ASTEROID trial findings presented at the European Lung Cancer Congress 2026 highlight an evolving strategy in early-stage non-small cell lung cancer (NSCLC). In patients unfit for surgery, treatment is commonly based on stereotactic body radiotherapy (SBRT), which provides excellent local control. However, despite these favorable local outcomes, a significant proportion of patients eventually develop distant metastases, underscoring a persistent unmet need in this setting.
In this context, combining radiotherapy with immunotherapy has emerged as a promising approach. The rationale lies in the potential synergistic interaction between radiation-induced tumor antigen release and immune checkpoint inhibition, which may enhance systemic antitumor responses.
At the European Lung Cancer Congress 2026, results from the randomized phase II ASTEROID trial evaluated whether adding durvalumab after SBRT could improve outcomes in patients with early-stage NSCLC treated with SBRT and durvalumab.

Study Design and Patient Population
The ASTEROID trial is a randomized, multicenter, open-label phase II study comparing SBRT alone with SBRT followed by durvalumab in patients with peripheral T1–T2N0M0 NSCLC.
Patients were randomized to receive either standard SBRT delivered in 3–4 fractions or SBRT followed by durvalumab 1500 mg administered every four weeks for up to 12 months. The primary endpoint of the study was time to progression, while disease-free survival and overall survival were secondary endpoints.
A total of 104 patients were enrolled between 2018 and 2024 across 12 Nordic centers. The study population reflects a typical real-world cohort of patients receiving SBRT, with a median age of 78 years and a predominance of early-stage tumors. Most patients had adenocarcinoma histology, and the majority had T1A tumors.

Key Efficacy Results
The addition of immunotherapy demonstrated a signal toward improved tumor control in early-stage NSCLC treated with SBRT and durvalumab.
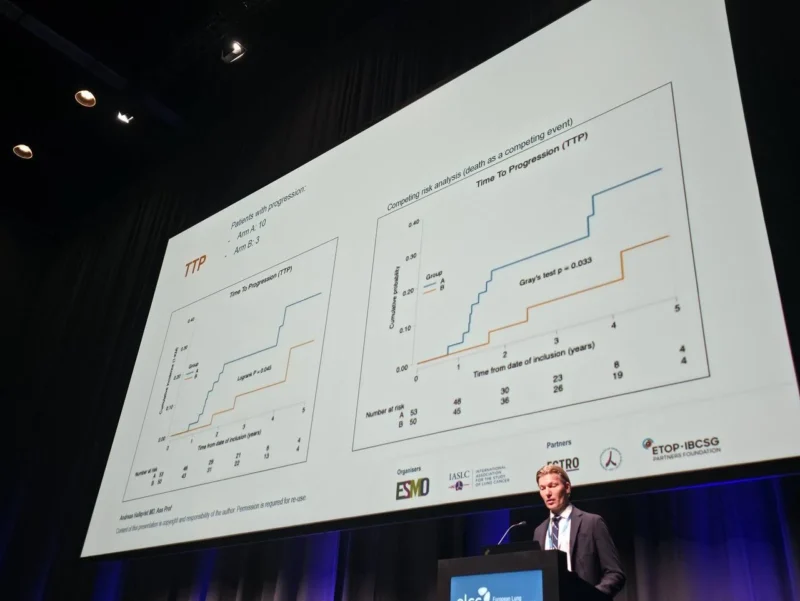
At a median follow-up of 33 months, fewer progression events were observed in the combination arm compared with SBRT alone. Specifically, 10 patients in the SBRT-only group experienced progression, compared with 3 patients in the SBRT plus durvalumab group, translating into a statistically significant improvement in time to progression (p=0.045, based on a predefined one-sided alpha level).
This finding suggests that adding durvalumab may reduce the risk of disease progression, potentially delaying recurrence in patients treated with SBRT.
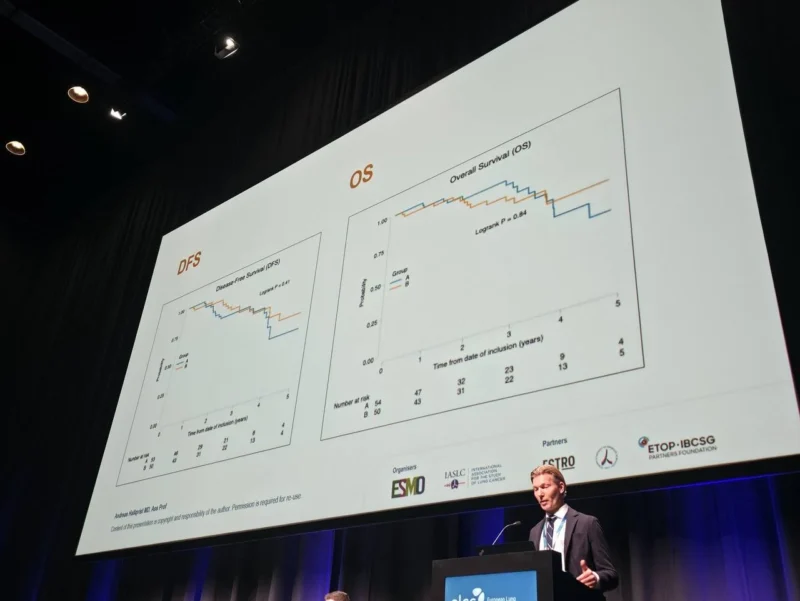
However, despite this improvement in tumor control, no differences were observed in disease-free survival or overall survival between the two treatment arms at the time of analysis. This highlights the complexity of treating this patient population, where competing risks such as age and comorbidities may influence long-term outcomes.
Safety and Tolerability
The safety profile of the combination was consistent with expectations for SBRT and immunotherapy.
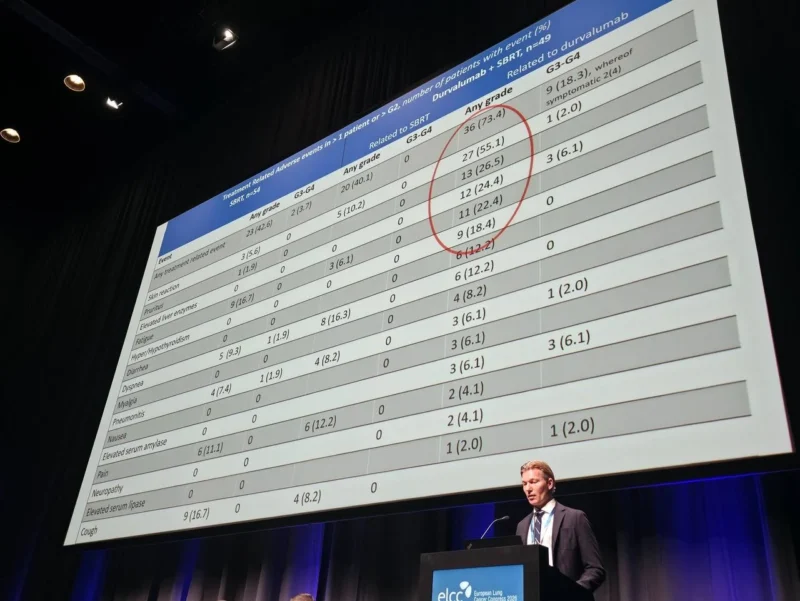
Adverse events related to SBRT alone were observed in 41% of patients, primarily consisting of low-grade symptoms such as fatigue, cough, pain, and dyspnea. Only one case of grade 3 pneumonitis was reported in the SBRT-only arm.
In the combination arm, adverse events related to durvalumab were reported in 73% of patients, with most events being grade 1 or 2. The most common toxicities included skin reactions, pruritus, and fatigue.
Two patients in the combination arm experienced grade 3 adverse events, including skin toxicity and pneumonitis. These findings suggest that while the addition of immunotherapy increases the incidence of adverse events, the overall safety profile remains manageable in a carefully selected population.
Clinical Implications
The ASTEROID trial provides important insight into the potential role of immunotherapy in early-stage NSCLC treated with SBRT and durvalumab.
While SBRT alone achieves excellent local control, it does not address the risk of distant relapse. The addition of durvalumab appears to enhance systemic disease control, as reflected by improved time to progression.
However, the absence of a survival benefit at this stage underscores the need for cautious interpretation. Patients included in this study were older and often had significant comorbidities, which may limit the ability to detect differences in overall survival.
These findings suggest that patient selection will be critical in determining who may benefit most from this approach. Future studies may focus on identifying biomarkers or clinical characteristics that predict response to combined radiotherapy and immunotherapy.
Key Takeaway
The phase II ASTEROID trial demonstrates that early-stage NSCLC treated with SBRT and durvalumab results in improved time to progression compared with SBRT alone, indicating enhanced tumor control.
Numerically fewer progression events were observed with durvalumab (3 vs 10), supporting improved tumor control despite no difference in DFS or OS.
However, the lack of improvement in survival endpoints highlights the need for further research to clarify the clinical value of this strategy in this population.
You can read full abstract here


