Long-term outcomes remain a critical question in early-stage non-small cell lung cancer (NSCLC), particularly for patients who achieve a pathological complete response (pCR) after neoadjuvant therapy. While pCR is widely considered a strong prognostic marker, its durability over time and interaction with immunotherapy strategies require deeper evaluation.
New data from the phase III KEYNOTE-671 trial provide important insights into this question, demonstrating that perioperative pembrolizumab continues to deliver sustained benefit even among patients who already achieve pCR.

Background
The KEYNOTE-671 study previously established perioperative pembrolizumab combined with neoadjuvant chemotherapy as a new standard approach in resectable early-stage NSCLC. The regimen significantly improved event-free survival (EFS), overall survival (OS), major pathological response, and pCR rates compared with chemotherapy alone.
However, an important clinical question remained:
Does pembrolizumab continue to provide benefit in patients who already achieve pCR?
This updated 5-year analysis focuses specifically on that subgroup.
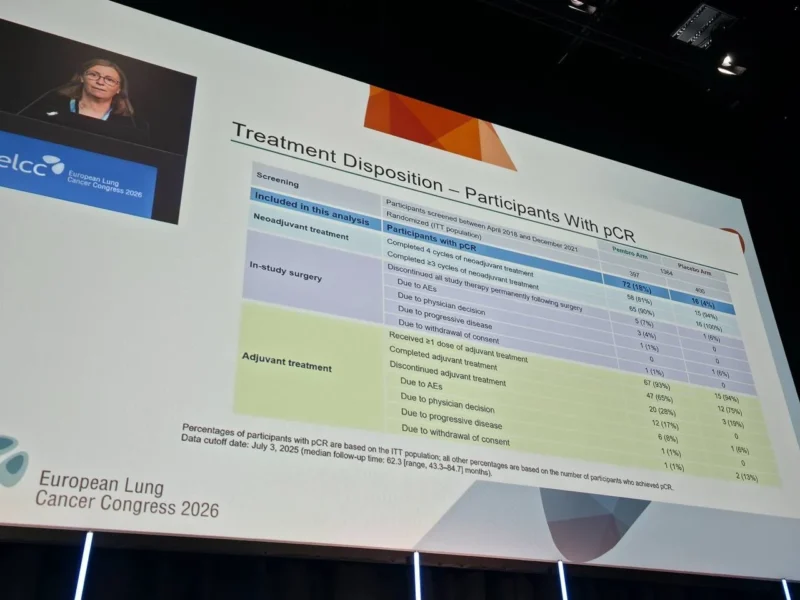
Methods
This randomized, phase III trial enrolled patients with previously untreated, resectable stage II to IIIB (N2) NSCLC. Participants were assigned in a 1:1 ratio to receive:
- Pembrolizumab plus neoadjuvant chemotherapy
- Placebo plus neoadjuvant chemotherapy
This was followed by surgery and adjuvant pembrolizumab or placebo for up to one year.
The primary endpoint remained event-free survival, with pCR assessed centrally by blinded pathology review.
Results
Pathological Complete Response Rates At the time of analysis, pCR was achieved in:
- 18.1% of patients in the pembrolizumab arm
- 4.0% in the placebo arm
This represents a substantial absolute improvement of 14.2%, reinforcing the strong impact of perioperative immunotherapy on tumor eradication.
Importantly, the benefit of pembrolizumab in achieving pCR was consistent across all baseline subgroups, including disease stage, nodal involvement, and PD-L1 expression.
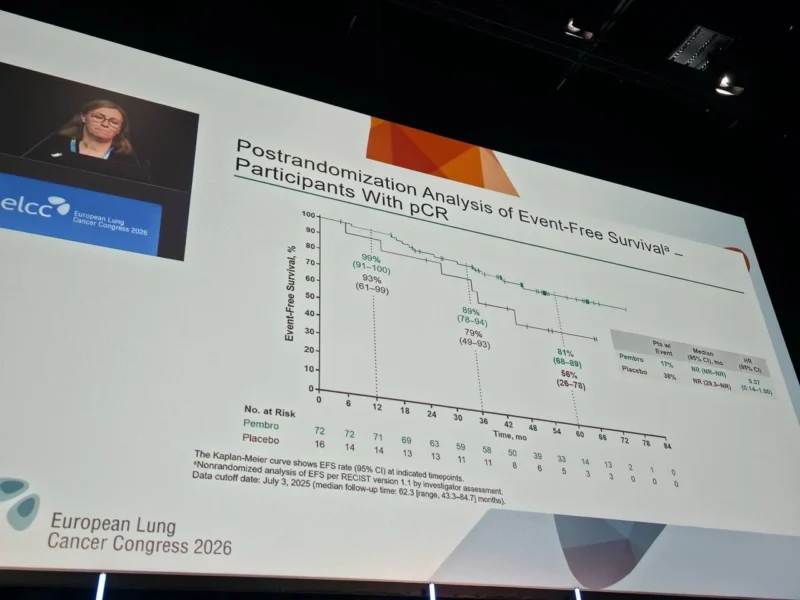
Long-Term Event-Free Survival
With a median follow-up exceeding 5 years, the durability of benefit becomes clear.
Among patients who achieved pCR:
- 5-year EFS was 80.8% with pembrolizumab
- 5-year EFS was 55.7% with placebo
This corresponds to a hazard ratio of 0.37, indicating a 63% reduction in the risk of events, even in this already favorable subgroup.
These findings are particularly important because they challenge the assumption that achieving pCR alone is sufficient and that additional therapy may not provide further benefit.
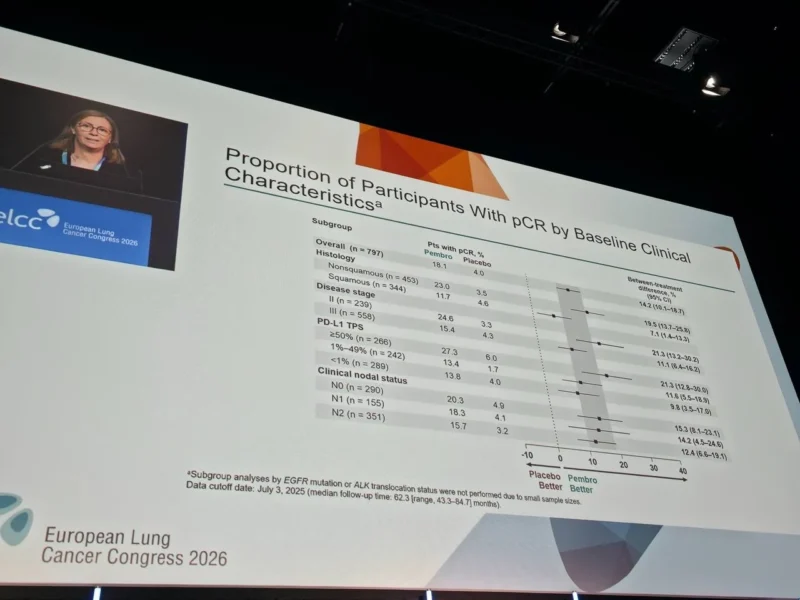
Subgroup Insights
The increase in pCR rates with pembrolizumab was observed across clinically relevant subgroups:
- Stage II disease: pCR 24.6% vs 3.3%
- Stage III disease: pCR 15.4% vs 4.3%
- PD-L1 ≥50%: pCR 27.3% vs 6.0%
- PD-L1 <1%: pCR 13.8% vs 4.0%
This consistency highlights that the benefit of perioperative immunotherapy extends beyond biomarker-selected populations and supports broad applicability in clinical practice.
Clinical Interpretation
This analysis delivers a key message:
Even among patients who achieve pCR, perioperative pembrolizumab continues to provide meaningful long-term benefit.
Traditionally, pCR has been viewed as a surrogate for excellent outcomes, often associated with reduced recurrence risk. However, these results suggest that:
- pCR does not fully eliminate recurrence risk
- Immunotherapy may enhance durability of response
- Continued systemic therapy contributes to long-term disease control
In other words, pembrolizumab is not only increasing pCR rates, it is deepening and sustaining the benefit of pCR.
Implications for Practice
These findings further strengthen the role of perioperative immunotherapy in early-stage NSCLC. They suggest that:
- Achieving pCR should not lead to de-escalation of systemic therapy prematurely
- Perioperative pembrolizumab provides additive benefit beyond tumor eradication
- Treatment strategies should focus on both achieving and maintaining remission
This represents a shift from viewing pCR as an endpoint to understanding it as part of a broader therapeutic continuum.
Conclusion
The 5-year results from KEYNOTE-671 confirm that perioperative pembrolizumab not only increases pCR rates but also significantly improves long-term outcomes in patients who achieve pCR.
These data reinforce the paradigm that immunotherapy plays a critical role across the entire treatment course, from neoadjuvant to adjuvant settings, and should remain a cornerstone of care in resectable early-stage NSCLC.
As long-term survival continues to improve, the focus in lung cancer is clearly evolving, from achieving response to ensuring its durability.
Read full abstract here


