Beamion LUNG-1 Trial , presented by John Heymach (Houston, TX, United States of America) during ELCC, reported efficacy and safety results for zongertinib in treatment-naïve patients with HER2-mutant NSCLC and in patients with active brain metastases, including those with no prior brain radiotherapy.
Background
Zongertinib is described in the abstract as an oral, irreversible tyrosine kinase inhibitor that selectively inhibits HER2 while sparing wild-type EGFR, with the aim of minimizing EGFR-related toxicities. In this phase Ib analysis of Beamion LUNG-1, investigators evaluated zongertinib in two important groups: treatment-naïve patients with HER2-mutant NSCLC and patients with active brain metastases, a population often underrepresented in clinical trials.
Methods
Patients with advanced or metastatic non-squamous NSCLC harboring a HER2 mutation within the tyrosine kinase domain received zongertinib 120 mg once daily.
The abstract reports results from two cohorts. Cohort 2 included treatment-naïve patients, and its primary endpoint was objective response defined as best overall complete or partial response by RECIST v1.1. Secondary endpoints included duration of response and progression-free survival.
Cohort 4 included patients who were either treatment-naïve or previously treated and had active, symptomatic brain metastases. The primary endpoint in this cohort was objective response in CNS lesions assessed by RANO-BM. All endpoints were evaluated by blinded independent central review.
Results
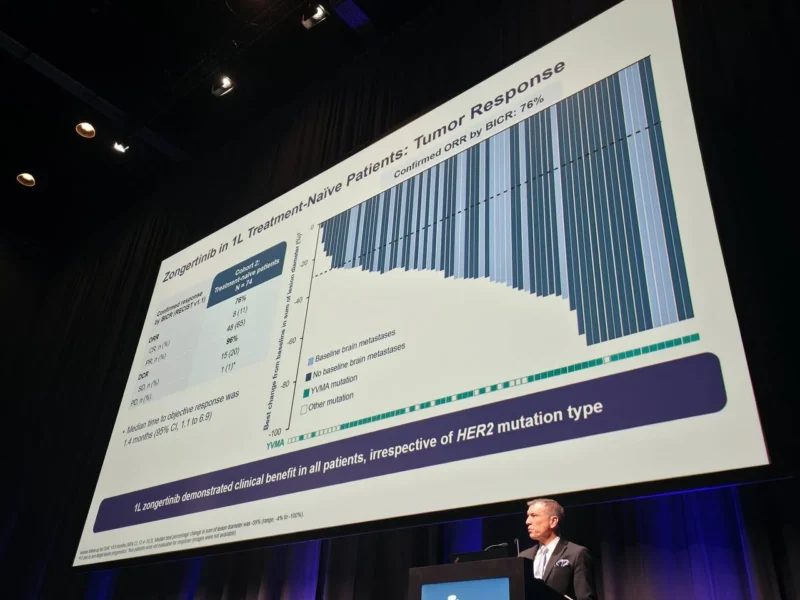
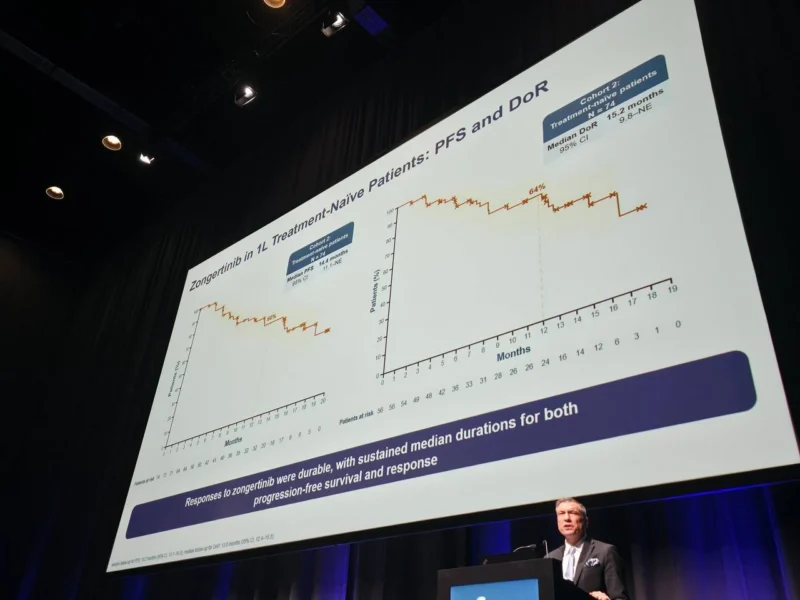
As of August 21, 2025, 74 patients had received zongertinib 120 mg in cohort 2. The confirmed objective response rate was 76% with a 95% confidence interval of 65% to 84%. Median duration of response was 15.2 months with a 95% confidence interval of 9.8 months to not evaluable, and median progression-free survival was 14.4 months with a 95% confidence interval of 11.1 months to not evaluable.
Treatment-related adverse events occurred in 91% of patients in cohort 2, with grade 3 or higher events in 19%. These adverse events were reported to be mainly low-grade diarrhea.
In cohort 4, 30 patients received zongertinib 120 mg. The confirmed intracranial objective response rate by RANO-BM was 47% with a 95% confidence interval of 30% to 64%. Median intracranial duration of response was 6.9 months with a 95% confidence interval of 2.9 months to not evaluable, and median intracranial progression-free survival was 8.2 months with a 95% confidence interval of 4.1 to 11.3 months.
Among the 8 treatment-naïve patients in cohort 4, the confirmed intracranial ORR was 50%, corresponding to 4 patients, with a 95% confidence interval of 22% to 79%.
Among 17 patients with centrally confirmed measurable CNS disease and no prior brain radiotherapy, the confirmed intracranial ORR was 59% with a 95% confidence interval of 36% to 78%. In this subgroup, median intracranial duration of response was 6.2 months with a 95% confidence interval of 2.7 months to not evaluable.
Conclusion
According to the abstract, zongertinib showed durable systemic and intracranial efficacy in treatment-naïve patients with advanced or metastatic HER2-mutant NSCLC. The study also showed notable intracranial efficacy in patients with active brain metastases, including those without prior brain radiotherapy, a population the abstract notes is commonly excluded from trials. The reported treatment-related adverse events were mostly low-grade.