in the MEN1 gene are commonly found in patients with multiple endocrine neoplasia type 1, a hereditary condition associated with a high risk of developing NETs in the pancreas, pituitary, and parathyroid glands.
In pancreatic NETs specifically, mutations in the DAXX and ATRX genes have been identified as prognostic markers. These mutations are often associated with better-differentiated tumors and may predict a more favorable clinical outcome compared to tumors lacking these alterations.
Another increasingly used method is next-generation sequencing (NGS), which analyzes multiple cancer-related genes simultaneously. NGS can detect actionable mutations that may make patients eligible for clinical trials or targeted therapies, helping clinicians tailor treatment plans based on the molecular profile of each tumor.
How Are NETs Treated?
Neuroendocrine tumors (NETs) are a diverse group of cancers that require a personalized treatment approach. The choice of therapy depends on the tumor’s location, grade (slow- or fast-growing), whether it produces hormones, and whether it has spread to other organs. Advances in treatment have significantly improved patient outcomes, with many therapies targeting both the cancer itself and its hormone-related symptoms .
Below is a detailed overview of the current treatment options for NETs, including traditional methods and emerging therapies from recent studies.
Surgery
Surgical removal remains the gold standard for localized NETs, offering the best chance for a cure. The type of surgery depends on the tumor’s location:
- Gastrointestinal (GI) NETs: Resection of the affected bowel, sometimes including lymph nodes.
- Pancreatic NETs: Enucleation (removal of just the tumor) or pancreatic resection (removal of part of the pancreas).
- Lung NETs: Lobectomy (removal of a lung lobe) or segmentectomy (removal of a portion of the lung).
Even in cases where NETs have spread, debulking surgery (removing as much of the tumor as possible) may improve symptoms and prolong survival.
Somatostatin Analogs
Somatostatin analogs (SSAs) such as octreotide and lanreotide are essential in managing neuroendocrine tumors (NETs), particularly for alleviating symptoms associated with hormone hypersecretion and potentially inhibiting tumor progression.
Octreotide, commercially known as Sandostatin, is effective in treating carcinoid syndrome by reducing symptoms like flushing and diarrhea. Additionally, studies suggest that octreotide may have antiproliferative effects, contributing to prolonged progression-free survival in patients with well-differentiated NETs. Pub Med.
Lanreotide, marketed as Somatuline Depot, has demonstrated efficacy in improving progression-free survival among patients with advanced gastroenteropancreatic NETs.The CLARINET study reported that lanreotide significantly prolonged progression-free survival compared to placebo. New England Journal of Medicine, 2014.
Both SSAs are generally well-tolerated, with common side effects including gastrointestinal disturbances and injection site reactions. Their role in symptom management and potential tumor growth inhibition makes them integral components of NET treatment strategies.
Targeted Therapy
Unlike traditional chemotherapy, targeted therapies work by interfering with specific molecules involved in cancer growth. Two key drugs approved for NETs are:
- Everolimus (Afinitor): Blocks the mTOR pathway, reducing tumor cell growth. Approved for pancreatic, lung, and gastrointestinal NETs.
- Sunitinib (Sutent): Inhibits tumor blood vessel formation (angiogenesis), mainly used for pancreatic NETs.
These drugs have shown significant improvement in survival rates, especially in patients with inoperable or metastatic NETs .
Peptide Receptor Radionuclide Therapy
One of the most promising therapies for NETs is Peptide Receptor Radionuclide Peptide Receptor Radionuclide Therapy, or PRRT, is an advanced treatment that delivers targeted radiation to cancer cells—specifically designed for patients with certain neuroendocrine tumors (NETs). This therapy uses a small protein (peptide) to find cancer cells and deliver a radioactive dose directly to them.
PRRT involves injecting a peptide that mimics natural hormones. This peptide is linked to a radioactive compound such as Lutetium-177. When the treatment is given, it travels through your bloodstream and attaches only to tumor cells that have specific receptors (usually somatostatin receptors). Once attached, the radioactive element releases a controlled dose of radiation that kills the cancer cells—while leaving most healthy cells unharmed.The most common PRRT drug is Lutathera® (177Lu-DOTATATE). This was approved by the FDA in 2018 to treat patients with gastroenteropancreatic neuroendocrine tumors (GEP-NETs) that are positive for somatostatin receptors.PRRT may be recommended if your neuroendocrine tumor is advanced, has spread, or is not responding to other treatments. PRRT is typically given through an IV every 8 weeks for a total of 4 cycles. Most patients tolerate it well. Common side effects include nausea, fatigue, and mild changes in blood counts, which are usually temporary.
Chemotherapy
Chemotherapy plays a limited role in the treatment of slow-growing, well-differentiated neuroendocrine tumors (NETs), but it remains a cornerstone in managing high-grade neuroendocrine carcinomas, which are aggressive and poorly differentiated. These tumors often require systemic treatment from the outset due to their rapid progression and high metastatic potential.
One of the most commonly used chemotherapy regimens for pancreatic NETs is the combination of capecitabine and temozolomide (CAPTEM). This regimen has shown promising results, with studies reporting notable objective response rates and prolonged progression-free survival, particularly in patients with well-differentiated pancreatic NETs.In contrast, for high-grade or poorly differentiated tumors, especially those resembling small cell carcinoma, platinum-based chemotherapy regimens—such as cisplatin or carboplatin combined with etoposide—are often used as first-line treatment. These regimens have demonstrated response rates as high as 70% in certain high-grade neuroendocrine carcinomas, making them a critical therapeutic option despite their higher toxicity profile.
While chemotherapy typically involves more side effects than targeted therapies or somatostatin analogs, it continues to serve an essential role in the management of aggressive and rapidly progressing NETs, particularly when other treatments are ineffective or not feasible. PubMed Central.
Immunotherapy
Immunotherapy, a treatment approach that harnesses the body’s immune system to identify and destroy cancer cells, is still an emerging strategy in the management of neuroendocrine tumors (NETs). While its success has been transformative in other cancer types, the role of immunotherapy in NETs remains under investigation and is primarily being evaluated through ongoing clinical trials.
One area of research focuses on immune checkpoint inhibitors, such as pembrolizumab and nivolumab, which target PD-1 or PD-L1 proteins. These proteins act as “brakes” on immune cells, and by blocking them, checkpoint inhibitors aim to unleash the immune system’s ability to recognize and attack tumor cells. Although initial studies have shown modest activity in NETs, certain subgroups—particularly high-grade or PD-L1–positive tumors—may benefit more significantly.
Another promising avenue is the development of combination therapies, where immunotherapy is paired with other treatments such as peptide receptor radionuclide therapy (PRRT) or targeted therapies. These strategies aim to boost the immune response by simultaneously damaging tumor cells and increasing their visibility to immune surveillance.As research continues, immunotherapy may become a more prominent option in select NET populations, particularly when integrated with other modalities to enhance effectiveness.Early results are promising, especially for high-grade NETs, but further research is needed .
Liver-Directed Therapies
Since many NETs spread to the liver, specialized treatments focus on reducing tumor burden in the liver:
- Hepatic Artery Embolization (HAE): Cuts off blood supply to liver tumors.
- Radioembolization (Y-90 Microspheres): Delivers radiation directly to liver tumors.
- Liver Transplantation: Considered for select patients with liver-only NET metastases.
These therapies can significantly reduce symptoms and prolong survival, especially when surgery is not an option.
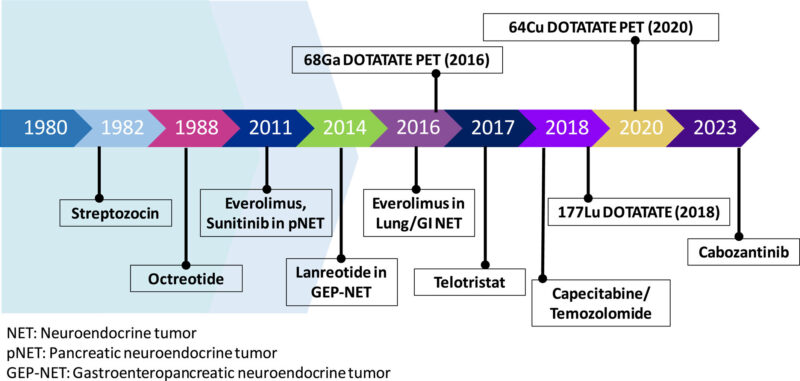
Emerging Therapies and Clinical Trials
Advancements in neuroendocrine tumor (NET) treatment are continually evolving, with several novel approaches showing promise in improving patient outcomes. Here are some of the key developments:
Targeted Therapies for NETs
Targeted therapies aim to attack specific molecules involved in tumor growth, and they have become increasingly important in the treatment of NETs. One promising agent is cabozantinib, a multi-targeted tyrosine kinase inhibitor. According to findings presented at the ESMO Congress 2023, cabozantinib improved progression-free survival (PFS) in patients with both pancreatic and non-pancreatic NETs. This suggests its potential role as a new treatment for NETs, particularly for those whose tumors are resistant to traditional therapies .

You Can Read More About Cabozantinib on Oncodaily
- Lurbinectedin + Irinotecan for GEP-NECs-A phase I/II study presented at the ESMO Sarcoma and Rare Cancers Congress 2024 examined the combination of lurbinectedin and irinotecan for patients with poorly differentiated gastroenteropancreatic neuroendocrine carcinomas (GEP-NECs). The results indicated that this combination achieved disease stabilization and partial response in some patients, with an improved median progression-free survival (PFS) compared to previous therapies. Lurbinectedin, a drug already approved for small-cell lung cancer, works by inhibiting transcription factors critical for tumor growth. The synergistic effect with irinotecan, which acts on different cellular processes, makes this regimen a potential option for refractory GEP-NECs .
- Peptide Receptor Radionuclide Therapy (PRRT)-Another promising approach for treating NETs, especially those of the gastrointestinal tract, is peptide receptor radionuclide therapy (PRRT). This treatment involves the use of radioactive molecules attached to peptides that specifically target neuroendocrine tumor cells, delivering localized radiation to kill cancer cells. PRRT has gained attention for its ability to improve progression-free survival in patients with advanced, well-differentiated NETs .
Immunotherapy in NETs
The role of immunotherapy in treating NETs is also under investigation. While still in the early stages, clinical trials are evaluating immune checkpoint inhibitors, such as nivolumab and pembrolizumab, to determine their effectiveness in combination with other treatments. Early results show some promise in enhancing the body’s immune response against NET cells, particularly in patients with high tumor mutational burden.
- Tumor Microenvironment Targeting-A newer area of interest is tumor microenvironment (TME)-targeting therapies. The tumor microenvironment plays a crucial role in the growth and spread of NETs, and therapies designed to alter this environment are being explored. By targeting the blood vessels that supply tumors or modulating immune cells within the TME, researchers aim to limit tumor progression and enhance the effectiveness of traditional treatments.
These ongoing advancements highlight a shift towards personalized, precision medicine in NET treatment, where therapies are tailored based on the molecular and genetic profile of the tumor. As these studies progress, they offer hope for better outcomes and more targeted therapies for patients with NETs.
Choosing the Right Treatment: A Personalized Approach
Every NET case is different, and treatment decisions should be based on multiple factors:
- Tumor location and grade
- Presence of metastases
- Hormone production
- Somatostatin receptor expression (for PRRT eligibility)
- Overall health and patient preferences
A multidisciplinary team of oncologists, endocrinologists, and nuclear medicine specialists is essential to tailor the best treatment plan for each patient.
Why Early Diagnosis of Neuroendocrine Tumors Matters?
Early diagnosis of neuroendocrine tumors (NETs) significantly impacts treatment options, patient outcomes, and overall survival. Because NETs are often slow-growing and produce vague symptoms, they are frequently misdiagnosed or detected late. Detecting them early allows for more effective treatment and a better prognosis.
Increased Treatment Options
When NETs are diagnosed at an early stage, they are often localized and easier to remove with surgery—the only potential cure for most NETs. In contrast, late-stage NETs that have spread to other organs (e.g., liver, bones) require systemic therapies like chemotherapy, targeted therapy, or peptide receptor radionuclide therapy (PRRT), which may only control but not cure the disease.
Improved Survival Rates
The survival outlook for patients with neuroendocrine tumors (NETs) varies widely and is strongly influenced by both the tumor’s grade and the stage at diagnosis. When NETs are detected early and remain localized to the organ of origin, the prognosis is generally favorable, with five-year survival rates exceeding 90% in many cases. These patients often respond well to surgery and may require only limited additional therapy.(American Cancer Society).As the disease progresses and spreads to regional lymph nodes, survival rates decline but remain encouraging with appropriate treatment. These regional NETs often benefit from a combination of surgery, targeted therapy, and somatostatin analogs, depending on the tumor’s biology and hormone activity.
However, once NETs metastasize to distant organs—most commonly the liver—the five-year survival rate drops significantly. In metastatic disease, survival rates typically fall below 30%(American Cancer Society), reflecting the challenges of controlling widespread tumor burden. Despite this, recent advances in therapies such as PRRT, targeted agents, and improved diagnostic tools have led to notable gains in progression-free and overall survival, offering new hope for patients with advanced disease.
Prevention of Severe Symptoms and Complications
Many NETs produce hormones that can cause serious symptoms, such as:
- Carcinoid Syndrome: Flushing, diarrhea, wheezing, and heart valve damage.
- Insulinomas: Dangerous low blood sugar levels.
- Gastrinomas: Severe acid reflux and stomach ulcers.
Early detection allows doctors to manage these hormone-related symptoms before they lead to life-threatening complications.
Delayed Diagnosis Leads to Mismanagement
Many NETs take years to diagnose because symptoms resemble common conditions like irritable bowel syndrome (IBS), asthma, or acid reflux. Studies show that patients wait an average of 52 months (over 4 years) from symptom onset to diagnosis.This delay increases the risk of tumor spread and limits treatment options(American Cancer Society).
Advances in Early Detection Methods
Recent advancements in imaging and biomarkers have improved early NET detection:
- Gallium-68 DOTATATE PET/CT scans can detect even small NETs.
- Chromogranin A (CgA) and 5-HIAA tests help identify functional NETs.
- Liquid biopsy and genetic testing may help detect NETs at even earlier stages
Are Neuroendocrine Tumors Dangerous?
The severity of NETs depends on their location, size, and whether they are slow-growing (low grade) or aggressive (high grade). Some NETs, especially those in the pancreas and GI tract, can produce hormones that cause additional complications. However, survival rates have improved due to advances in treatment .
As research accelerates and precision therapies evolve, neuroendocrine tumors are no longer a silent enigma—early diagnosis, targeted treatment, and patient-centered care are now transforming outcomes and redefining hope.
You Can Watch More on OncoDaily Youtube TV
Written by Armen Gevorgyan, MD
FAQ
What are neuroendocrine tumors?
NETs are abnormal growths that develop in neuroendocrine cells, which have traits similar to nerve cells and hormone-producing cells. They can occur anywhere in the body but are most commonly found in the lungs, appendix, small intestine, rectum, and pancreas.
How common are neuroendocrine tumors?
Although historically considered rare, the incidence of NETs has been increasing. Recent statistics suggest that more than 100,000 people in the United States have NETs, surpassing the number of individuals with other, better-known cancers like pancreatic or stomach cancer.
What causes neuroendocrine tumors?
In most cases, the exact cause is unknown. However, certain inherited conditions, such as multiple endocrine neoplasia types 1 and 2 and Von Hippel-Lindau disease, can increase the risk of developing NETs.
What are the symptoms of neuroendocrine tumors?
Symptoms vary depending on the tumor’s location and whether it secretes hormones. Some NETs may not cause noticeable symptoms, while others can lead to flushing, diarrhea, wheezing, or abdominal pain due to hormone release.
How are neuroendocrine tumors diagnosed?
Diagnosis may involve a combination of imaging tests, blood and urine tests to detect hormone levels, and biopsy procedures to examine tissue samples.
Are there different types of neuroendocrine tumors?
Yes, NETs are classified based on their location (e.g., gastrointestinal tract, pancreas, lungs), grade, and ability to produce hormones that cause symptoms.
What treatment options are available for neuroendocrine tumors?
Treatment depends on factors like tumor type, location, size, and whether it has spread. Options may include surgery, medication to control hormone-related symptoms, targeted therapies, chemotherapy, and radiation therapy.
Can neuroendocrine tumors be cured?
If detected early and localized, NETs can often be surgically removed, potentially leading to a cure. However, if the cancer has spread, treatment focuses on managing the disease and controlling symptoms.
What is the prognosis for someone with a neuroendocrine tumor?
Prognosis varies widely based on factors like tumor type, grade, stage, and overall health. Some NETs grow slowly, allowing for long-term management, while others are more aggressive.
Are neuroendocrine tumors hereditary?
While most NETs are sporadic, a small percentage are associated with inherited syndromes like multiple endocrine neoplasia type 1 (MEN1), which can increase the risk of developing these tumors.