KRAS mutations are among the most common oncogenic drivers in lung cancer, yet not all KRAS subtypes have been equally targetable. While KRAS G12C inhibitors have recently changed the treatment landscape, KRAS G12D-Mutant NSCLC remains a major unmet need, affecting approximately 5% of patients and lacking any approved targeted therapies.
At European Lung Cancer Congress 2026, new data from a first-in-human phase I study introduce setidegrasib, a novel protein degrader designed to selectively target KRAS G12D. This approach represents a shift from conventional inhibition toward oncoprotein degradation, offering a new therapeutic strategy in KRAS G12D-Mutant NSCLC.

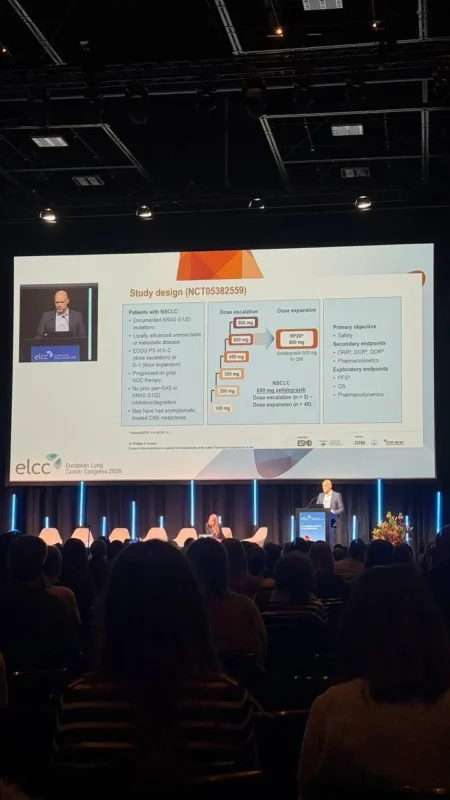
Study Design and Patient Population
This phase I study (NCT05382559), conducted by Astellas Pharma, evaluated setidegrasib at a dose of 600 mg administered intravenously once weekly, identified as the recommended phase II dose.
Patients enrolled had locally advanced or metastatic solid tumors harboring KRAS G12D mutations, including a dedicated cohort of individuals with KRAS G12D-Mutant NSCLC. All patients had received prior standard-of-care treatments, making this a heavily pretreated population with limited therapeutic options.
The NSCLC cohort included 45 patients, with a median of two prior lines of therapy. A notable proportion were light or non-smokers, and more than one-third of evaluable tumors were PD-L1 negative, reflecting a population that often derives limited benefit from immunotherapy.
Key Efficacy Results
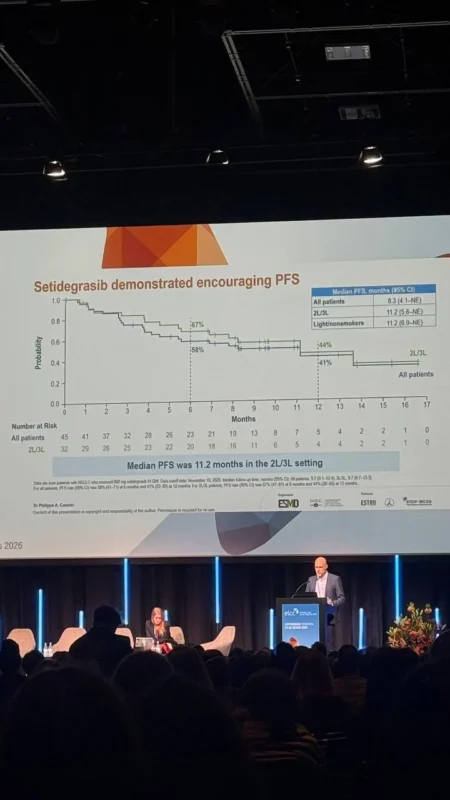
Setidegrasib demonstrated encouraging antitumor activity in KRAS G12D-Mutant NSCLC, particularly given the refractory nature of the population studied.
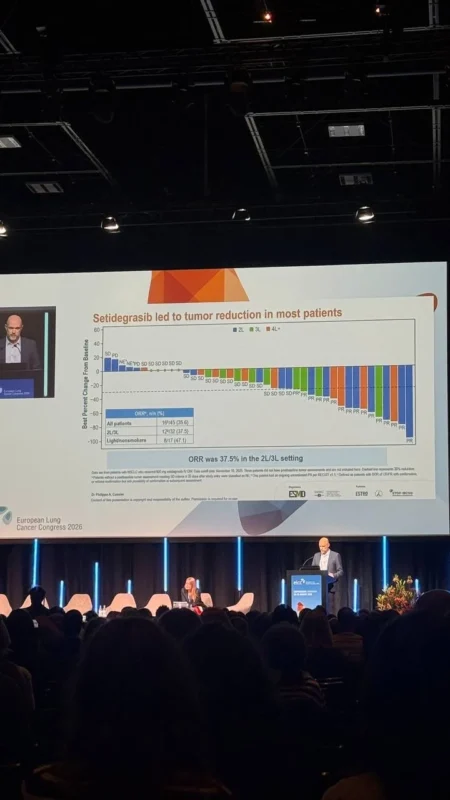
The objective response rate reached 35.6%, indicating that more than one-third of patients experienced meaningful tumor shrinkage. In addition, the disease control rate was 84.4%, suggesting that the majority of patients derived clinical benefit, either through response or disease stabilization.
Importantly, responses appeared durable. The median duration of response was not reached at the time of analysis, with 75.7% of responses ongoing at six months and 50.4% at one year. These findings are particularly notable in KRAS G12D-Mutant NSCLC, where durable responses have historically been difficult to achieve.
Progression-free survival further supports the activity of this agent. Median PFS was 8.3 months, with improved outcomes observed in certain subgroups. Patients who had received prior platinum-based chemotherapy and immune checkpoint inhibitors demonstrated a median PFS of 11.2 months, suggesting that setidegrasib retains activity even after standard therapies have failed.
Additionally, light or non-smokers appeared to derive greater benefit, with response rates approaching 47% and disease control rates exceeding 90%, although these findings require further validation in larger cohorts.
Safety Profile
Setidegrasib demonstrated a manageable and favorable safety profile, which is critical in a heavily pretreated population.
The most common treatment-related adverse events were infusion-related reactions, occurring in approximately 77.8% of patients. These events were exclusively grade 1–2 and typically presented as rash, pruritus, or urticaria after the first dose, with decreased frequency in subsequent administrations.
Other commonly observed adverse events included nausea and elevations in liver enzymes. Grade 3 or higher treatment-related adverse events occurred in 13.3% of patients, and serious adverse events were reported in 6.7%.
Importantly, no treatment-related adverse events led to discontinuation or death, supporting the tolerability of this novel therapeutic approach.
Mechanism of Action and Clinical Implications
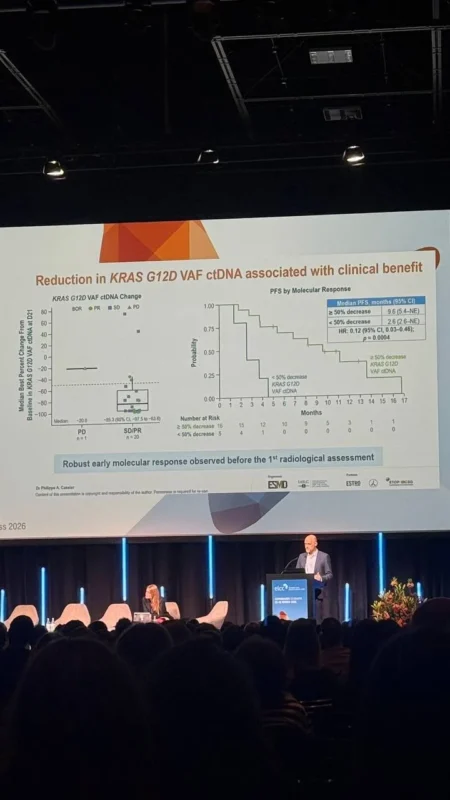
Setidegrasib represents a first-in-class protein degrader targeting KRAS G12D, distinguishing it from traditional small-molecule inhibitors. Instead of blocking KRAS signaling, this strategy promotes degradation of the mutant KRAS protein itself, potentially leading to more sustained pathway suppression.
Preclinical and translational data presented alongside this study indicate that setidegrasib effectively suppresses KRAS signaling and may also induce tumor immunomodulatory effects, raising the possibility of future combination strategies with immunotherapy or other targeted agents.
This mechanism is particularly relevant in KRAS G12D-Mutant NSCLC, where resistance to existing therapies and lack of direct targeting options have limited progress.
Key Takeaway
Setidegrasib demonstrates clinically meaningful activity and a manageable safety profile in KRAS G12D-Mutant NSCLC, a population with no approved targeted therapies to date.
The observed response rates, durability of benefit, and tolerability profile support further clinical development and highlight the potential of protein degradation as a novel therapeutic strategy in lung cancer.
If confirmed in later-phase trials, this approach could represent a major step forward in precision oncology for KRAS G12D-Mutant NSCLC, addressing a long-standing gap in targeted treatment.
Read full abstract here


