Michel Frank Ferrazo, Purchased Materials Technician at ABL Antibióticos do Brasil Ltda, shared a post on LinkedIn:
“In vivo CAR-T engineering is moving from concept to a broader preclinical platform landscape.
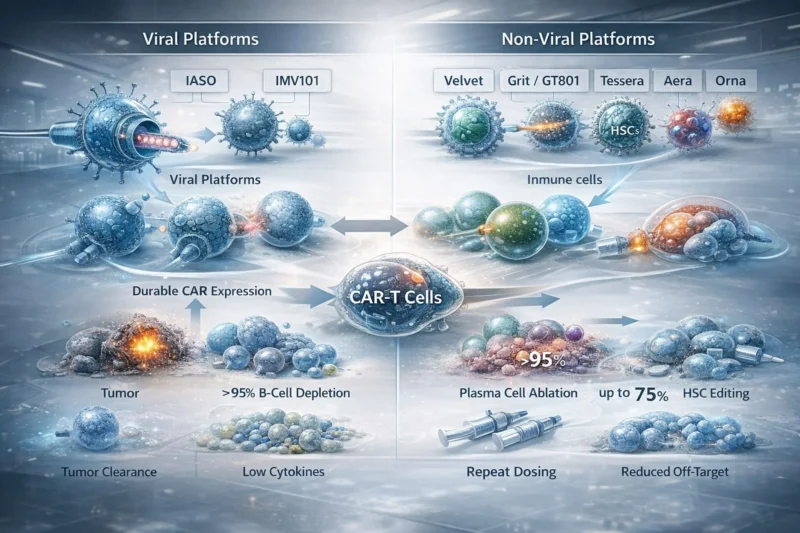
A concise correspondence from the 2025 ASH meeting highlights how both viral and non-viral delivery systems are being designed to reprogram endogenous immune cells directly in vivo, with the goal of addressing key ex vivo CAR-T constraints such as manufacturing complexity, cost, lymphodepletion-related burden, and safety concerns.
What stands out is the diversity of engineering strategies now being explored: targeted lentiviral systems for resting or niche-resident T cells, T cell-directed LNP approaches carrying CAR mRNA, and platforms extending beyond T-cell redirection into HSC editing. The paper summarizes signals of strong preclinical activity, including complete tumor clearance in some models, >95% B-cell depletion in selected settings, and up to 75% HSC editing in one platform.
The translational angle is also interesting. The summarized programs span hematologic malignancies, multiple myeloma, lupus, RA, and other autoimmune contexts, suggesting that in vivo CAR approaches may evolve as more flexible immune engineering modalities rather than a single product class.
At the same time, the article is careful about the open questions: viral durability may come with non-reversible expression and integration concerns, while non-viral systems may require repeat dosing. Most evidence discussed remains preclinical and often based on immunodeficient models, so immunogenicity, clearance, and immune-related adverse events in humans are still unresolved.
Preclinical/early evidence; clinical impact remains to be validated.
A useful snapshot of where the field is heading: not just better CAR-T manufacturing, but potentially new operating models for in vivo immune reprogramming. Curious to hear your thoughts.”
Other articles featuring Michel Frank Ferrazo on OncoDaily.


