Rabi Hanna, Pediatric Blood and Marrow Transplant Program Director, and Chairman at the Department of Pediatric Hematology-Oncology and BMT at Cleveland Clinic, shared a post on LinkedIn:
“I am excited to share on behalf of all co-investgaitors the NEJM publication of the RUBY Trial sponsored by Editas Medicine.
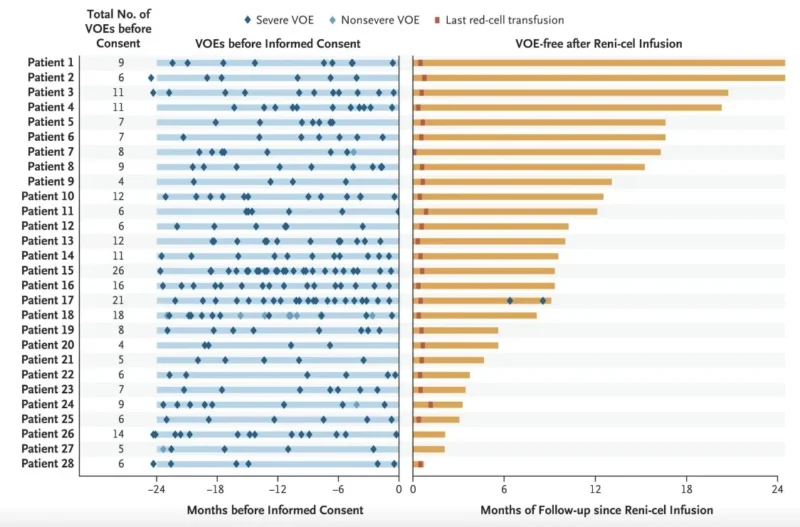
Study summary: ‘Reni-cel’ The first in-human CRISPR/Cas12a gene-editing therapy for Sickel cell diseases shows excellent improvements in clinical outcomes. Out of 28 patients treated, 27 have experienced a complete resolution of painful sickle cell crises and the safety and toxicity as consistent with other GeneEditing therapies that use myeloablative busulfan conditioning therapy. Other biological benefits showed that fetal hemoglobin was>40% by month 3 and total hemoglobin levels recovering to mean13.8 g/dL around 6 month.
It is immensely rewarding to add new, highly effective therapeutic options for patients living with SCD. What makes this science so elegant is that by utilizing CRISPR to target the HBG1/HBG2 promoters, we are beautifully mimicking the natural, protective experiment seen in individuals who have mutations occurring in HgbG1/G2 and causing hereditary persistence of fetal hemoglobin (HPFH)
Furthermore, this success highlights the incredible breadth of CRISPR applications and our expanding ability to safely target different genes to treat complex diseases. However, as we celebrate this massive leap forward, we must also recognize the work that still lies ahead to fully realize the promise of genetic medicine:
Regulatory Evolution: We need regulatory frameworks to advance and adapt to match the radical pace of these technological breakthroughs, particularly to accelerate treatments for ultra-rare disorders.
Better Conditioning Therapies: We must develop improved conditioning regimens that rely on less chemotherapy so we can significantly decrease long-term toxicities for our patients.
Manufacturing and Efficiency: Because stem cell collection can be exceptionally challenging in SCD, we need continued improvements in manufacturing and gene-editing efficiency.
Equitable Access: Finally, we need more creative financial models and public/private partnerships to increase access and ensure these life-changing therapies reach all patients globally.
I want to extend my deepest gratitude to all the investigators involved in this milestone, and a very special thank you to our phenomenal clinical and research teams at Cleveland Clinic Children’s and Cleveland Clinic who worked tirelessly to perform this study. This progress wouldn’t be possible without your dedication.”
Title: CRISPR-Cas12a Gene Editing of HBG1 and HBG2 Promoters to Treat Sickle Cell Disease
Authors: Rabi Hanna, Haydar Frangoul, Luis Pineiro, Christopher McKinney, Markus Mapara, Jignesh Dalal, Hemalatha G. Rangarajan, Harold Atkins, Akshay Sharma, Kai-Hsin Chang, Michael C. Jaskolka, Keunpyo Kim, Qifeng Yu, Baisong Mei, Olubunmi Afonja, Mark C. Walters
Read the Full Article.

Other Articles Featuring Rabi Hanna On OncoDaily.


