Balazs Halmos, Professor of Medicine at Montefiore Health System, shared a post by Toni Choueiri on LinkedIn, adding:
“Disappointing news for the added value of anti-LAG3, fianlimab, despite trends in high dose arm. With that, now a decade into alternate checkpoint inhibitor trials- TIM-3, TIGIT, LAG-3– real breakthroughs just continue to… lag”
Quoting Toni Choueiri, the Director of the Lank Center for Genitourinary Oncology at Dana-Farber Cancer Institute (DFCI), Co-Leader of the Kidney Cancer Program at Dana-Farber/Harvard Cancer Center, and the Jerome and Nancy Kohlberg Chair and Professor of Medicine at Harvard Medical School on X:
“1L Melanoma PD1 vs PD1+LAG3 from is (-) another setback for the IO/ICI field. We keep pressing. Some interesting trends and signals.”
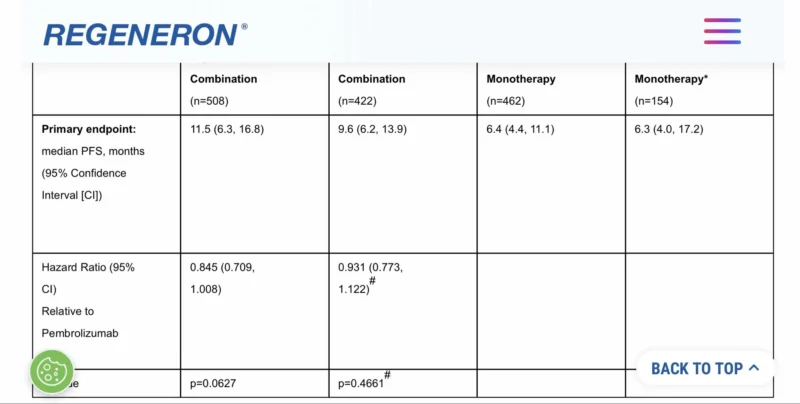
The failure of Regeneron’s Phase 3 first-line advanced melanoma trial evaluating the anti-LAG–3 antibody fianlimab in combination with cemiplimab (anti-PD-1) represents a significant setback in managing adaptive resistance.
In the tumor microenvironment, chronic antigen exposure drives cytotoxic T cells into terminal exhaustion. While anti-PD-1 monotherapy temporarily rejuvenates these cells, it frequently triggers a compensatory up-regulation of alternative inhibitory receptors, most notably LAG-3, TIGIT, and TIM-3. Dual checkpoint blockade aims to synergistically reverse this exhaustion by simultaneously disrupting non-redundant inhibitory pathways.
A Decade of Alternative Checkpoint Failures
Despite clear preclinical synergy and the clinical validation of Bristol Myers Squibb’s dual anti-PD-1/anti-LAG-3 combination (Opdualag), translating alternative checkpoint targets into Phase 3 success has proven exceptionally difficult over the last ten years.
Regeneron‘s combination failed to achieve statistical significance for its primary endpoint of Progression-Free Survival (PFS), joining a long list of high-profile Phase 3 misses across the anti-TIGIT and anti-TIM-3 classes.
These failures underscore the immense complexity of the tumor microenvironment. For clinicians, the data demonstrate that simply adding a second antibody to block co-expressed inhibitory receptors is not a guaranteed mechanism to overcome primary or adaptive anti-PD-1 resistance.
Other articles about Fianlimab on OncoDaily.