Triple-negative breast cancer remains one of the most aggressive subtypes of breast cancer, with a high risk of early relapse, rapid progression, and poor long-term survival. In the metastatic setting, the disease has historically been difficult to treat because of its biologic heterogeneity and the lack of hormone receptor or HER2 expression, which limits the use of many targeted therapies. Although the addition of pembrolizumab to chemotherapy improved outcomes for patients with PD-L1–positive disease in KEYNOTE-355, the prognosis for many patients has remained poor, and better first-line options have been urgently needed (Cortes et al., 2020; Cortes et al., 2022).
Now, results from the phase 3 ASCENT-04/KEYNOTE-D19 trial, published in the New England Journal of Medicine, suggest that combining sacituzumab govitecan with pembrolizumab may offer a new first-line standard for patients with previously untreated, PD-L1–positive, locally advanced unresectable or metastatic triple-negative breast cancer (Tolaney et al., 2026).
Read About Triple-Negative Breast Cancer on OncoDaily
Why This Study Matters
Roughly 40% of triple-negative breast cancers are PD-L1 positive, making immunotherapy an important part of treatment for a meaningful proportion of patients. Pembrolizumab plus chemotherapy has been the preferred first-line option in this setting based on KEYNOTE-355, where it improved both progression-free survival and overall survival compared with chemotherapy alone in tumors with a combined positive score of 10 or greater (Cortes et al., 2020; Cortes et al., 2022). However, many patients still progress early, and almost half of patients with metastatic triple-negative breast cancer never receive treatment beyond first line, underscoring the importance of achieving deeper and more durable benefit upfront (Skinner et al., 2021; Punie et al., 2025).
Sacituzumab govitecan is a Trop-2–directed antibody–drug conjugate linked to SN-38, the active metabolite of irinotecan. It has already shown clear benefit in previously treated metastatic triple-negative breast cancer in the ASCENT trial, where it improved progression-free survival and overall survival over chemotherapy (Bardia et al., 2021; Bardia et al., 2024). The key question in ASCENT-04/KEYNOTE-D19 was whether moving sacituzumab govitecan earlier, and pairing it with pembrolizumab, could improve outcomes beyond those seen with standard chemotherapy plus pembrolizumab.
Copyright © 2026 Massachusetts Medical Society.
Study Design
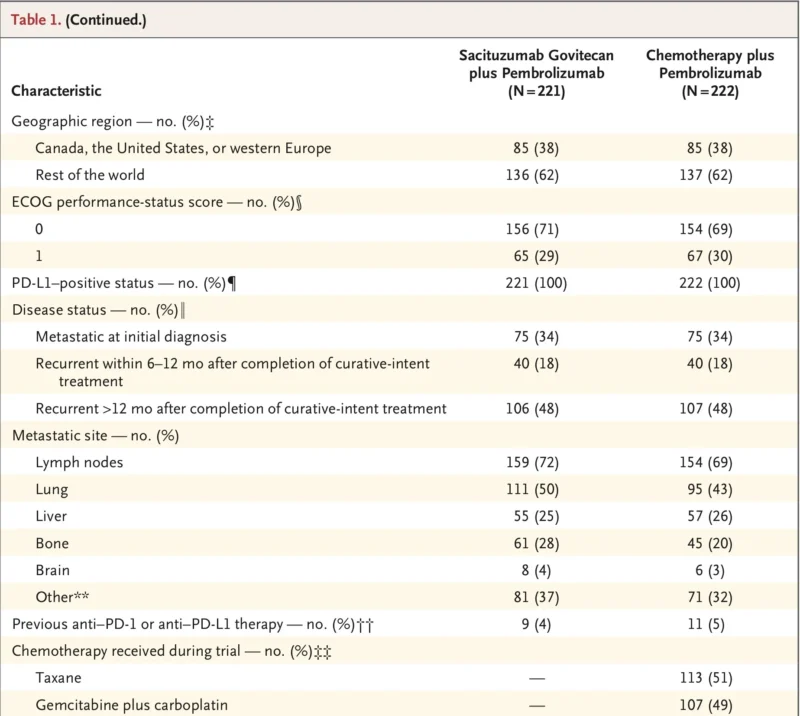
ASCENT-04/KEYNOTE-D19 was an international, open-label, phase 3 trial conducted across 186 sites in 28 countries. The study enrolled adults with previously untreated, PD-L1–positive, locally advanced unresectable or metastatic triple-negative breast cancer. PD-L1 positivity was defined as a combined positive score of at least 10, centrally confirmed using the PD-L1 IHC 22C3 assay. Triple-negative status was also centrally confirmed (Tolaney et al., 2026).
Patients were randomized 1:1 to receive either sacituzumab govitecan plus pembrolizumab or chemotherapy plus pembrolizumab. In the control arm, chemotherapy could be paclitaxel, nab-paclitaxel, or gemcitabine-carboplatin, reflecting real-world first-line options. The primary endpoint was progression-free survival by blinded independent central review. Secondary endpoints included overall survival, objective response, duration of response, and safety (Tolaney et al., 2026).
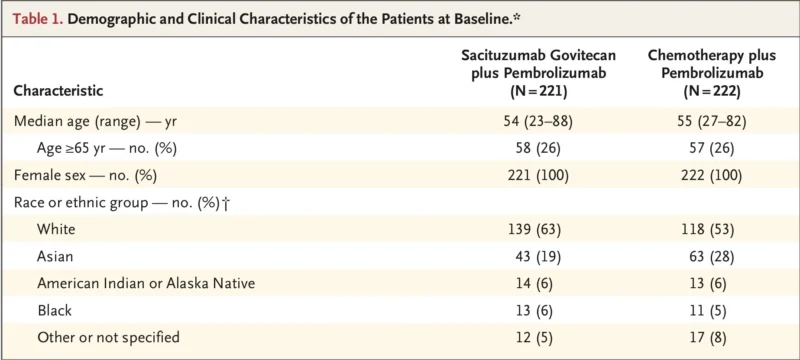
A total of 443 patients were randomized, with 221 assigned to sacituzumab govitecan plus pembrolizumab and 222 assigned to chemotherapy plus pembrolizumab. Baseline characteristics were generally balanced between the two groups. Median age was 54 to 55 years, and about one-third of patients had metastatic disease at initial diagnosis. Nearly half had recurrent disease more than 12 months after curative-intent treatment (Tolaney et al., 2026).
Progression-Free Survival: A Clear Win for the Antibody–Drug Conjugate Combination
The central finding of the trial was a statistically significant improvement in progression-free survival with sacituzumab govitecan plus pembrolizumab.
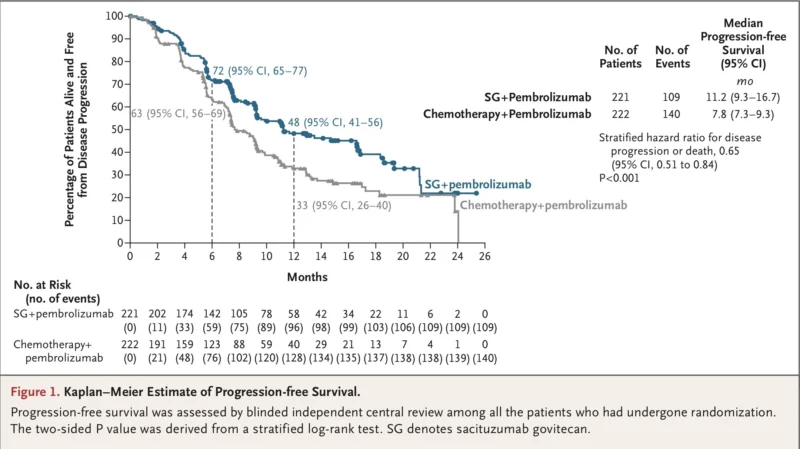
Median progression-free survival was 11.2 months with sacituzumab govitecan plus pembrolizumab compared with 7.8 months with chemotherapy plus pembrolizumab. This translated into a 35% reduction in the risk of disease progression or death, with a hazard ratio of 0.65 (95% CI, 0.51 to 0.84; P<0.001) (Tolaney et al., 2026).
This is clinically meaningful. In a disease where the first-line setting may be the best chance to meaningfully alter trajectory, extending median progression-free survival by more than three months is important. It is even more notable because the control arm already included pembrolizumab, a regimen that had previously set the standard in this population (Cortes et al., 2022).
The benefit was also supported by investigator-assessed progression-free survival, which was 11.3 months with sacituzumab govitecan plus pembrolizumab versus 8.3 months with chemotherapy plus pembrolizumab, with a hazard ratio of 0.67 (Tolaney et al., 2026).
Subgroup analyses suggested that the progression-free survival benefit was broadly consistent across clinically relevant groups, including age, performance status, geographic region, menopausal status, and disease recurrence pattern. Encouragingly, benefit also appeared to extend to harder-to-treat populations, including patients with liver metastases and those with recurrence within 6 to 12 months after curative-intent treatment (Tolaney et al., 2026).
Copyright © 2026 Massachusetts Medical Society.
Response Rate and Duration of Response
Although formal hypothesis testing was not conducted for response endpoints at this interim stage, the efficacy pattern still favored the sacituzumab govitecan combination.
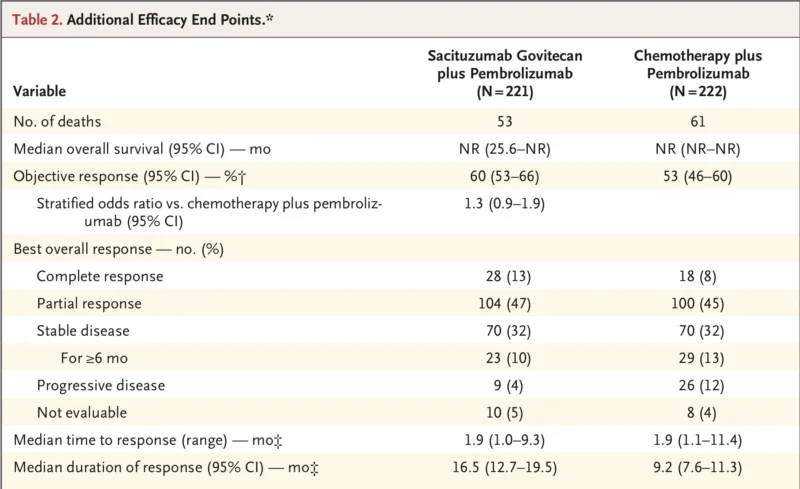
The objective response rate was 60% with sacituzumab govitecan plus pembrolizumab compared with 53% with chemotherapy plus pembrolizumab. Complete responses occurred in 13% versus 8% of patients, respectively (Tolaney et al., 2026).
What stands out even more than the response rate is the durability of response. Among patients who responded, median duration of response was 16.5 months with sacituzumab govitecan plus pembrolizumab, compared with 9.2 months with chemotherapy plus pembrolizumab (Tolaney et al., 2026).
That difference is substantial. In metastatic triple-negative breast cancer, response depth matters, but response duration often matters more. A regimen that can hold disease control longer may help delay symptomatic progression, reduce the need for therapy changes, and preserve quality of life for a longer period.
Overall Survival: Not Mature Yet
At the time of the primary analysis, overall survival data were still immature. Only about 26% of the total study population had died, and the median overall survival had not yet been reached in either group (Tolaney et al., 2026).
This means the study cannot yet claim an overall survival advantage. That is an important limitation, especially in an era where crossover and post-protocol treatments can confound long-term analyses. In fact, many patients in the chemotherapy plus pembrolizumab arm later received sacituzumab govitecan after progression, either through the trial crossover phase or commercially, which may dilute a survival difference over time (Tolaney et al., 2026).
Still, the progression-free survival benefit, combined with longer duration of response and lower discontinuation rates, already supports the clinical relevance of the regimen.
Copyright © 2026 Massachusetts Medical Society.
Safety and Tolerability
Safety was closely watched, especially because this regimen combines immunotherapy with an antibody–drug conjugate rather than standard chemotherapy.
Grade 3 or higher adverse events occurred in 71% of patients receiving sacituzumab govitecan plus pembrolizumab and 70% of patients receiving chemotherapy plus pembrolizumab. On the surface, these rates look similar. However, an important difference emerged in treatment discontinuation. Discontinuation due to adverse events occurred in only 12% of patients in the sacituzumab govitecan arm compared with 31% in the chemotherapy arm (Tolaney et al., 2026).
This is a major point. The toxicity burden may be numerically similar at the high-grade level, but it appears that adverse events with sacituzumab govitecan plus pembrolizumab were more manageable, allowing more patients to stay on therapy.
The most common adverse events with sacituzumab govitecan plus pembrolizumab were diarrhea, nausea, and neutropenia. The most common grade 3 or higher events were neutropenia, diarrhea, and fatigue. In the chemotherapy arm, common toxicities included neutropenia, fatigue, and anemia, with more thrombocytopenia and peripheral neuropathy than in the sacituzumab govitecan group (Tolaney et al., 2026).
Deaths due to adverse events occurred in 3% of patients in each arm, showing that serious toxicity remains a concern regardless of regimen in this fragile disease setting (Tolaney et al., 2026).
Copyright © 2026 Massachusetts Medical Society.
How Does This Fit Into the Current Treatment Landscape?
These results are important because they move sacituzumab govitecan from a later-line rescue strategy into the frontline setting for a biologically selected population. Until now, sacituzumab govitecan has been firmly established in previously treated metastatic triple-negative breast cancer based on ASCENT (Bardia et al., 2021). ASCENT-04/KEYNOTE-D19 suggests that the drug may have even greater impact when used earlier, before resistance accumulates and before patients clinically deteriorate.
This trial also supports a larger trend in breast oncology: antibody–drug conjugates are no longer just salvage therapies. They are becoming legitimate platforms for combination strategies, including with checkpoint inhibitors. Preclinical and translational interest in combining antibody–drug conjugates with immunotherapy has grown over recent years, and this study provides some of the strongest clinical evidence yet that such a strategy can translate into real benefit (Nicolò et al., 2022).
Final Thoughts
ASCENT-04/KEYNOTE-D19 delivers one of the most important metastatic triple-negative breast cancer updates of the year. In patients with previously untreated PD-L1–positive advanced triple-negative breast cancer, sacituzumab govitecan plus pembrolizumab significantly improved progression-free survival over chemotherapy plus pembrolizumab, with a median of 11.2 months versus 7.8 months and a hazard ratio of 0.65 (Tolaney et al., 2026).
The regimen also produced a numerically higher response rate, a much longer duration of response, and lower treatment discontinuation due to adverse events. While overall survival remains immature, the data strongly suggest that this combination may redefine first-line treatment in this setting.
For a disease where early control matters enormously, and where many patients may never reach a second-line treatment, that matters.
Read full article here


