Dr. Richard Goldberg, a leading medical oncologist who helped establish the global standard of care for metastatic colorectal cancer, is raising urgent concerns about how modern cancer therapies are evaluated. In a recent perspective published in The Cancer Letter, he argues that current regulatory frameworks, particularly those used by the U.S. Food and Drug Administration, remain rooted in chemotherapy-era assumptions. Goldberg calls for a shift toward evaluation standards that better reflect the biology of immunotherapy, emphasizing that traditional metrics such as early tumor shrinkage may fail to capture meaningful long-term survival benefits.
The Young Patients Behind the Numbers
The rise of colorectal cancer in younger adults has become one of the most alarming trends in modern oncology. These are patients in their 20s, 30s, and 40s, often in the middle of building careers, raising families, and living with the expectation that cancer is still a distant risk. Instead, many are being diagnosed only after symptoms become impossible to ignore.
Colorectal cancer is no longer a disease defined mainly by older age. In a troubling shift, it became the leading cause of cancer-related death in Americans younger than 50 in 2023, rising from the fifth-leading cause in the early 1990s, according to a research letter published in JAMA. At the same time, overall cancer mortality in this age group declined by 44% from 1990 to 2023, falling from 25.5 to 14.2 per 100,000. Colorectal cancer moved in the opposite direction, with mortality increasing by 1.1% annually since 2005. (Siegel et al., 2026).
That contrast is more than a statistic. It is a warning that progress in oncology is not reaching every disease, every patient group, or every clinical setting equally. It also raises a harder question: when science advances, are regulatory systems moving fast enough to help patients benefit from it?
The problem is not only incidence. Mortality is rising as well, and that makes this trend especially serious. As highlighted in the data cited by Dr. Richard Goldberg, three in four patients younger than 50 are diagnosed with advanced colorectal cancer, suggesting that prevention, awareness, and early detection are still falling short.
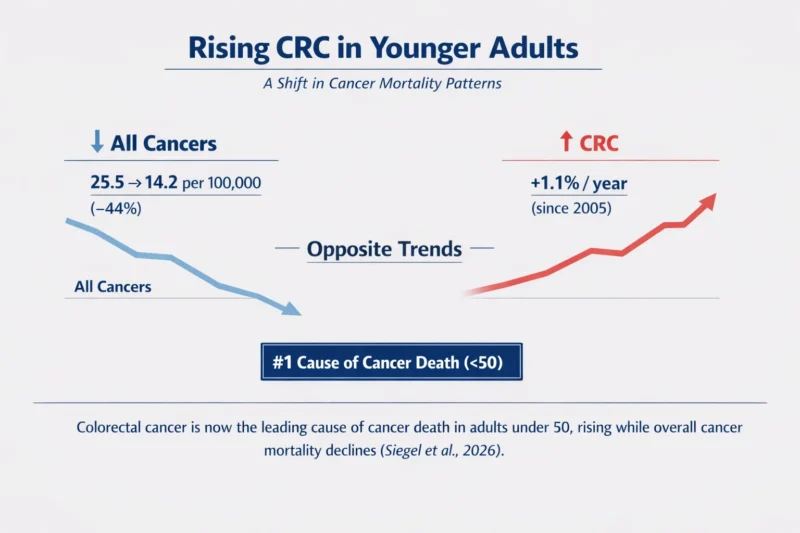
Screening recommendations have moved earlier, with average-risk screening now recommended beginning at age 45, and even earlier for people with a family or genetic risk.(Siegel et al., 2023) But awareness remains inconsistent, and many younger adults do not see themselves, or are not seen by clinicians, as possible colorectal cancer patients until the disease is already advanced.
Why This Matters Even More in Metastatic Disease
For many patients with metastatic colorectal cancer, especially those with microsatellite-stable disease, which accounts for about 95% of cases, treatment options remain limited. Standard surgery, radiation, and chemotherapy can still play important roles, but after progression, many approved therapies offer only modest gains. For some patients, median survival may remain less than one year, often at the cost of substantial toxicity.
This is where the urgency of innovation becomes real. New immune-based therapies and combinations are being studied in an effort to improve outcomes in a disease that has long remained difficult to treat once it becomes metastatic and resistant. But according to Goldberg, the regulatory system evaluating these therapies is still anchored to assumptions built in the chemotherapy era.
The Central Problem: Old Metrics for New Therapies
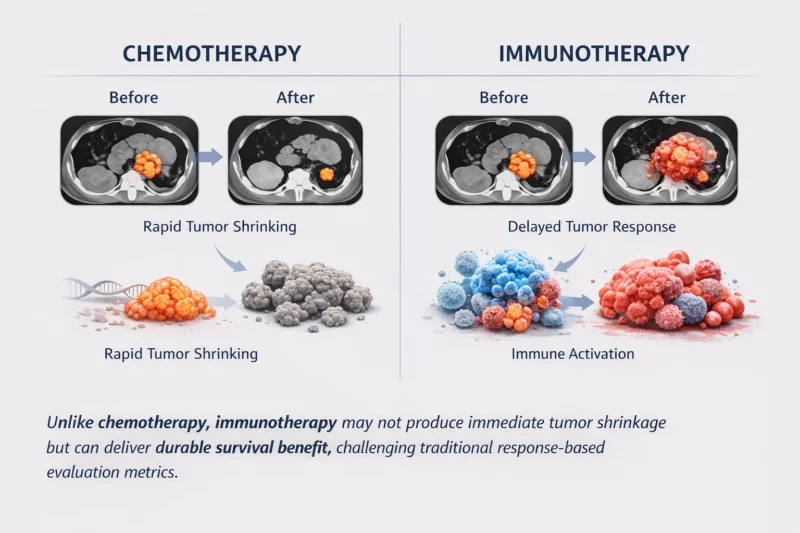
In his recent piece in The Cancer Letter, Dr. Goldberg argues that the FDA continues to rely too heavily on objective response rate, or tumor shrinkage, when evaluating therapies for accelerated approval. That framework made sense when cytotoxic chemotherapy dominated oncology. These drugs often produce visible tumor shrinkage within days or weeks, making radiographic response an acceptable early signal of clinical benefit.
These discussions reflect a systemic challenge: when risk–benefit assessments become overly conservative even after safety is established the result is not just institutional discomfort, but lost time for patients.
Immunotherapy is Different.
Rather than directly killing cancer cells in the way chemotherapy does, immunotherapy works by helping the immune system recognize and attack tumor cells. That process may take longer. Tumors may initially appear stable. In some cases, lesions may even seem larger early on because of immune-cell infiltration and inflammation. Yet these same patients may later experience prolonged disease control and meaningful survival benefit.
As Goldberg explains,
“In clinical practice, we’ve adapted our approach. We understand that therapies like immunotherapy may not show immediate results, but the survival benefits they bring are unparalleled.”
Why Response Rate Alone Is No Longer Enough
The core of Goldberg’s argument is not that standards should be weakened. It is that standards should be updated to reflect how modern therapies actually work.
A therapy that does not produce dramatic early radiographic responses may still be helping patients live substantially longer. In metastatic colorectal cancer, where options are limited and prognosis can be poor, that matters enormously. A treatment that extends survival to one, two, or even three years without the burden of traditional chemotherapy-related toxicity is not a marginal advance. It is clinically meaningful benefit.
From the patient perspective, these are not abstract endpoint debates. They are questions of time, function, and quality of life. For someone expected to survive six months, an additional year or two may mean seeing children grow older, returning home without debilitating toxicity, or simply living with less suffering.
Goldberg argues that disease control, durability of effect, and landmark survival at one, two, and three years should carry far greater weight in regulatory decisions involving immunotherapy. In the current era, early response rate alone may no longer be a reliable surrogate for true patient benefit.
A Broader Debate Around FDA Delays
This argument is emerging at a time of wider scrutiny of the FDA’s review processes. As noted in reporting from POLITICO (Lim, D., & Gardner, L. (2026), a recent Senate Committee on Aging hearing featured bipartisan concern about regulatory delays, inconsistent review practices, shifting standards, late-stage data requests, and limited use of existing flexibility for diseases with high unmet need.
“Delays are not neutral,” says Goldberg. “They cost lives, especially in fast-moving diseases like metastatic CRC.”
Those concerns were not limited to colorectal cancer. But colorectal cancer, particularly in younger adults, offers one of the clearest examples of why the issue matters. When a disease is rising, when patients are being diagnosed late, and when treatment innovation is urgently needed, delays in evaluation are not neutral. They have consequences. Goldberg’s point is that once safety is established, excessive conservatism in judging benefit can cost lives. The consequence is not merely slower paperwork. It is lost time for patients who may not have much of it.
Modernizing Regulation Is Not Lowering the Bar
Calls for regulatory modernization are sometimes misread as calls for reduced rigor. That is not what this discussion is about. The FDA’s role in protecting patients remains essential. Oncology cannot afford approvals based on weak evidence or unsafe assumptions. But a scientifically rigorous system must also be biologically informed. If the mechanism of action has changed, the logic of evaluation must change with it. That means recognizing that immunotherapy may not behave like chemotherapy on the first scan. It means accepting that delayed benefit can still be real benefit. It means looking beyond shrinkage and incorporating endpoints that better capture the value of modern cancer treatment.
In practice, this may require greater emphasis on durability, long-term survival, immune-related response patterns, and disease control in settings where traditional endpoints fail to tell the full story.
You Can Also Read Is Colorectal Cancer Treatment Moving in the Wrong Direction for Adults Under 50? by OncoDaily

The Stakes for Younger Adults
The timing of this debate is especially important because early-onset colorectal cancer is no longer a future concern. It is a present crisis.
The JAMA data show that while mortality from lung cancer, leukemia, breast cancer, and brain cancer in younger adults has continued to decline, colorectal cancer is now moving against the current. It is becoming deadlier in a population that has historically been considered outside the main screening window and outside the traditional image of colorectal cancer.
That should force action on multiple fronts. Prevention and earlier diagnosis must improve. Red-flag symptoms must be recognized earlier by both clinicians and the public. Screening uptake must expand. But innovation must also move faster from trial to clinic when evidence shows that therapies may offer real benefit. A rising mortality curve cannot be met with outdated assumptions.
Science Has Changed. Regulation Must Change Too
Oncology has entered an era in which the most meaningful therapeutic gains do not always appear in the earliest scan-based endpoints. In some cases, the patients who benefit most are not the ones whose tumors shrink fastest. They are the ones who live longer, stay well longer, and gain time that older models of evaluation may fail to predict.
That is the message at the center of Goldberg’s argument, and it arrives at a moment when colorectal cancer in younger adults is demanding more than awareness alone. It is demanding alignment between research, clinical urgency, and regulatory judgment.
If colorectal cancer is now the leading cause of cancer death in Americans under 50, then the response cannot be incremental. Prevention must improve. Detection must come earlier. And when science offers promising new strategies, regulators must be prepared to evaluate them with tools that fit the therapies of today, not the treatments of decades past.
The biology has changed. The patients have changed. The stakes have changed.
The standards must change too.


