ADEPPT Trial , presented by Jarushka Naidoo (Dublin, Ireland) at ELCC, explored the efficacy, safety, and quality-of-life outcomes of adagrasib in underrepresented populations with pretreated KRAS G12C-mutant non-small cell lung cancer, including elderly patients and those with ECOG performance status 2.
Background
The phase II ADEPPT trial evaluated adagrasib in patients with pretreated KRAS G12C-mutant non-small cell lung cancer from underrepresented populations often not well represented in earlier studies. These included elderly patients and patients with ECOG performance status 2. The study assessed efficacy, toxicity, and quality of life in these two groups.
Background
KRAS G12C mutations occur in about 10% to 15% of non-small cell lung cancer cases, identifying a clinically relevant subgroup for targeted treatment. Adagrasib, a second-generation KRAS G12C inhibitor, has already demonstrated activity in pretreated NSCLC, particularly in patients with ECOG performance status 0–1. Still, this does not fully reflect everyday practice, where many patients are older or have ECOG PS 2 and are therefore often underrepresented in prospective trials.
As a result, evidence on treatment outcomes in these more vulnerable populations remains limited. The ADEPPT study was designed to address this gap by evaluating the efficacy, safety, and quality-of-life impact of adagrasib in elderly patients and in those with ECOG PS 2.
Methods
ADEPPT (NCT05673187) is a prospective, international, phase II trial of adagrasib 600 mg twice daily, given until progression or unacceptable toxicity, in patients with KRAS G12C-mutant pretreated NSCLC.
The trial included two parallel single-arm cohorts. Cohort A enrolled patients aged 70 years or older with ECOG PS 0–1. Cohort B enrolled patients aged 18 years or older with ECOG PS 2.
The primary endpoint was confirmed objective response rate by 12 weeks (cORR) according to RECIST v1.1. A sample size of 34 patients per cohort provided 80% power to reject the null hypothesis of ORR ≤15% versus 35%, with a success criterion of at least 10 responders by 12 weeks, using a one-sided alpha of 2.5% and an exact binomial test.
Secondary endpoints included progression-free survival, overall survival, safety, and quality of life, assessed with the NCCN-Functional Assessment of Cancer Therapy Lung Symptom Index-17.
Results
Between 12 June 2023 and 18 February 2025, 32 of the planned 34 patients were enrolled in each cohort. In cohort A and cohort B, 66% and 62% of patients were male, 97% in both cohorts were former or current smokers, 100% and 91% had adenocarcinoma, and 16% and 32% had baseline brain lesions, respectively. Median follow-up was 16.8 months in cohort A and 18.5 months in cohort B, while mean relative dose intensity reached 72% and 79%.
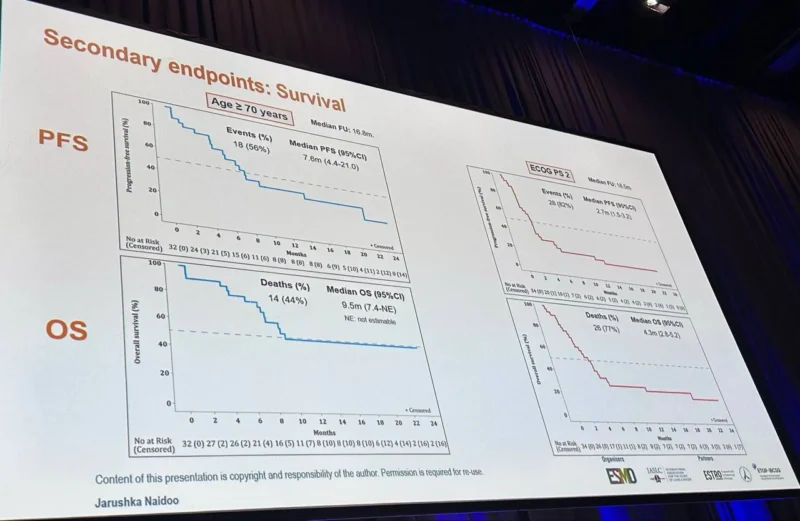
In cohort A, which included elderly patients with ECOG PS 0–1, the study met its primary endpoint. The confirmed objective response rate at 12 weeks was 31%, corresponding to 10 confirmed responses (1 complete and 9 partial). The overall objective response rate was 34%, with a median progression-free survival of 7.6 months and a median overall survival of 9.5 months. The median duration of response was not reached at the time of analysis.
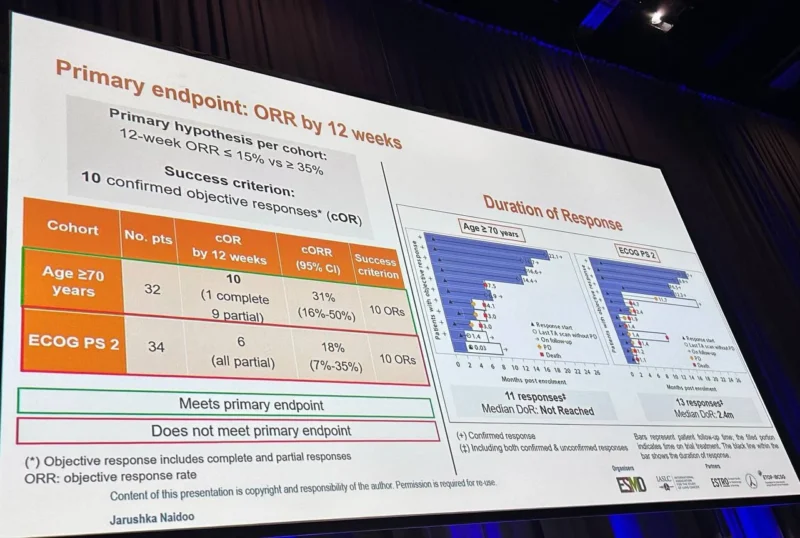
In cohort B, which enrolled patients with ECOG PS 2, the primary endpoint was not met. The confirmed objective response rate at 12 weeks was 18%, with 6 confirmed responses (all partial). The overall objective response rate was 38%, while median progression-free survival was 2.7 months, median overall survival was 4.3 months, and median duration of response was 2.4 months.
Safety And Quality Of Life
Grade 3 or higher treatment-related adverse events were reported in 41% of patients in cohort A and 62% in cohort B. There was one fatal treatment-related adverse event, reported as sudden death NOS, in cohort B.
At 12 weeks, the mean increase in the quality-of-life index from baseline was 2.3 in cohort A and 6.6 in cohort B, based on 18 and 11 patients still on treatment, respectively.
Conclusion
Based on the abstract, adagrasib demonstrated meaningful clinical benefit with a manageable safety profile in elderly patients with ECOG PS 0–1. In patients with ECOG PS 2, activity was described as limited, although quality of life improved.