Graft-versus-Host Disease (GVHD) is a potentially life-threatening complication of allogeneic hematopoietic stem cell transplantation (allo-HSCT), a curative therapy for various malignant and non-malignant hematologic disorders.
Transplant is not merely the replacement of marrow – it is the transfer of an immunity. Accordingly, ongoing efforts aim to better regulate donor immunity without compromising its anti-leukemia effect. Against this backdrop, it is worth exploring the evolving landscape of GVHD management.
Why GVHD Happens
For GVHD to occur, immunocompetent cells should be transplanted into immunodeficient recipients, and these transferred cells should recognize alloantigens in the host. Other important factors influencing the risk and severity of GVHD include:
1. Donor-host factors: The incidence increases with unmatched donor transplants, HLA disparity, and sex mismatching.
2. Stem-cell source factors: Cryopreservation of marrow before infusion and the use of umbilical cord blood decrease the incidence.
3. Immune modulation factors: The incidence is lower with triple therapy (cyclosporine, methotrexate and prednisone) as compared with double therapy (cyclosporine and MTX). In preclinical settings, statins can inhibit antigen-presenting cells function and reduce the expression of MHC class II.
4. Chemotherapy and radiation therapy: High-dose chemotherapy causes tissue damage and increases circulating cytokine levels, thereby enhancing the ability of immune cells in the graft to recognize host antigens.
5. Regimens, including total body irradiation, are associated with increased incidence and severity of GVHD compared to chemotherapy alone (Justiz Vaillant et al., 2024).
Acute vs Chronic GVHD
Acute GVHD is mediated by donor T lymphocytes recognizing recipient antigens, followed by their activation, proliferation, inflammatory cytokine release and cytolytic effects that eliminate the patient’s HLA-disparate cells. Chronic GVHD is mediated by T-cell, B-cell and macrophage activation with eventual inflammation and fibrosis.
According to the NIH classification:
- Classic acute GVHD develops within 100 days of transplantation with typical acute features.
- Persistent, recurrent, or late-onset acute GVHD shows acute manifestations beyond day 100.
- Classic chronic GVHD occurs after day 100 with characteristic chronic features.
- Overlap syndrome may arise at any time post-transplant and includes features of both acute and chronic GVHD.
Acute GVHD commonly affects the skin, liver, GI tract, developing manifestations such as maculopapular rash, jaundice, nausea, abdominal pain mucosal ulceration, and diarrhea. The rash eruption may spread diffusely and become confluent. In severe cases, bullous lesions resembling toxic epidermal necrolysis (TEN) may develop. It may also involve the lungs, kidneys, eyes, and hematopoietic system, and can reduce responsiveness to active immunization.
Chronic GVHD presentation often resembles autoimmune or fibrosing disorders and may involve almost any organ. Oral involvement may resemble lichen planus and carries an increased risk of progression to oral squamous cell carcinoma, which may behave more aggressively in stem cell transplant recipients.
Recurrent infections, often related to prolonged immunosuppression, remain an important cause of mortality. Ocular involvement is also associated with poorer prognosis and typically affects the ocular surface, presenting as dry eye syndrome or keratoconjunctivitis sicca. Oral ulcerations, bronchiolitis obliterans, joint stiffness, and hepatic dysfunction are also relatively common.
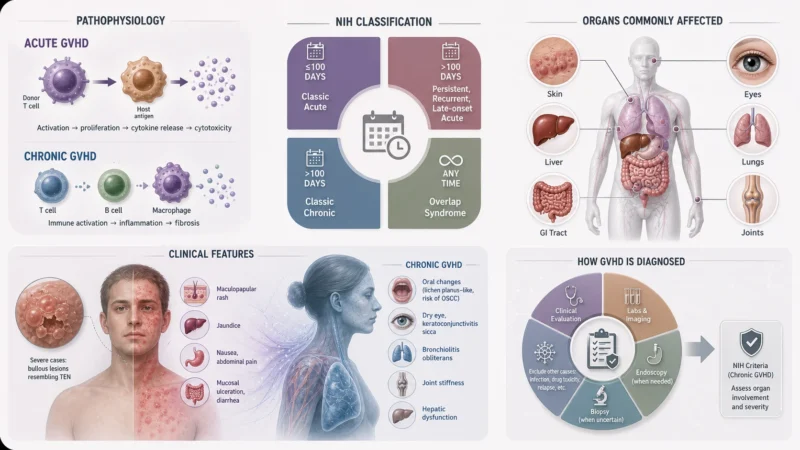
GVHD is primarily a clinical diagnosis. Laboratory tests, imaging, and endoscopy may help assess severity and exclude alternative causes such as infection, drug toxicity, relapse, or other transplant-related complications. Tissue biopsy of the skin, gastrointestinal tract, liver, or other affected organs can provide histologic support, particularly when the diagnosis is uncertain. In chronic GVHD, NIH consensus criteria are commonly used to classify organ involvement and disease severity.
The Balance: GVHD vs Graft-versus-Leukemia Effect
cGVHD remains a leading cause of non-relapse mortality in patients surviving more than two years after allogeneic HCT. At the same time, cGVHD has a biologically important protective role, as affected patients often experience lower relapse rates of their underlying malignancy because of an enhanced graft-versus-leukemia effect.
OS in patients with mild to moderate cGVHD is generally comparable to those without this complication, as the increased mortality risk may be offset by lower disease-related mortality. In contrast, severe forms are associated with long-term mortality rates of up to 50%, particularly in patients with prior severe acute GVHD.
Beyond its impact on survival, cGVHD substantially impairs quality of life, contributing to greater fatigue, increased medication burden, and progressive functional decline.
Investigational Biomarkers
The identification of biomarkers for acute GVHD diagnosis and prognosis has become a major area of research worldwide. Proteins including pro-inflammatory cytokines and factors associated with GVHD-specific organ injury, measured at defined time points, have been evaluated as potential biomarkers. Elevated levels of ST2 (suppression of tumorigenicity 2) are associated with an increased risk of severe aGVHD and may improve risk stratification for response to first-line therapy.
Plasma elafin levels rise at the onset of cutaneous aGVHD. In lower gastrointestinal aGVHD, regenerating islet-derived protein 3α, cytokeratin 18, and hepatocyte growth factor levels are significantly higher than in patients with non-GVHD diarrhea. These latter biomarkers may also be elevated in hepatic aGVHD, however, they do not reliably distinguish GVHD from other causes of hyperbilirubinemia.
Matrix metalloproteinase-3 has been proposed as diagnostic biomarker for bronchiolitis obliterans syndrome in cGVHD. Parametric response mapping is a promising CT-based tool for diagnosis and monitoring (Choe H. et al., 2025).
How GVHD Is Managed
Immunosuppressive medications are the cornerstone:
- Corticosteroids: Prednisone and methylprednisolone are often used initially.
- Calcineurin inhibitors: Cyclosporine and tacrolimus are used in combination with steroids.
- Mycophenolate mofetil, sirolimus, and azathioprine may also be used in combination.
Antithymocyte globulin may be used to suppress the immune system in severe cases of GVHD, particularly in patients who do not respond to initial treatments.
Biological therapies can target specific components:
- TNF inhibitors: Infliximab and etanercept for severe GI GVHD.
- IL-2 receptor antibodies: Basiliximab and daclizumab.
Photopheresis involves removing WBCs from the patient’s blood, exposing them to a photosensitizing agent, ultraviolet light and returning them to the patient. It is often used in chronic GVHD cases. Topical treatments may be used for skin involvement.
Supportive care includes managing symptoms and complications associated with infections, nutritional deficiencies, and electrolyte imbalances.
Approaches to Prevention
Newer strategies build on a CNI backbone by adding in-vivo immune modulation:
- posttransplant cyclophosphamide (PTCy)
- abatacept (a CTLA-4 immunoglobulin soluble fusion protein)
- sirolimus (mTOR inhibition)
Additionally, targeting immune pathways such as:
- T-cell trafficking blockade (vedolizumab)
- cytokine signaling blockade (JAK inhibitors)
- B-cell blockade (especially for cGVHD prevention)
PTCy and abatacept have significantly improved the safety and efficacy of allogeneic transplantation in both HLA-mismatched and HLA-matched donor settings (Gooptu et al., 2026).
Steroid-Resistant GVHD
Approximately 50% of patients with acute GVHD fail to respond to standard first-line therapy with systemic high-dose glucocorticoids, and in grade 4 disease, response rates do not exceed 30%. Real-world data suggest that steroid resistance is more frequent in patients with GI GVHD. Outcomes remain poor, with a median survival of less than 12 months, particularly because this group is prone to infections, bleeding and surgical events.
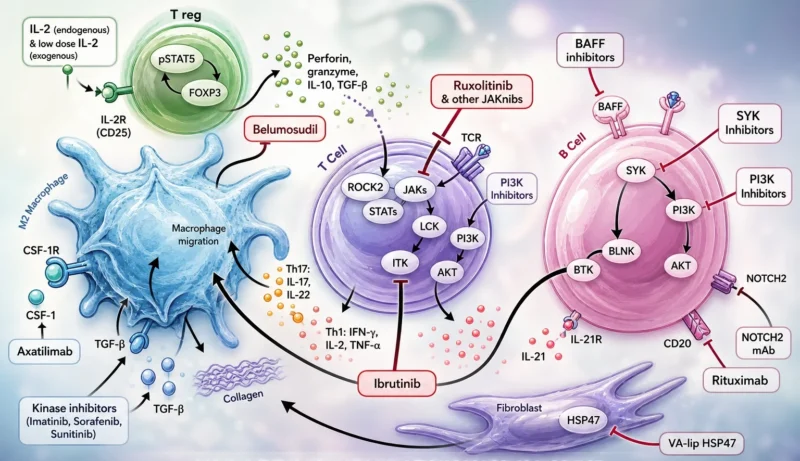
Currently approved second-line therapies for SR-cGVHD include Ibrutinib, Ruxolitinib (also for SR-aGVHD), Belumosudil, and Axatilimab. Optimal sequencing beyond ruxolitinib (the standard second-line therapy) remains uncertain and is generally guided by the predominant organ involvement and the safety profile of each agent.
Belumosudil inhibits ROCK2, thereby modulating T-cell differentiation, reducing fibrosis, and suppressing inflammatory cytokine signaling. In the ROCKstar trial ORR was 75%, with a median response duration of 54 weeks.
Axatilimab targets CSF1R expressed on macrophages and monocytes. In the phase II AGAVE-201 trial, it demonstrated the highest ORR at 74%, with 60% durable responses at 12 months. Combination use with ruxolitinib showed 47% response rates in pulmonary cGVHD.
Ibrutinib irreversibly inhibits BTK and ITK, affecting both B-cell receptor signaling and T-cell inflammatory pathways. FDA approval followed clinical data demonstrating an ORR of 67% across affected organ systems. Among responders, 71% maintained response for at least 20 weeks. Real-world analyses suggest higher activity in hepatic cGVHD.
Ongoing Research
Mesenchymal stem cells (MSCs) have been explored as a potential treatment option for cGVHD, potentially through expansion of regulatory T cells, particularly in steroid-refractory disease. Ongoing research aims to optimize cell source, dosing, and treatment schedules.
Rovadicitinib is an oral, selective dual JAK1/2 and ROCK1/2 inhibitor designed to target both inflammatory and fibrotic pathways in cGVHD. With further validation, it may become an option for heavily pretreated patients. Pimicotinib (CSF1R inhibitor) is being investigated as a potential therapy for cGVHD.
Artificial intelligence is increasingly being explored in steroid-refractory cGVHD management. By integrating clinical data, imaging, and multi-omics information, AI models may help identify therapeutic targets, predict treatment response and improve patient stratification. Recent work has demonstrated the feasibility of autonomous algorithms capable of independently recommending Ruxolitinib prophylaxis strategies in transplant settings (Choe et al., Denk et al., 2025, El Jurdi et al., Saadeh et al., 2026).
You Can Watch More on OncoDaily TV
Current Limitations
While the cumulative efforts of the scientific community, together with funding agencies, have led to meaningful progress in cGVHD prevention and treatment, it remains neither obsolete nor trivial. After more than four decades, corticosteroids continue to be the standard first-line systemic treatment, although they remain suboptimal and are associated with significant morbidity during long-term use.
Once patients develop SR-cGVHD, which occurs in ~50% of cases, many go on to receive multiple subsequent lines of therapy. This often results in increased morbidity, declining quality of life, and substantial healthcare costs. Currently, treatment algorithms are largely based on regulatory approval, drug availability, side-effect profile, cost, and physician experience/preference, with many patients failing to achieve or maintain CR over time.
All four agents approved for SR-cGVHD were supported by open-label studies, with only Ruxolitinib having a best available therapy comparator arm. Future efforts are expected to focus on developing treatment algorithms that are preemptive, targeted and tailored to each patient’s underlying pathomechanisms, leading to more effective and less toxic approaches.
Life Beyond Transplant
Even when the malignancy is controlled, patients may continue to experience persistent physical limitations that can significantly disrupt work, relationships and daily life.
The emotional burden can be equally profound, as many patients face uncertainty, loss of independence, social isolation and the expectation that they should feel “finished” once the transplant is over.
For this reason, survivorship care must extend beyond immunosuppression alone and include rehabilitation, mental health care, symptom-directed follow-up and clear communication that recovery is often gradual rather than linear.
You can also read: Acute Lymphoblastic Leukemia (ALL) in Adults vs Children: Why Such Differences in Survival
Written by Susanna Mikayelyan, MD
FAQ
Can GVHD occur years after a stem cell transplant?
Yes. While acute GVHD often develops earlier after transplant, chronic GVHD can appear months or even years later. Some patients may also experience persistent or late-onset manifestations that require long-term follow-up.
Is GVHD a sign that the transplant is working?
Not necessarily, but mild GVHD can reflect donor immune activity, which may be associated with a beneficial graft-versus-leukemia effect. However, clinically significant GVHD can also cause major complications and should not be considered desirable.
Why do some patients develop GVHD while others do not?
Risk depends on several factors, including donor-recipient HLA compatibility, donor type, stem-cell source, conditioning intensity, age, immune prophylaxis, and individual biological differences.
Can GVHD be mistaken for infection or medication side effects?
Yes. Rash, diarrhea, liver dysfunction, dry eyes, and fatigue may also result from infections, drug toxicity, relapse, or other transplant-related complications. This is why diagnosis often requires careful clinical evaluation.
How does GVHD affect quality of life?
GVHD may contribute to fatigue, pain, skin tightness, dry eyes, reduced mobility, sleep disturbance, emotional stress, and difficulty returning to normal daily activities or work.
Are newer treatments replacing traditional immunosuppression?
Newer targeted therapies such as JAK inhibitors, ROCK2 inhibitors, and CSF1R-targeted agents are expanding options, especially in steroid-refractory disease. However, conventional immunosuppression still remains central in many settings.



