Mamtha Balla, Transplant Oncology-ID fellow at MD Anderson Cancer Center and Hematology oncology Fellow at The University of Toledo, shared a post on X:
“Myelodysplastic Syndrome (MDS) – High-Yield Summary.
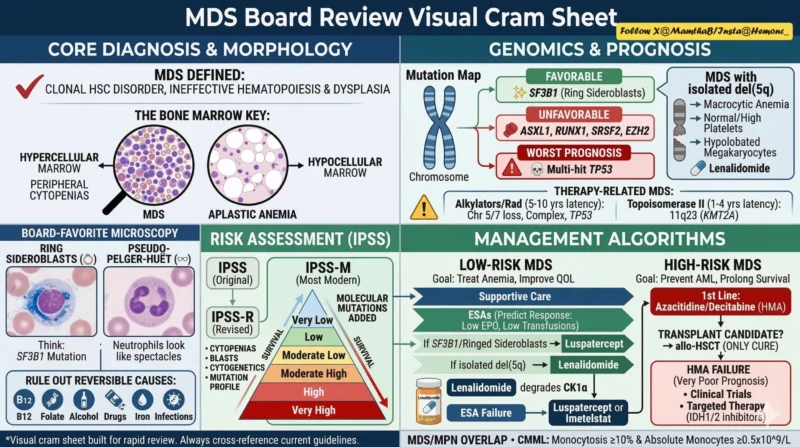
Memory mnemonic. Mutations that have a unfavorable prognosis. Terrible (TP53), Awful (ASXL1), Rigid (RUNX1), and Splicing (SRSF2) mutations cause Early death (EZH2).
Basics and Pathogenesis
- MDS = clonal HSC disorder – ineffective hematopoiesis, dysplasia, cytopenias, AML risk.
- Median age ~73, incidence rises sharply after 70; parallels CHIP.
- Model: CHIP – CCUS – low‑risk MDS – high‑risk MDS – AML.
Diagnosis and Morphology
- Needs ≥1 cytopenia + ≥10% dysplasia in ≥1 lineage ± blasts/cytogenetics.
- Marrow: usually hypercellular with ineffective hematopoiesis (vs hypocellular in aplastic anemia).
- Morph ‘buzzwords’: ring sideroblasts (SF3B1), pseudo-Pelger‑Huët neutrophils, micromegakaryocytes, blasts.
Key Mutations and Genomics
- SF3B1 – ring sideroblasts, anemia, generally favorable prognosis.
- Multi‑hit TP53 (biallelic/high VAF) – complex karyotype, very poor prognosis, lenalidomide resistance, therapy‑related disease.
- More driver mutations = worse prognosis; IPSS‑M formally incorporates mutation profile.
Cytogenetics to Memorize
- Isolated del(5q) – macrocytic anemia, normal/high Plt, hypolobated megakaryocytes, treat with lenalidomide.
- -7/del(7q), del(17p), complex karyotype = higher‑risk; some are MDS‑defining even without dysplasia.
- AML‑defining (even with low blasts): NPM1, inv(3), t(6;9), core‑binding factor.
Risk Scores (Boards Love This)
- IPSS‑R: blasts + cytogenetics + depth/number of cytopenias – very low – very high.
- IPSS‑M: IPSS‑R + mutations (TP53, ASXL1, RUNX1, EZH2, SRSF2, U2AF1 etc) – 6 risk groups.
- Multi‑hit TP53 = worst group in IPSS‑M.
CHIP / ICUS / CCUS
- CHIP: clonal mutations (DTA etc) without cytopenias – ~0.5–1%/yr risk MDS/AML + CV mortality.
- ICUS: cytopenia, no dysplasia, no clone.
- CCUS: cytopenia + clonal mutation, no morphologic MDS; higher progression risk – MDS.
- MDS/MPN Overlap Have both cytopenias/dysplasia AND proliferation; must exclude BCR‑ABL1/PDGFRA/B etc.
- CMML: monocytes ≥0.5 ×10⁹/L and ≥10% WBC; – MD‑CMML (WBC <13k),
- MP‑CMML (WBC ≥13k, RAS‑mutant, more aggressive).
- MDS/MPN with neutrophilia (aCML), and MDS/MPN with SF3B1 + thrombocytosis (RARS‑T) – think CSF3R/JAK2/CALR/MPL.
Therapy‑Related and Germline
- Alkylators/radiation – t‑MDS after 5–10 yrs, complex karyotype, TP53, monosomy 5/7
- Topo II inhibitors – 1-4 yrs, 11q23 rearrangements, usually no TP53.
- Germline: dyskeratosis congenita (telomerase), Fanconi (DNA repair), RUNX1, GATA2, DDX41 (older‑onset familial MDS)
Low‑Risk MDS – Treatment Ladder
- Goal: fix cytopenias, reduce transfusions, improve QoL (not necessarily cure).
- Step 1: check serum EPO. Low EPO/low transfusion – ESA (EPO/darbepoetin).
- ESA failure + SF3B1/ring sideroblasts – luspatercept (TGF‑β ligand trap)
- del(5q) – lenalidomide (cereblon – CK1α degradation on 5q; p53‑dependent killing)
- ESA‑failure, non‑del(5q) – luspatercept, lenalidomide, imetelstat (telomerase inhibitor, myelosuppression), or HMA/clinical trial
- Heavy transfusion low‑risk – consider iron chelation (deferasirox) esp if transplant candidate, target ferritin <1000.
High‑Risk MDS – Core Strategy
- Goal: prevent AML transformation + prolong survival.[ppl-ai-file-upload.s3.amazonaws]
- First‑line: HMAs (azacitidine, decitabine; IV or oral ASTX727), disease control and bridge to transplant; azacitidine improves OS.
- HMA failure – median survival ~4-5 months – prioritize clinical trials, transplant if possible, targeted therapy.
- IDH1 – ivosidenib; IDH2 – enasidenib (oral, ~40-50% response, ~14-17 mo OS)
- Transplant – Boards’ Favorite Line.
- Allo‑HSCT is the ONLY curative therapy for MDS.
- Age ≠ absolute contraindication; RIC allows HSCT in 60-70+ with meaningful survival via graft‑versus‑leukemia effect.
High‑/very‑high IPSS‑R/IPSS‑M – early transplant when feasible; low‑risk – delay until progression or poor biology. Donor vs no‑donor studies: donor/HSCT arm – better OS in Int‑2/high‑risk MDS.
Ultra‑High‑Yield One‑Liners
- Hypercellular marrow + cytopenias + dysplasia in an elderly patient = MDS until proven otherwise.
- Macrocytic anemia + thrombocytosis + isolated del(5q) – lenalidomide.
- Ring sideroblasts + SF3B1 – good prognosis, treat anemia with luspatercept.
- Multi‑hit TP53 or complex karyotype – very poor prognosis; think therapy‑related, HMA/HSCT if possible.
- High‑risk: HMA now, allo‑HSCT when you can, trials/targets when you can’t.”
Other articles about MDS on OncoDaily.


