The treatment landscape of non-small cell lung cancer (NSCLC) has evolved rapidly with the development of targeted therapies. However, patients with EGFR exon 20 insertion NSCLC continue to represent a challenging subgroup, often associated with limited response to conventional EGFR tyrosine kinase inhibitors and poorer clinical outcomes.
For patients who have already received platinum-based chemotherapy, treatment options remain particularly limited, highlighting the need for novel therapeutic strategies.
At the European Lung Cancer Congress 2026, final overall survival results from the phase II BECOME study provide important insights into a combination approach using becotarug and osimertinib in this difficult-to-treat population.

Study Design and Treatment Strategy
The BECOME study evaluated the combination of becotarug, an anti-EGFR monoclonal antibody, with osimertinib in patients with platinum-pretreated EGFR exon 20 insertion-positive NSCLC.
Patients received becotarug at a dose of 6 mg/kg every two weeks alongside osimertinib 160 mg once daily. The analysis focused on overall survival outcomes, as well as subgroup analyses based on insertion location and baseline circulating tumor DNA (ctDNA) status.
A total of 112 patients were included in the intention-to-treat population, with a median follow-up of 26.7 months, allowing for a meaningful assessment of long-term outcomes.

Overall Survival Outcomes
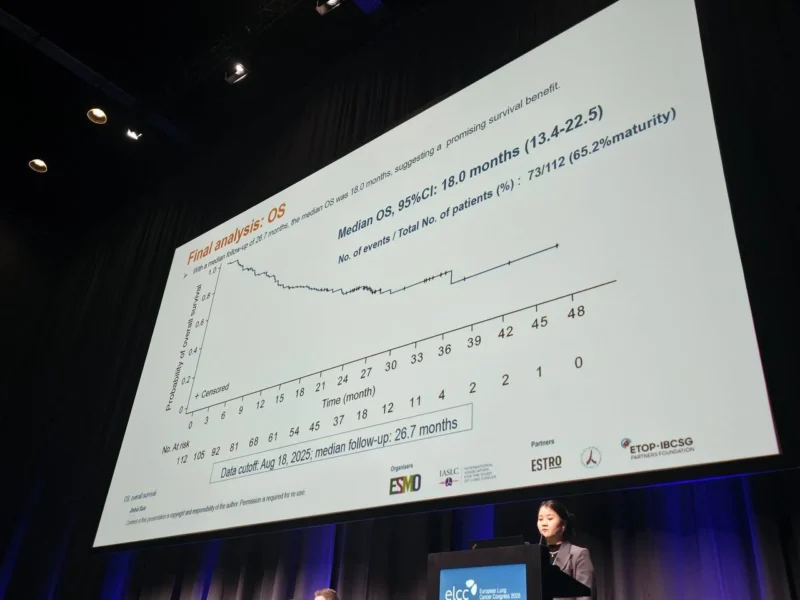
The combination of becotarug and osimertinib demonstrated encouraging survival outcomes in this heavily pretreated population.
Median overall survival reached 18.0 months, suggesting clinically meaningful benefit in a setting where treatment options are limited. These findings reinforce the potential role of targeted combination strategies in improving outcomes for patients with EGFR exon 20 insertion mutations.
Impact of Mutation Subtypes
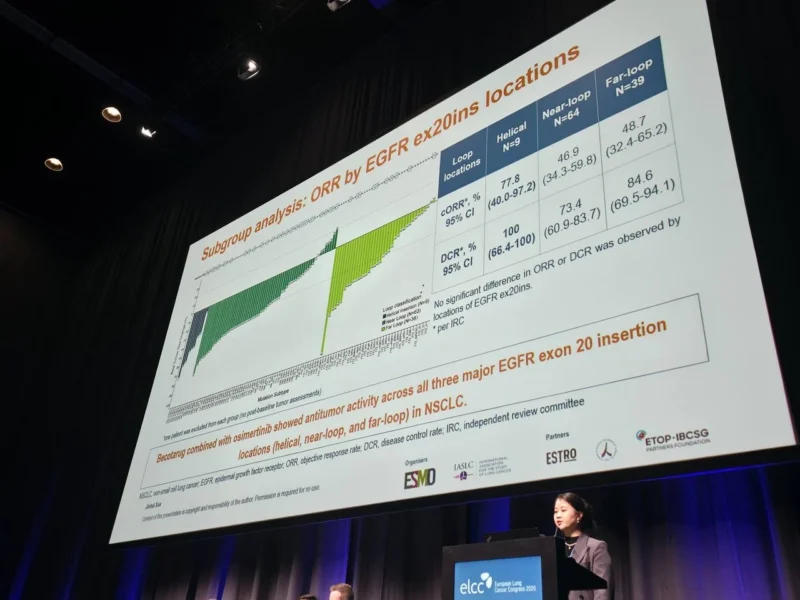
A key aspect of this analysis was the evaluation of outcomes based on the location of EGFR exon 20 insertions.
Patients were categorized into three groups: helical insertions, near-loop insertions, and far-loop insertions. Differences in clinical outcomes were observed across these subgroups, highlighting the biological heterogeneity within EGFR exon 20 insertion NSCLC.
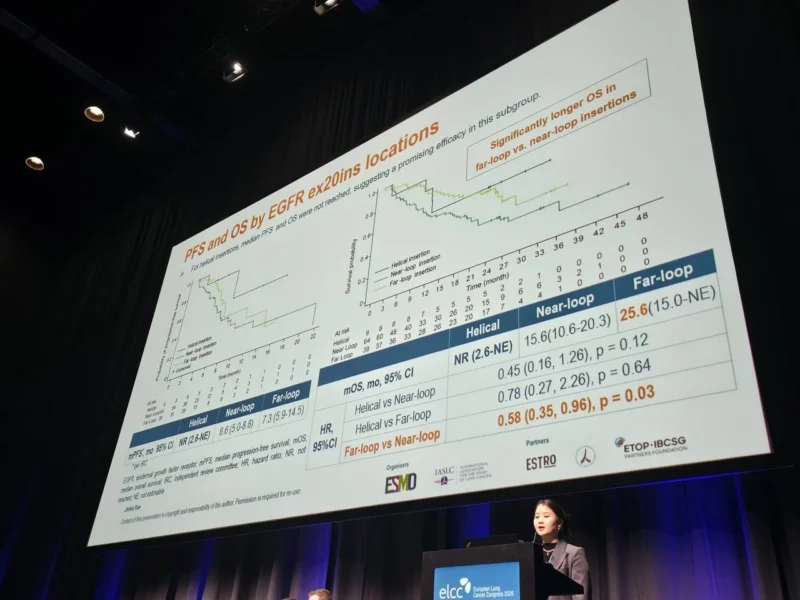
Notably, patients with far-loop insertions demonstrated significantly improved overall survival compared with those with near-loop insertions, with a hazard ratio of 0.58 and a statistically significant difference. Median overall survival in this subgroup reached 25.6 months, compared with 15.6 months in patients with near-loop insertions.
These findings suggest that insertion location may serve as an important predictive factor for treatment response and survival outcomes, emphasizing the need for more refined molecular stratification in clinical practice.
Role of ctDNA as a Prognostic Marker
The study also explored the impact of baseline circulating tumor DNA status on treatment outcomes.
Among patients with available paired tissue and blood samples, differences in survival outcomes were observed between ctDNA-positive and ctDNA-negative groups.
Patients with ctDNA-negative disease demonstrated substantially longer progression-free survival and overall survival compared with those who were ctDNA-positive. Median progression-free survival reached 14.5 months in ctDNA-negative patients, compared with 6.9 months in ctDNA-positive patients. Similarly, median overall survival was 36.6 months versus 19.2 months, respectively, with statistically significant differences.
While response rates and disease control rates did not differ significantly between groups, these findings indicate that ctDNA status may provide valuable prognostic information, potentially identifying patients more likely to achieve durable benefit from this combination therapy.
Clinical Implications
The results from the BECOME study highlight several important considerations for clinical practice.
First, the combination of becotarug and osimertinib demonstrates meaningful survival benefit in a population with limited treatment options, supporting further investigation of this approach.
Second, the observed differences in outcomes based on insertion subtype underscore the importance of molecular heterogeneity within EGFR exon 20 insertion NSCLC.
Third, the strong prognostic impact of ctDNA status suggests that liquid biopsy may play an increasingly important role in guiding treatment decisions and risk stratification.
Together, these findings move the field closer toward a more personalized approach to treating EGFR exon 20 insertion NSCLC.
Key Takeaway
The phase II BECOME study demonstrates that becotarug plus osimertinib provides clinically meaningful survival benefit in platinum-pretreated EGFR exon 20 insertion NSCLC, with outcomes influenced by mutation subtype and ctDNA status.
These results highlight the potential of combination targeted therapy and reinforce the importance of molecular profiling in optimizing treatment strategies for this challenging patient population.
Read full abstract here


