Karen Canfell, Lead of Cancer Elimination Collaboration (CEC) at the University of Sydney, shared Jesper Bonde’s, Molecular biologist, HPV and cancer expert, extensive lab manager experience at AHH-Hvidovre Hospital, post on LinkedIn, adding:
“Congratulations Jesper Bonde and colleagues on this important implementation trial of HPV vs cytology screening in Denmark! The study confirms increased detection of precancerous disease with primary HPV screening, in a large cohort.”
Quoting Jesper Bonde’s post:
“Outcome of the Danish phased implementation of HPV screening now reported in International Journal of Cancer.
The Danish Nationwide Health Care Policy (HCP) Trial represents one of the first real-world evaluations of primary HPV screening versus high-quality cytology-based screening within a population-based national program.
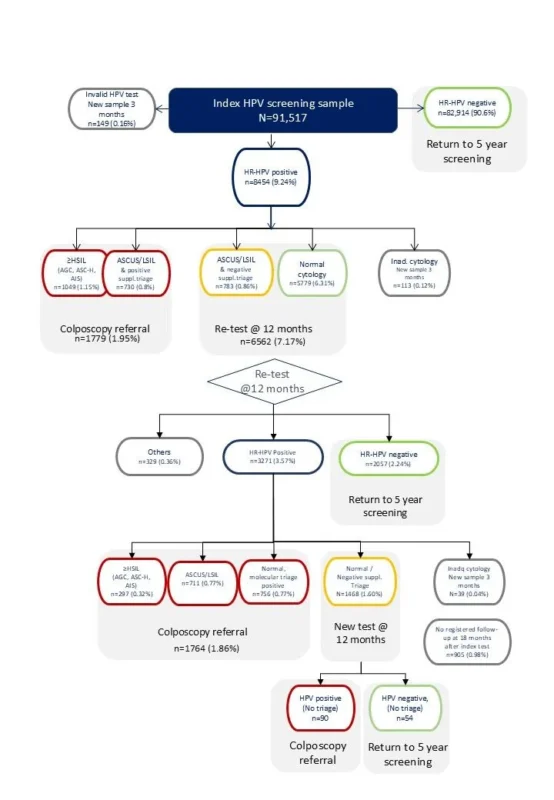
Conducted across all five Danish regions in 2021, the trial included 178,323 women aged 30-59 years undergoing cervical cancer screening: HPV screening: 91,517 women, Cytology screening: 86,806 women, Follow-up: 18 months nationwide.
Importantly, the program implemented innovative risk-based HPV screening algorithms integrating HPV testing with molecular and cytology triage to optimize follow-up recommendations in routine clinical practice.
The findings were striking:
- HPV screening detected significantly more cervical disease
- Detection of ≥CIN2 increased by 51%
- Detection of ≥CIN3 increased by 35%
- HPV screening more effectively identified women at increased risk, even within a setting like Denmark, characterized by exceptionally high-quality cytology screening.
The study also provided critical insight into implementation and healthcare resource implications:
- More repeat testing followed positive HPV results
- Fewer immediate colposcopy referrals occurred after index screening
- However, cumulative colposcopy referrals increased over time (6.3% vs 4.9%), consistent with the prevalence-based detection inherent to HPV testing.
Beyond demonstrating improved disease detection, this nationwide implementation trial offered policymakers a real-world understanding of the operational consequences of transitioning to HPV-based screening at scale.
Most importantly, evidence from this HCP trial directly informed Denmark’s national transition in from cytology-based to primary HPV-based cervical cancer screening. An example of how population-level evidence can shape national cancer prevention policy and future screening strategies worldwide.”
Title: Health Care Policy Implementation Trial of Primary Human Papillomavirus-Based Cervical Screening in Denmark
Authors: Jesper Bonde, Jeppe Bennekou Schroll, Birgitte Tønnes Pedersen, Elsebeth Lynge, Marianne Waldstrøm, Petra Hall Viborg, Anna Frandsen, Rikke Holst Andersen, Susanne Nielsen, Doris Schledermann, Bettina Kjær Kristensen, Berit Andersen, on behalf of the National Danish Cervical Cancer Screening Steering Committee
Other articles featuring Karen Canfell on OncoDaily.


