Brain metastasis remains one of the most clinically difficult events in extensive-stage small cell lung cancer, even as first-line treatment has changed with the addition of immune checkpoint inhibitors. A new prospective cohort study by Wenli Cao and colleagues addresses a question that has become increasingly important in the chemoimmunotherapy era: does adding immunotherapy to chemotherapy reduce the risk of brain metastasis in patients with extensive-stage small cell lung cancer who have no brain involvement at baseline?
Their answer is sobering. In this cohort, chemoimmunotherapy did not significantly lower the cumulative incidence of brain metastasis compared with chemotherapy alone. Instead, prophylactic cranial irradiation remained the only independently validated protective strategy against brain metastasis in this setting (Cao et al., 2026).
Why This Question Matters
Small cell lung cancer is one of the most aggressive thoracic malignancies, and most patients are diagnosed with extensive-stage disease. Brain metastasis is especially relevant in this population because the central nervous system is one of the most common sites of failure. Historically, survival after brain metastasis in small cell lung cancer has been poor, and although systemic treatment has improved overall outcomes, longer survival also creates more opportunity for intracranial progression to emerge over time (Cao et al., 2026; Slotman et al., 2007).
This creates a clinical paradox. Modern chemoimmunotherapy regimens such as those used in IMpower133 and CASPIAN have improved survival in extensive-stage small cell lung cancer, but it has remained unclear whether they also protect against later brain metastasis. Some subgroup and retrospective analyses have suggested delayed intracranial progression or reduced need for cranial radiotherapy, while others have shown no meaningful reduction in new brain metastases. That uncertainty has direct implications for surveillance, prophylactic cranial irradiation, and future CNS-directed strategies (Horn et al., 2018; Paz-Ares et al., 2019; Chen et al., 2022; Lu et al., 2024).
How The Study Was Designed
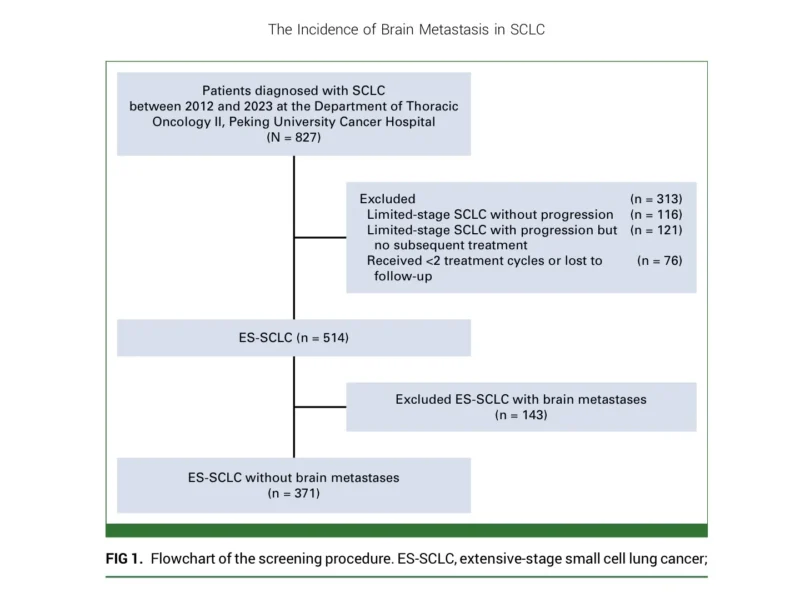
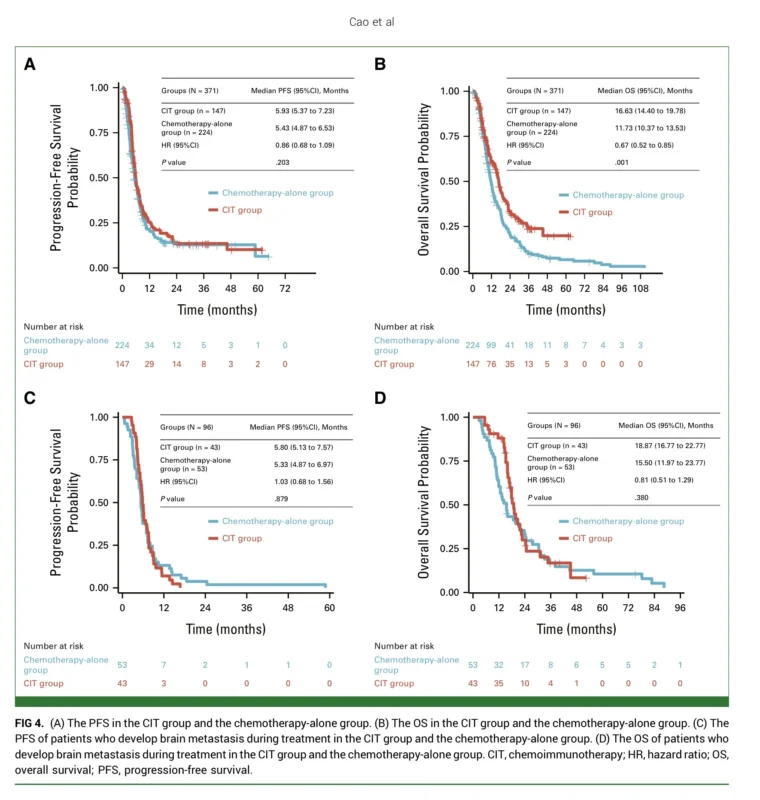
To address this issue more directly, the investigators analyzed a prospective cohort of 371 patients with histologically confirmed extensive-stage small cell lung cancer treated at Peking University Cancer Hospital between 2012 and 2023. All patients had baseline brain imaging showing no brain metastasis and none developed brain involvement within the first two months after diagnosis. This is important because the study was designed specifically to understand new brain metastasis development in patients who started systemic therapy without known intracranial disease (Cao et al., 2026).
Patients were divided into two groups according to first-line treatment. The chemoimmunotherapy group included 147 patients, while 224 received chemotherapy alone. The primary endpoint was cumulative incidence of brain metastasis using competing-risk methods, with death treated as a competing event. Multivariable Fine-Gray regression was used to identify independent risk factors. The authors also performed sensitivity analyses, including inverse probability of treatment weighting, to account for baseline differences and the changing treatment era (Cao et al., 2026).
Brain Metastasis Was Common and Often an Early Event
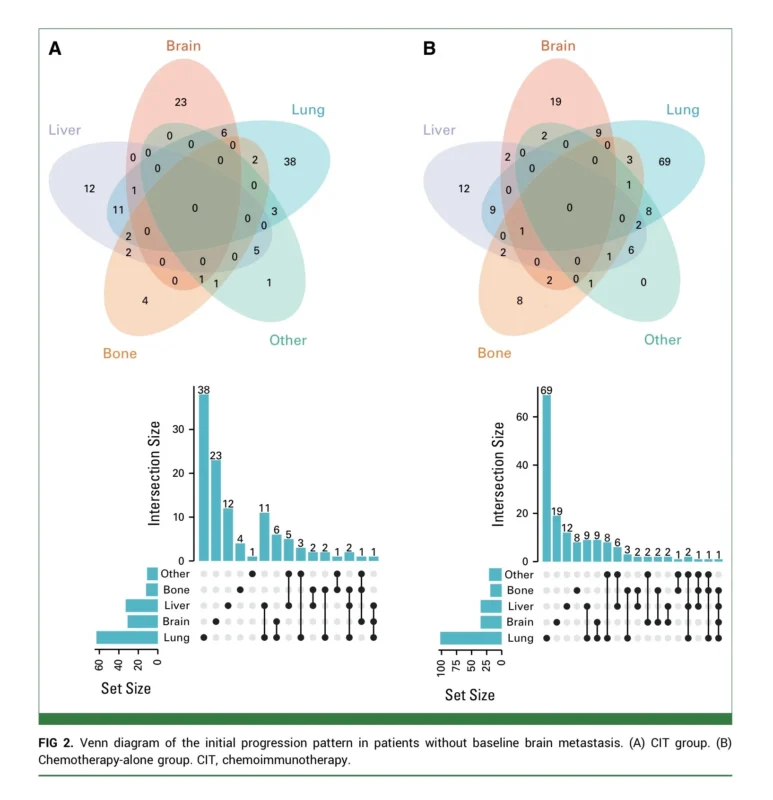
Over a median follow-up of 37.73 months, 96 of 371 patients, or 25.88%, developed brain metastasis. In 17.79% of the full cohort, brain metastasis was the first site of progression, and in 11.32% it was an isolated intracranial event. These numbers matter because they show that the brain is not simply one of several late metastatic sites. In a significant proportion of patients, it is the first clinically meaningful site of failure (Cao et al., 2026).
The pattern of progression was also informative. Lung tumor progression remained common, and liver metastasis as an initial progression event was numerically higher in the chemoimmunotherapy group, although this difference was not statistically significant. Among patients who eventually developed brain metastasis, the number of lesions at first intracranial progression and the initial brain-directed treatment approach were similar between treatment groups (Cao et al., 2026).
Perhaps one of the most clinically useful observations was the timing. The peak hazard for new brain metastasis occurred at approximately eight months after initiation of systemic therapy in both groups. That is later than the median progression-free survival of first-line treatment, suggesting that brain relapse risk may continue to rise even after extracranial progression has already occurred. This timing supports the idea that sustained CNS surveillance remains important in the first year, even when patients have already shown disease progression elsewhere (Cao et al., 2026).
Immunotherapy Did Not Reduce Brain Metastasis Incidence
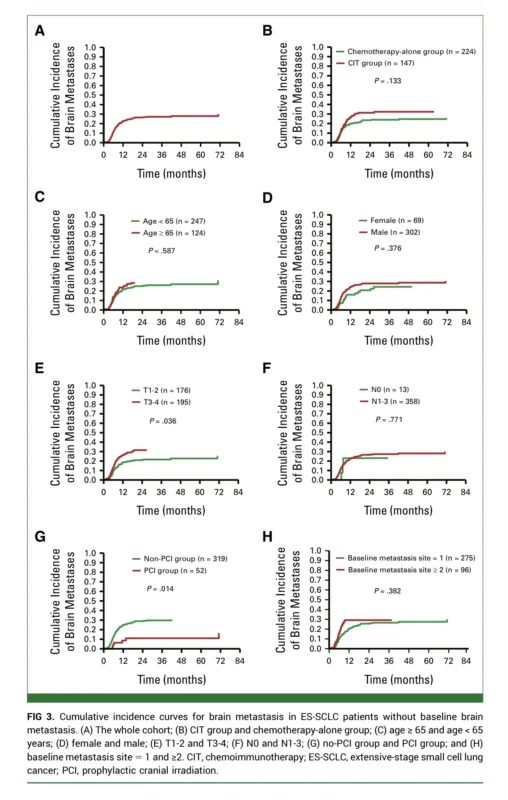
The central result of the study is clear. At one year, the cumulative incidence of brain metastasis was 26.37% in the chemoimmunotherapy group and 19.93% in the chemotherapy-alone group. At two years, the rates were 31.28% and 23.48%, respectively. These differences were not statistically significant, with a Gray’s test P value of .133. In univariable Fine-Gray analysis, chemoimmunotherapy was not associated with reduced brain metastasis risk, and this remained true after multivariable adjustment, where the adjusted subdistribution hazard ratio was 1.255 (95% CI, 0.711 to 2.214; P = .434) (Cao et al., 2026).
That result is important because it argues against a strong protective CNS effect from adding immunotherapy to chemotherapy in this specific setting. The confidence intervals do not rule out smaller clinically relevant effects, but they make a major reduction in brain metastasis risk unlikely. Sensitivity analyses restricted to the modern treatment era and propensity-weighted models gave similar results, which strengthens the credibility of the finding (Cao et al., 2026).
This conclusion aligns with prior randomized evidence. IMpower133 and CASPIAN both improved survival but did not clearly reduce the rate of new brain metastasis in patients without baseline intracranial disease. The present study adds prospective real-world evidence that supports the same overall message: immunotherapy may improve overall disease control and survival, but it does not appear to solve the CNS problem in extensive-stage small cell lung cancer (Horn et al., 2018; Paz-Ares et al., 2019; Chen et al., 2022).
Prophylactic Cranial Irradiation Still Matters
While chemoimmunotherapy did not reduce brain metastasis incidence, prophylactic cranial irradiation did. In multivariable Fine-Gray analysis, PCI was independently associated with a significantly lower risk of brain metastasis, with an adjusted subdistribution hazard ratio of 0.431 (95% CI, 0.195 to 0.955; P = .038) (Cao et al., 2026).
This is one of the most relevant clinical findings in the paper. PCI remains controversial in extensive-stage small cell lung cancer, especially after the Japanese randomized trial showed no overall survival benefit in patients staged with MRI and followed closely with surveillance imaging (Takahashi et al., 2017). Neurocognitive concerns have also made many clinicians more selective about its use. But the present study suggests that, whatever its survival impact, PCI still appears to reduce the risk of subsequent brain metastasis in this population (Cao et al., 2026; Takahashi et al., 2017).
Interestingly, PCI was not associated with significantly improved progression-free or overall survival in this cohort. That does not necessarily weaken its relevance. It instead reinforces the difference between preventing a clinically meaningful site of failure and improving global survival in a disease where systemic progression remains common. The clinical value of PCI may therefore lie in preventing intracranial relapse and preserving neurologic function rather than clearly extending survival on its own.
Survival Improved With Chemoimmunotherapy, But Not Through CNS Protection
The survival data help put these findings into perspective. In the full cohort, chemoimmunotherapy significantly improved overall survival compared with chemotherapy alone, with median overall survival of 16.63 months versus 11.73 months. Progression-free survival, however, was similar between groups. Among patients who did go on to develop brain metastasis, neither overall survival nor progression-free survival differed significantly between treatment groups (Cao et al., 2026).
This suggests that the survival benefit of chemoimmunotherapy in extensive-stage small cell lung cancer is real, but it is not being driven by prevention of brain metastasis. Put differently, systemic benefit does not automatically translate into CNS benefit. That distinction matters in daily practice because it means clinicians should not assume that a stronger first-line systemic regimen is enough to control later intracranial risk.
What This Means for Practice
The practical message from this study is straightforward. Brain metastasis remains a major and early risk in extensive-stage small cell lung cancer even when patients begin treatment without baseline brain involvement. Adding immunotherapy to chemotherapy improves overall survival, but it does not significantly reduce the incidence of later brain metastasis. PCI remains the only evidence-based preventive strategy supported by this analysis, although its use still requires individualized judgment given the lack of a demonstrated survival benefit and concerns about toxicity (Cao et al., 2026).
The study also highlights the importance of surveillance. Because the peak incidence of brain metastasis occurred around eight months after treatment initiation, close CNS imaging during the first year appears especially relevant. Earlier detection could allow treatment while lesions are smaller and asymptomatic, which may improve the feasibility of local approaches such as stereotactic radiosurgery in selected patients (Cao et al., 2026; Aizer et al., 2025).
The Bigger Unanswered Question
The broader implication is that extensive-stage small cell lung cancer still lacks an effective CNS-directed systemic strategy. If immunotherapy improves survival without meaningfully preventing brain metastasis, then the next phase of research needs to focus more directly on the biology of CNS spread, blood-brain barrier penetration, immune privilege within the brain microenvironment, and rational combinations that specifically address intracranial risk (Cao et al., 2026).
This study does not close the debate around PCI, and it does not prove that all patients should receive it. But it does make one point very clear: the CNS remains a major sanctuary site in extensive-stage small cell lung cancer, and current systemic progress has not yet changed that reality.