At the European Lung Cancer Congress 2026, the phase III LATIFY trial evaluated a biologically driven combination strategy in a challenging treatment setting: patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) who had progressed after both PD-(L)1 inhibitors and platinum-based chemotherapy.
Ceralasertib, a selective inhibitor of the ATR (ataxia telangiectasia and Rad3-related) DNA damage response kinase, was combined with durvalumab, an anti–PD-L1 antibody, with the rationale that targeting DNA repair pathways could enhance tumor immunogenicity and restore sensitivity to immunotherapy.

Study Design and Patient Population
LATIFY was a randomized, open-label, multicenter phase III trial enrolling 594 patients with locally advanced or metastatic NSCLC without actionable genomic alterations. All patients had progressed on or after prior PD-(L)1 inhibitor therapy and platinum-based chemotherapy.
Participants were randomized 1:1 to receive:
- Ceralasertib (240 mg orally, twice daily on Days 1–7) + durvalumab (1500 mg IV on Day 8 of a 28-day cycle)
- Docetaxel (75 mg/m² IV every 3 weeks)
Treatment continued until disease progression or unacceptable toxicity.
The primary endpoint was overall survival (OS), with key secondary endpoints including progression-free survival (PFS), objective response rate (ORR), and safety.
Baseline characteristics were well balanced between treatment arms, and the median follow-up was approximately 19–20 months.

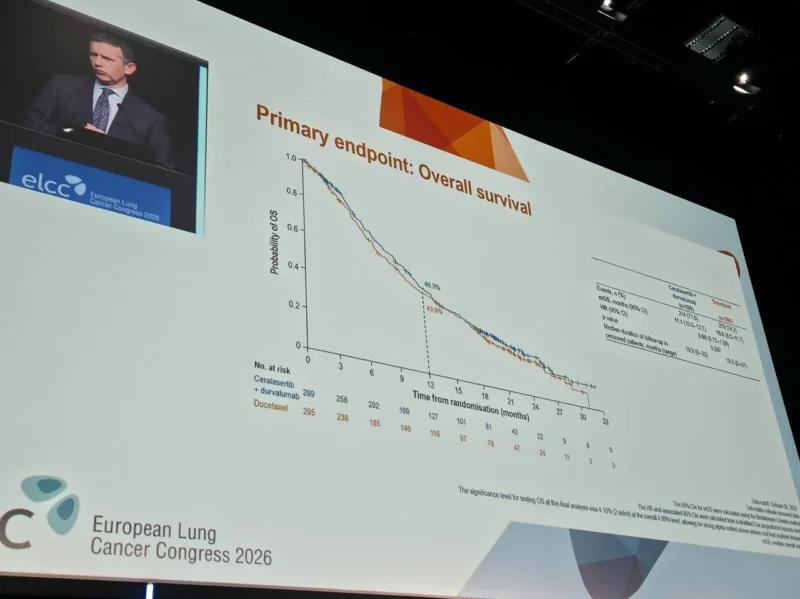
Overall Survival: No Significant Benefit
At the final analysis, the combination of ceralasertib plus durvalumab did not demonstrate a statistically significant improvement in overall survival compared with docetaxel.
- Median OS: 11.1 months (C+D) vs 10.0 months (docetaxel)
- Hazard ratio: 0.90 (95% CI, 0.75–1.09; p = 0.287)
- 12-month OS rate: 46.3% vs 43.6%
While there was a numerical trend favoring the experimental arm, the difference did not reach statistical significance, and survival outcomes remained broadly comparable between groups.
Importantly, OS results were consistent across predefined subgroups, suggesting no clear population that derived a differential survival benefit.
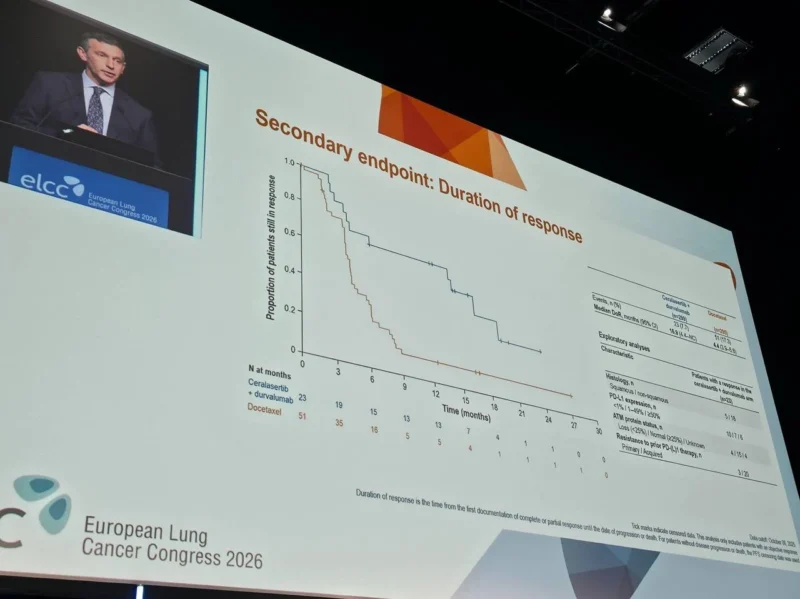
While ceralasertib plus durvalumab did not improve survival outcomes, responses appeared more durable, with a median duration of response of 16.9 months compared to 4.4 months with docetaxel.
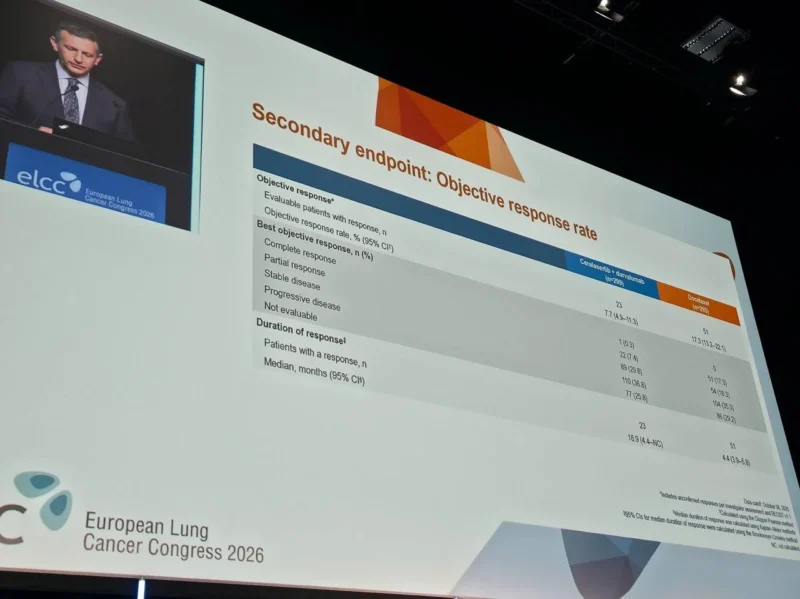
Progression-Free Survival and Response Rates
Similarly, no meaningful improvements were observed in progression-free survival.
- Median PFS: 4.1 months in both arms
- Hazard ratio: 0.87 (95% CI, 0.73–1.04; p = 0.133)
- 6-month PFS rate: 35.3% vs 33.5%
Objective response rates were, in fact, higher in the docetaxel arm:
- ORR: 7.7% with ceralasertib + durvalumab
- 17.3% with docetaxel
These findings indicate that the addition of ATR inhibition to PD-L1 blockade did not translate into improved tumor shrinkage or disease control in this setting.
Safety Profile: Improved Tolerability
Despite the lack of efficacy benefit, the combination demonstrated a more favorable safety profile compared with docetaxel.
Grade ≥3 treatment-related adverse events:
- 26.9% (C+D) vs 43.8% (docetaxel)
Treatment discontinuation due to adverse events:
- 7.7% vs 11.3%
Overall, adverse events were consistent with the known safety profiles of each agent, and no unexpected safety signals were identified.
The improved tolerability may be clinically relevant, particularly for patients unable to tolerate cytotoxic chemotherapy, although this must be weighed against the lack of efficacy advantage.
Biological Rationale vs Clinical Reality
The LATIFY trial was grounded in a strong biological hypothesis. ATR inhibition is thought to increase DNA damage, enhance tumor antigen presentation, and potentially sensitize tumors to immune checkpoint blockade.
However, the results highlight a recurring challenge in oncology drug development: promising preclinical rationale does not always translate into clinical benefit.
In this case, combining ceralasertib with durvalumab did not overcome resistance to prior immunotherapy, nor did it improve key clinical outcomes compared with standard chemotherapy.
Clinical Implications
For patients with advanced NSCLC who progress after PD-(L)1 inhibitors and chemotherapy, treatment options remain limited. Docetaxel continues to represent a standard of care, despite modest efficacy.
The LATIFY results indicate that:
The ceralasertib plus durvalumab combination should not replace docetaxel in this setting
ATR inhibition, at least in this context, does not provide sufficient clinical benefit
Future strategies may need to focus on better patient selection, alternative combinations, or different mechanisms of resistance
Conclusion
The phase III LATIFY trial demonstrates that ceralasertib plus durvalumab does not significantly improve survival outcomes compared with docetaxel in patients with previously treated advanced NSCLC.
While the combination showed manageable toxicity and improved tolerability, this did not translate into meaningful clinical benefit.
These findings underscore the continued need for more effective post-immunotherapy treatment strategies and highlight the importance of validating biological hypotheses in large, randomized trials.


